JENTADUETO Drug Patent Profile
✉ Email this page to a colleague
When do Jentadueto patents expire, and what generic alternatives are available?
Jentadueto is a drug marketed by Boehringer Ingelheim and is included in two NDAs. There are eleven patents protecting this drug and one Paragraph IV challenge.
This drug has three hundred and seventy-three patent family members in forty-five countries.
The generic ingredient in JENTADUETO is linagliptin; metformin hydrochloride. There are nineteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the linagliptin; metformin hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Jentadueto
A generic version of JENTADUETO was approved as linagliptin; metformin hydrochloride by SUNSHINE on August 30th, 2021.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for JENTADUETO?
- What are the global sales for JENTADUETO?
- What is Average Wholesale Price for JENTADUETO?
Summary for JENTADUETO
| International Patents: | 373 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 3 |
| Clinical Trials: | 1 |
| Patent Applications: | 34 |
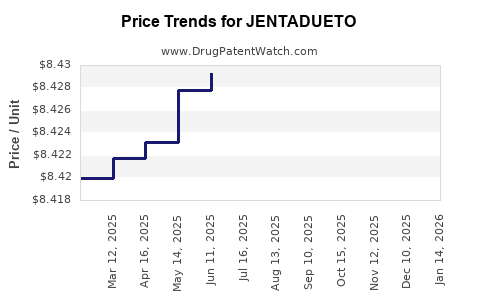
| Drug Prices: | Drug price information for JENTADUETO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for JENTADUETO |
| What excipients (inactive ingredients) are in JENTADUETO? | JENTADUETO excipients list |
| DailyMed Link: | JENTADUETO at DailyMed |
Recent Clinical Trials for JENTADUETO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Boehringer Ingelheim | Phase 1 |
Pharmacology for JENTADUETO
| Drug Class | Biguanide Dipeptidyl Peptidase 4 Inhibitor |
| Mechanism of Action | Dipeptidyl Peptidase 4 Inhibitors |
Paragraph IV (Patent) Challenges for JENTADUETO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| JENTADUETO | Tablets | linagliptin; metformin hydrochloride | 2.5 mg/500 mg 2.5 mg/850 mg 2.5 mg/1000 mg | 201281 | 8 | 2015-05-04 |
US Patents and Regulatory Information for JENTADUETO
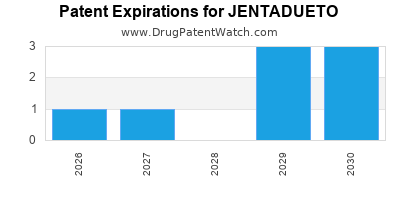
JENTADUETO is protected by nine US patents and two FDA Regulatory Exclusivities.
Expired US Patents for JENTADUETO
EU/EMA Drug Approvals for JENTADUETO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Jentadueto | linagliptin, metformin hydrochloride | EMEA/H/C/002279 Treatment of adult patients with type-2 diabetes mellitus:Jentadueto is indicated as an adjunct to diet and exercise to improve glycaemic control in adult patients inadequately controlled on their maximal tolerated dose of metformin alone, or those already being treated with the combination of linagliptin and metformin.Jentadueto is indicated in combination with a sulphonylurea (i.e. triple combination therapy) as an adjunct to diet and exercise in adult patients inadequately controlled on their maximal tolerated dose of metformin and a sulphonylurea. |
Authorised | no | no | no | 2012-07-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for JENTADUETO
When does loss-of-exclusivity occur for JENTADUETO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1175
Patent: COMPOSICION FARMACEUTICA QUE COMPRENDE UN INHIBIDOR DE LA DIPEPTIDIL-PEPTIDASA-4 (DPP4) Y UN FARMACO ACOMPANANTE
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 09232043
Patent: DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 0911273
Patent: inibidor de dpp-iv combinado com outro agente antidiabético, comprimidos que compreendem tais formulações, seu uso e processo para sua preparação
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 20450
Patent: NOUVELLES FORMULATIONS, COMPRIMES COMPRENANT DE TELLES FORMULATIONS, LEUR UTILISATION ET LEUR PROCEDE DE PREPARATION (NEW FORMULATIONS, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Chile
Patent: 09000809
Patent: Composicion farmaceutica que comprende un inhibidor de la dpp4 1-[(4-metil-quinazolin-2-il)metil]-3-metil-7-(2-butin-1-il)-8-(3-(r)-amino-piperidin-1-il)-xantina y metformina o sus sales, junto a l-arginina para estabilizar al inhibidor de dpp4; procedimiento de preparacion de dicha composicion.
