COTELLIC Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cotellic, and what generic alternatives are available?
Cotellic is a drug marketed by Genentech Inc and is included in one NDA. There are seven patents protecting this drug.
This drug has one hundred and eighty-four patent family members in forty-three countries.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this compound. Additional details are available on the cobimetinib fumarate profile page.
DrugPatentWatch® Generic Entry Outlook for Cotellic
Cotellic was eligible for patent challenges on November 10, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 30, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for COTELLIC?
- What are the global sales for COTELLIC?
- What is Average Wholesale Price for COTELLIC?
Summary for COTELLIC
| International Patents: | 184 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 10 |
| Clinical Trials: | 35 |
| Patent Applications: | 1 |
| Drug Prices: | Drug price information for COTELLIC |
| What excipients (inactive ingredients) are in COTELLIC? | COTELLIC excipients list |
| DailyMed Link: | COTELLIC at DailyMed |
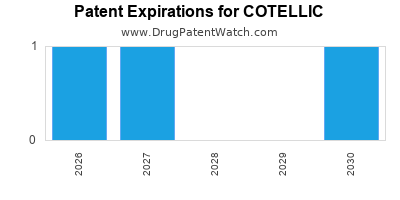

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for COTELLIC
Generic Entry Date for COTELLIC*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for COTELLIC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Birmingham | Phase 2/Phase 3 |
| Cancer Research UK | Phase 2/Phase 3 |
| Royal Marsden NHS Foundation Trust | Phase 2/Phase 3 |
Pharmacology for COTELLIC
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Kinase Inhibitors |
US Patents and Regulatory Information for COTELLIC
COTELLIC is protected by seven US patents and four FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of COTELLIC is ⤷ Subscribe.
This potential generic entry date is based on patent 10,590,102.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | 11,597,699 | ⤷ Subscribe | ⤷ Subscribe | ||||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | 11,087,354*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | 11,254,649*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for COTELLIC
When does loss-of-exclusivity occur for COTELLIC?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5483
Patent: SAL DE FUMARATO CRISTALINA DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-YODOFENILAMINO)FENIL][3-HIDROXI-3-(PIPERIDIN-2-IL)AZETIDIN-1-IL]-METANONA
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 16288209
Patent: Crystalline fumarate salt of (S)-(3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl) (3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl)-methanone
Estimated Expiration: ⤷ Subscribe
Patent: 21200202
Patent: CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 2017028516
Patent: sal de fumarato cristalino de (s)-[3,4-difluoro-2-(2-fluoro-4-iodofenilamino)fenil][3-hidroxi-3-(piperidin-2-il) azetidin-1-il]-metanona
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 90222
Patent: SEL CRISTALLIN DE FUMARATE DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL][3-HYDROXY-3-(PIPERIDIN-2-YL)AZETIDIN-1-YL]-METHANONE (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE)
Estimated Expiration: ⤷ Subscribe
