Market Dynamics and Financial Trajectory for Benicar (Olmesartan)
Introduction to Benicar (Olmesartan)
Benicar, also known by its generic name olmesartan, is an antihypertensive medication developed and marketed by Daiichi Sankyo. It is used to treat high blood pressure and has been a significant player in the pharmaceutical market.
Market Drivers
The market for Benicar is driven by several key factors:
Rising Prevalence of Hypertension
The increasing prevalence of hypertension and related cardiovascular conditions is a major driver. As lifestyles become more sedentary and stress levels rise, the demand for effective antihypertensive medications like Benicar continues to grow[1].
Emphasis on Preventive Care
There is a growing emphasis on cardiovascular risk reduction and preventive care. Both physicians and patients recognize the importance of maintaining healthy blood pressure levels to prevent heart attacks, strokes, and other cardiovascular events, leading to increased prescriptions for Benicar[1].
Personalized Medicine
The trend towards personalized medicine is also impacting the market. Tailored treatment plans that include medications like Benicar are becoming more common, contributing to its market presence[1].
Generic Versions and Cost-Efficiency
The introduction of generic versions of olmesartan has made the medication more cost-efficient and accessible to patients. This shift has driven market growth by making the treatment more affordable[1][4].
Financial Performance of Benicar
Peak Revenue Years
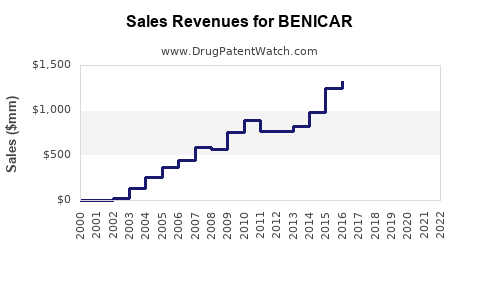
Benicar was one of Daiichi Sankyo's mainstay products, generating significant revenue. In the 2013 fiscal year, it earned $3.1 billion for the company, and in 2014, it generated $2.4 billion. However, revenue began to decline as the patent expired and generic versions entered the market[4].
Decline Post-Patent Expiry
In 2016, Daiichi Sankyo lost exclusive rights to Benicar in the U.S., and generic versions started to dominate the market. This led to a substantial decline in revenue from Benicar. By 2016, the annual revenue from the drug had dropped to $661 million, a significant reduction from its peak years[4].
Impact of Generic Competition
The entry of generic versions of olmesartan into the market had a profound impact on Daiichi Sankyo's revenue. It was estimated that generic versions would account for $950 million in sales in the 2016-2017 fiscal year, further eroding the company's market share and revenue from the drug[4].
Financial Trajectory Post-2016
Quarterly and Annual Revenue
In the financial reports for 2016, it was noted that the revenue from Benicar/Benicar HCT in the U.S. was $79.5 billion for the year, with a slight increase in some quarters but an overall decline compared to previous years[2].
Revenue Comparison
The revenue from Benicar in different regions showed varying trends. For instance, in Japan, the revenue from Olmetec (the Japanese brand name for olmesartan) declined, while in the U.S., the revenue from Benicar/Benicar HCT showed some stability but was ultimately on a downward trend due to generic competition[2].
Legal and Regulatory Challenges
Litigation and Settlements
Benicar has been involved in several lawsuits related to its side effects, which have further impacted Daiichi Sankyo's financials. In 2017, the company spent $300 million to settle lawsuits related to Benicar, adding to the financial strain[5].
Strategic Response by Daiichi Sankyo
Focus on New Products and Markets
To address the decline in revenue from Benicar, Daiichi Sankyo has been focusing on developing new drugs and expanding into other markets, particularly in the specialty area of oncology. The company aims to center its business around oncology treatments by 2025, recognizing the large and growing market in this area[4].
U.S. Market Growth
Despite challenges, the U.S. market remains a key target for growth for Daiichi Sankyo. The company sees ongoing stable growth in the U.S. pharmaceutical market, despite pressures on pricing and insurance coverage[4].
Key Takeaways
- The market for Benicar is driven by the rising prevalence of hypertension, emphasis on preventive care, personalized medicine, and the availability of generic versions.
- The financial performance of Benicar peaked in the early 2010s but declined significantly after the patent expiry and the introduction of generic versions.
- Daiichi Sankyo has faced legal challenges related to Benicar and has been investing in new product development and market expansion, particularly in oncology.
- The U.S. market remains crucial for Daiichi Sankyo’s growth strategy despite the challenges posed by generic competition and regulatory pressures.
FAQs
What is Benicar used for?
Benicar, or olmesartan, is used to treat high blood pressure and reduce the risk of cardiovascular events such as heart attacks and strokes.
Why did the revenue from Benicar decline?
The revenue from Benicar declined significantly after the patent expired and generic versions of the drug entered the market, reducing Daiichi Sankyo's exclusive rights and market share.
How has Daiichi Sankyo responded to the decline in Benicar revenue?
Daiichi Sankyo has focused on developing new drugs, particularly in the oncology sector, and is targeting growth in the U.S. pharmaceutical market despite the challenges.
What were the financial impacts of the lawsuits related to Benicar?
In 2017, Daiichi Sankyo spent $300 million to settle lawsuits related to Benicar, adding to the financial strain on the company.
What is Daiichi Sankyo's long-term strategy?
Daiichi Sankyo aims to center its business around oncology treatments by 2025 and continues to target growth in the U.S. pharmaceutical market.
Sources
- IMARC Group - Olmesartan (Benicar) Manufacturing Plant Project Report 2024
- Daiichi Sankyo - Reference Data
- CVS Health - Driving More Affordable, Accessible and Effective Care
- DrugWatch - Daiichi Sankyo - Benicar Manufacturer, History, Products
- BioPharma Dive - Daiichi spends $300M to settle Benicar suits