Estimated Expiration: ⤷ Subscribe
China
Patent: 1983073
Patent: Dpp-iv inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Patent: 3083672
Patent: DPP-IV inhibitor combined with further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Patent: 6215190
Patent: 与其它抗糖尿病药组合的DPP‑IV抑制剂、包含此类制剂的片剂,和其用途和制备方法 (Dpp-iv inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation)
Estimated Expiration: ⤷ Subscribe
Patent: 3648422
Patent: 与其它抗糖尿病药组合的DPP-IV抑制剂、包含此类制剂的片剂,和其用途和制备方法 (DPP-IV inhibitors in combination with other anti-diabetic agents, tablets comprising such formulations, and use and processes for preparation thereof)
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 51277
Patent: INHIBIDOR DE LA DPP-IV COMBINADO CON MAS DE UN AGENTE ANTIDIABETICO, COMPRIMIDOS QUE COMPRENDEN TALES FORMULACIONES, SU USO Y PROCEDIMIENTOS PARA SU PREPARACIÓN.
Estimated Expiration: ⤷ Subscribe
Denmark
Patent: 85410
Estimated Expiration: ⤷ Subscribe
Ecuador
Patent: 10010489
Patent: Nuevas formulaciones, comprimidos que comprenden tales formulaciones, su uso y procedimiento para su preparación
Estimated Expiration: ⤷ Subscribe
Eurasian Patent Organization
Patent: 9395
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, ВКЛЮЧАЮЩАЯ ИНГИБИТОР ДПП-4 И МЕТФОРМИН, СПОСОБ ЕЕ ПОЛУЧЕНИЯ И ТВЕРДАЯ ДОЗИРОВАННАЯ ФОРМА, ВКЛЮЧАЮЩАЯ УКАЗАННУЮ КОМПОЗИЦИЮ (PHARMACEUTICAL COMPOSITION COMPRISING A DPP-4 INHIBITOR AND METFORMIN, PROCESS FOR THE PREPARATION THEREOF AND SOLID DOSAGE FORM COMPRISING SAID COMPOSITION)
Estimated Expiration: ⤷ Subscribe
Patent: 8435
Patent: ПРИМЕНЕНИЕ ФАРМАЦЕВТИЧЕСКОЙ КОМПОЗИЦИИ В ВИДЕ ТАБЛЕТКИ С ПЛЕНОЧНЫМ ПОКРЫТИЕМ ДЛЯ ЛЕЧЕНИЯ САХАРНОГО ДИАБЕТА 2 ТИПА (USE OF A PHARMACEUTICAL COMPOSITION IN THE FORM OF A FILM-COATED TABLET IN TREATING TYPE 2 DIABETES MELLITUS)
Estimated Expiration: ⤷ Subscribe
Patent: 1001577
Patent: ИНГИБИТОР ДПП-4 В КОМБИНАЦИИ С ДОПОЛНИТЕЛЬНЫМ ПРОТИВОДИАБЕТИЧЕСКИМ АГЕНТОМ, ТАБЛЕТКИ, ВКЛЮЧАЮЩИЕ УКАЗАННЫЕ КОМПОЗИЦИИ, ИХ ПРИМЕНЕНИЕ И СПОСОБ ИХ ПОЛУЧЕНИЯ
Estimated Expiration: ⤷ Subscribe
Patent: 1300121
Patent: ИНГИБИТОР ДПП-4 В КОМБИНАЦИИ С ДОПОЛНИТЕЛЬНЫМ ПРОТИВОДИАБЕТИЧЕСКИМ АГЕНТОМ, ТАБЛЕТКИ, ВКЛЮЧАЮЩИЕ УКАЗАННЫЕ КОМПОЗИЦИИ, ИХ ПРИМЕНЕНИЕ И СПОСОБ ИХ ПОЛУЧЕНИЯ
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 85410
Patent: DPP-IV INHIBITEUR COMBINÉ AVEC UN AUTRE AGENT ANTI-DIABÉTIQUE, COMPRIMÉS COMPRENANT LESDITES FORMULATIONS, LEUR UTILISATION ET LEUR PROCÉDÉ DE PRÉPARATION (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Patent: 53403
Patent: INHIBITEUR DPP-IV COMBINÉ À UN AUTRE AGENT ANTIDIABÉTIQUE, COMPRIMÉS COMPRENANT DE TELLES FORMULATIONS, LEUR UTILISATION ET LEUR PROCÉDÉ DE PRÉPARATION (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Patent: 44374
Patent: INHIBITEUR DE DPP-IV COMBINÉ À UN AUTRE AGENT ANTIDIABÉTIQUE, COMPRIMÉS COMPRENANT DE TELLES FORMULATIONS, LEUR UTILISATION ET LEUR PROCÉDÉ DE PRÉPARATION (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Hong Kong
Patent: 49485
Patent: DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION
Estimated Expiration: ⤷ Subscribe
Hungary
Patent: 41649
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 88428
Estimated Expiration: ⤷ Subscribe
Patent: 22068
Estimated Expiration: ⤷ Subscribe
Patent: 11516456
Estimated Expiration: ⤷ Subscribe
Patent: 13237707
Patent: NEW FORMULATION, TABLET COMPRISING THE FORMULATION, THE USE AND PROCESS FOR THE PREPARATION
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 1232
Patent: INHIBIDOR DE DPP-IV COMBINADO CON OTRO AGENTE ANTIDIABETICO, TABLETAS QUE COMPRENDEN TALES FORMULACIONES, SU USO Y PROCEDIMIENTO PARA SU PREPARACION. (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION.)
Estimated Expiration: ⤷ Subscribe
Patent: 10010819
Patent: INHIBIDORES DE DPP-IV COMBINADO CON OTRO AGENTE ANTIDIABETICO, TABLETAS QUE COMPRENDEN ESAS FORMULACIONES, SU USO Y PROCEDIMIENTO PARA SU PREPARACION. (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION.)
Estimated Expiration: ⤷ Subscribe
Morocco
Patent: 200
Patent: صياغات جديدة، وأقراص تتكون من هذه الصيغ، استخدامها وطريقة تحضيرها
Estimated Expiration: ⤷ Subscribe
New Zealand
Patent: 7747
Patent: Combination of 1-[(methyl-quinazolin-2-yl)methyl]-3-methyl-7-(2-butyn-1-yl)-8-(piperidin-1-yl)-xanthine (BI-1356), metformin and a stabilising agent
Estimated Expiration: ⤷ Subscribe
Patent: 9580
Patent: Dpp-iv inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 091730
Patent: FORMULACIONES QUE COMPRENDEN UN INHIBIDOR DE DPP4
Estimated Expiration: ⤷ Subscribe
Patent: 140960
Patent: FORMULACIONES QUE COMPRENDEN UN INHIBIDOR DE DPP4
Estimated Expiration: ⤷ Subscribe
Poland
Patent: 85410
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 1005664
Patent: DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 1611314
Estimated Expiration: ⤷ Subscribe
Patent: 1775942
Estimated Expiration: ⤷ Subscribe
Patent: 110005690
Patent: DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION
Estimated Expiration: ⤷ Subscribe
Patent: 160042174
Patent: 추가의 항당뇨병제와 병용된 DPP-IV 억제제, 이러한 제형을 포함하는 정제, 이들의 용도 및 이들의 제조 방법 (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Patent: 170056021
Patent: 추가의 항당뇨병제와 병용된 DPP-IV 억제제, 이러한 제형을 포함하는 정제, 이들의 용도 및 이들의 제조 방법 (- DPP-IV Inhibitor combined with a further antidiabetic agent tablets comprising such formulations their use and process for their preparation)
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 96124
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 27816
Estimated Expiration: ⤷ Subscribe
Patent: 0946534
Patent: New formulations, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Patent: 1509941
Patent: New formulations, tablets comprising such formulations, their use and process for their preparation
Estimated Expiration: ⤷ Subscribe
Tunisia
Patent: 10000431
Patent: NEW FORMULATIONS, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION
Estimated Expiration: ⤷ Subscribe
Turkey
Patent: 1818886
Estimated Expiration: ⤷ Subscribe
Ukraine
Patent: 4136
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, КОТОРАЯ ВКЛЮЧАЕТ ИНГИБИТОР ДПП-4 В КОМБИНАЦИИ С ДОПОЛНИТЕЛЬНЫМ ПРОТИВОДИАБЕТИЧЕСКИМ АГЕНТОМ, И СПОСОБ ЕЕ ПОЛУЧЕНИЯ;ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ, ЩО ВКЛЮЧАЄ ІНГІБІТОР ДПП-4 У КОМБІНАЦІЇ З ДОДАТКОВИМ ПРОТИДІАБЕТИЧНИМ АГЕНТОМ, ТА СПОСІБ ЇЇ ОДЕРЖАННЯ (DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION)
Estimated Expiration: ⤷ Subscribe
Uruguay
Patent: 747
Patent: NUEVAS FORMULACIONES, COMPRIMIDOS QUE COMPRENDEN TALES FORMULACIONES, SU USO Y PROCEDIMIENTO PARA SU PREPARACION
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering JENTADUETO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Portugal | 2015754 | ⤷ Subscribe | |
| New Zealand | 332707 | USE OF DIPEPTIDYL PEPTIDASE IV INHIBITORS (DPIV) FOR LOWERING THE BLOOD GLUCOSE LEVEL IN MAMMALS | ⤷ Subscribe |
| Mexico | 2022001797 | TRATAMIENTO DE LA DIABETES EN PACIENTES CON CONTROL GLUCEMICO INSUFICIENTE A PESAR DE LA TERAPIA CON UN FARMACO ORAL O NO ORAL ANTIDIABETICO. (TREATMENT FOR DIABETES IN PATIENTS WITH INSUFFICIENT GLYCEMIC CONTROL DESPITE THERAPY WITH AN ORAL OR NON-ORAL ANTIDIABETIC DRUG.) | ⤷ Subscribe |
| Australia | 2013251292 | 8-[3-amino-piperidin-1-yl]-xanthines, the production thereof and the use of the same as medicaments | ⤷ Subscribe |
| Canada | 2738367 | TRAITEMENT DU DIABETE CHEZ DES PATIENTS DONT LA REGULATION GLYCEMIQUE EST INSUFFISANTE MALGRE UNE THERAPIE PAR UN MEDICAMENT ANTIDIABETIQUE ORAL OU NON ORAL (TREATMENT FOR DIABETES IN PATIENTS WITH INSUFFICIENT GLYCEMIC CONTROL DESPITE THERAPY WITH AN ORAL OR NON-ORAL ANTIDIABETIC DRUG) | ⤷ Subscribe |
| China | 101983073 | Dpp-iv inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation | ⤷ Subscribe |
| Eurasian Patent Organization | 016752 | ПРОМЕЖУТОЧНЫЕ СОЕДИНЕНИЯ ДЛЯ ПОЛУЧЕНИЯ ХИРАЛЬНЫХ 8-(3-АМИНОПИПЕРИДИН-1-ИЛ)КСАНТИНОВ (INTERMEDIATES FOR PRODUCING CHIRAL 8-(3-AMINOPIPERIDIN-1-YL)XANTHINES) | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for JENTADUETO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1532149 | PA2012022 | Lithuania | ⤷ Subscribe | PRODUCT NAME: LINAGLIPTINUM + METFORMINI HYDROCHLORIDUM; REGISTRATION NO/DATE: EU/1/12/780/001 - EU/1/12/780/034 20120720 |
| 1084705 | CR 2014 00066 | Denmark | ⤷ Subscribe | PRODUCT NAME: ALOGLIPTIN OG FARMACEUTISK SALTE DERAF, HERUNDER ALOGLIPTIN BENZOATE; REG. NO/DATE: EU/1/13/844/001-027 20130923 |
| 1084705 | C300709 | Netherlands | ⤷ Subscribe | PRODUCT NAME: ALOGLIPTIN; REGISTRATION NO/DATE: EU/1/13/844/001-027 20130919 |
| 0896538 | 07C0035 | France | ⤷ Subscribe | PRODUCT NAME: SITAGLIPTIN; REGISTRATION NO/DATE IN FRANCE: EU/1/07/383/001 DU 20070321; REGISTRATION NO/DATE AT EEC: EU/1/07/383/001-018 DU 20070321 |
| 1730131 | C01730131/03 | Switzerland | ⤷ Subscribe | PRODUCT NAME: EMPAGLIFLOZIN UND LINAGLIPTIN; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66132 29.03.2017 |
| 1084705 | CR 2014 00062 | Denmark | ⤷ Subscribe | PRODUCT NAME: SAXAGLIPTIN OG FARMACEUTISK SALTE DERAF, HERUNDER SAXAGLIPTIN HYDROCHLORID; REG. NO/DATE: EU/1/09/545/001-015 20091005 |
| 0896538 | CA 2007 00061 | Denmark | ⤷ Subscribe | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
JENTADUETO Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