Chile
Patent: 17003475
Patent: Sal de fumarato cristalina de (s)-[3,4-difluoro-2-(2-fluoro-4-yodofenilamino)fenil][3-hidroxi-3-(piperidin-2-il)azetidin-1-il]-metanona
Estimated Expiration: ⤷ Subscribe
China
Patent: 7810183
Estimated Expiration: ⤷ Subscribe
Patent: 8290395
Patent: 结晶反丁烯二酸盐 (Crystalline fumarate salts)
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 18000086
Patent: Sal de fumarato cristalina de (s)-[3,4-difluoro-2-(2-fluoro-4-yodofenilamino)fenil][3-hidroxi-3-(piperidin-2-il)azetidin-1-il]-metanona
Estimated Expiration: ⤷ Subscribe
Costa Rica
Patent: 180056
Patent: SAL DE FUMARATO CRISTALINA DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-YODOFENILAMINO)FENIL] [3-HIDROXI-3-(PIPERIDIN-2-IL)AZETIDIN-1-IL]-METANONA
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 17264
Patent: SEL CRISTALLIN DE FUMARATE DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHÉNYLAMINO)PHÉNYL][3-HYDROXY-3-(PIPÉRIDIN-2-YL)AZÉTIDIN-1-YL]-MÉTHANONE (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL][3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE)
Estimated Expiration: ⤷ Subscribe
Hong Kong
Patent: 52433
Estimated Expiration: ⤷ Subscribe
Israel
Patent: 6423
Patent: מלח פומראט גבישי של (s (-[4,3-דיפלואורו-2-(2-פלואורו-4-יודופנילאמינו)פניל] [3-הידרוקסי-3-(פיפרידין-2-איל)אזטידין-1-איל]-מתאנון (Crystalline fumarate salt of (s)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]-methanone)
Estimated Expiration: ⤷ Subscribe
Patent: 5052
Patent: מלח פומראט גבישי של (s)-[4,3-דיפלואורו-2-(2-פלואורו-4-יודופנילאמינו)פניל] [3-הידרוקסי-3-(פיפרידין-2-איל)אזטידין-1-איל]-מתאנון (Crystalline fumarate salt of (s)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]-methanone)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 38950
Estimated Expiration: ⤷ Subscribe
Patent: 18519318
Patent: (S)−[3,4−ジフルオロ−2−(2−フルオロ−4−ヨードフェニルアミノ)フェニル][3−ヒドロキシ−3−(ピペリジン−2−イル」)アゼチジン−1−イル]−メタノンの結晶性フマル酸塩
Estimated Expiration: ⤷ Subscribe
Patent: 21035967
Patent: (S)−[3,4−ジフルオロ−2−(2−フルオロ−4−ヨードフェニルアミノ)フェニル][3−ヒドロキシ−3−(ピペリジン−2−イル)アゼチジン−1−イル]−メタノンの結晶性フマル酸塩 (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE)
Estimated Expiration: ⤷ Subscribe
Patent: 23025000
Patent: (S)-[3,4-ジフルオロ-2-(2-フルオロ-4-ヨードフェニルアミノ)フェニル][3-ヒドロキシ-3-(ピペリジン-2-イル)アゼチジン-1-イル]-メタノンの結晶性フマル酸塩
Estimated Expiration: ⤷ Subscribe
Malaysia
Patent: 2545
Patent: CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 17017037
Patent: SAL DE FUMARATO CRISTALINA DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-YODO FENILAMINO) FENIL][3-HIDROXI-3-(PIPERIDIN-2-IL]AZETIDIN-1-IL]-META NONA. (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODO PHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE.)
Estimated Expiration: ⤷ Subscribe
Morocco
Patent: 301
Patent: SEL CRISTALLIN DE FUMARATE DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHÉNYLAMINO)PHÉNYL][3-HYDROXY-3-(PIPÉRIDIN-2-YL)AZÉTIDIN-1-YL]-MÉTHANONE
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 180692
Patent: SAL DE FUMARATO CRISTALINA DE (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-YODOFENILAMINO)FENIL][3-HIDROXI-3-(PIPERIDIN-2-IL)AZETIDIN-1-IL]-METANONA
Estimated Expiration: ⤷ Subscribe
Philippines
Patent: 017502414
Patent: CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE
Estimated Expiration: ⤷ Subscribe
Russian Federation
Patent: 62181
Patent: КРИСТАЛЛИЧЕСКАЯ ФУМАРАТНАЯ СОЛЬ (S)-[3,4-ДИФТОР-2-(2-ФТОР-4-ЙОДФЕНИЛАМИНО)ФЕНИЛ][3-ГИДРОКСИ-3-(ПИПЕРИДИН-2-ИЛ)АЗЕТИДИН-1-ИЛ]МЕТАНОНА (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL][3-HYDROXY-3-(PIPERIDINE-2-YL)AZETIDINE-1-YL]METHANONE)
Estimated Expiration: ⤷ Subscribe
Patent: 18103172
Patent: КРИСТАЛЛИЧЕСКАЯ ФУМАРАТНАЯ СОЛЬ (S)-[3,4-ДИФТОР-2-(2-ФТОР-4-ЙОДФЕНИЛАМИНО)ФЕНИЛ][3-ГИДРОКСИ-3-(ПИПЕРИДИН-2-ИЛ)АЗЕТИДИН-1-ИЛ]МЕТАНОНА
Estimated Expiration: ⤷ Subscribe
Patent: 21132394
Patent: КРИСТАЛЛИЧЕСКАЯ ФУМАРАТНАЯ СОЛЬ (S)-[3,4-ДИФТОР-2-(2-ФТОР-4-ЙОДФЕНИЛАМИНО)ФЕНИЛ][3-ГИДРОКСИ-3-(ПИПЕРИДИН-2-ИЛ)АЗЕТИДИН-1-ИЛ]МЕТАНОНА
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 202105196Y
Patent: CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 1708760
Patent: CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 2695324
Estimated Expiration: ⤷ Subscribe
Patent: 180021775
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 10556
Estimated Expiration: ⤷ Subscribe
Patent: 75187
Estimated Expiration: ⤷ Subscribe
Patent: 1718535
Patent: Crystalline fumarate salt of (S)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]-methanone
Estimated Expiration: ⤷ Subscribe
Patent: 2108568
Patent: Crystalline fumarate salt of (S)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]-methanone
Estimated Expiration: ⤷ Subscribe
Ukraine
Patent: 4728
Patent: КРИСТАЛІЧНА ФУМАРАТНА СІЛЬ (S)-[3,4-ДИФТОР-2-(2-ФТОР-4-ЙОДФЕНІЛАМІНО)ФЕНІЛ][3-ГІДРОКСИ-3-(ПІПЕРИДИН-2-ІЛ)АЗЕТИДИН-1-ІЛ]МЕТАНОНУ (CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE)
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering COTELLIC around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Mexico | 2020013162 | COMPRIMIDOS DE LIBERACION INMEDIATA QUE CONTIENEN UN FARMACO Y PROCESOS PARA FORMAR LOS COMPRIMIDOS. (IMMEDIATE-RELEASE TABLETS CONTAINING A DRUG AND PROCESSES FOR FORMING THE TABLETS.) | ⤷ Subscribe |
| Eurasian Patent Organization | 019983 | ИНГИБИТОРЫ MEK И СПОСОБЫ ИХ ПРИМЕНЕНИЯ (MEK INHIBITORS AND METHODS OF USING SAME) | ⤷ Subscribe |
| China | 110251512 | 包括给药COBIMETINIB和威罗菲尼的黑色素瘤的组合疗法 (Combination therapies for melanoma comprising administering cobimetinib and vemurafinib) | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for COTELLIC
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1934174 | C01934174/01 | Switzerland | ⤷ Subscribe | PRODUCT NAME: COBIMETINIB; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 65620 24.08.2015 |
| 1934174 | 22/2016 | Austria | ⤷ Subscribe | PRODUCT NAME: COBIMETINIB ALS EIN EINZELNES STEREOISOMER ODER ALS EINE MISCHUNG VON STEREOISOMEREN DAVON, EINSCHLIESSLICH PHARMAZEUTISCH ANNEHMBARER SALZE UND SOLVATE, INSBESONDERE COBIMETINIB HEMIFUMARAT; REGISTRATION NO/DATE: EU/1/15/1048 (MITTEILUNG) 20151124 |
| 1934174 | 122016000033 | Germany | ⤷ Subscribe | PRODUCT NAME: COBIMETINIB, OPTIONAL ALS PHARMAZEUTISCH ZULAESSIGES SALZ ODER SOLVAT DAVON, INSBESONDERE COBIMETINIB-HEMIFUMARAT; REGISTRATION NO/DATE: EU/1/15/1048 20151120 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
COTELLIC Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


