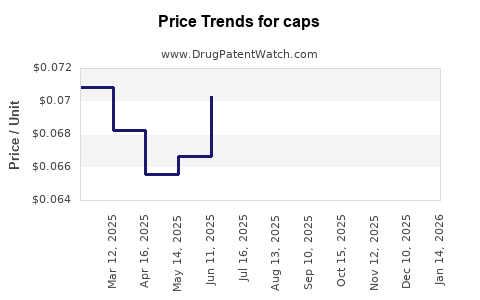
Caps - Generic Drug Details
✉ Email this page to a colleague
US Patents and Regulatory Information for caps
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Averitas | QUTENZA | capsaicin | PATCH;TOPICAL | 022395-001 | Nov 16, 2009 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Averitas | QUTENZA | capsaicin | PATCH;TOPICAL | 022395-001 | Nov 16, 2009 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Averitas | QUTENZA | capsaicin | PATCH;TOPICAL | 022395-001 | Nov 16, 2009 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |