OXYCONTIN Drug Patent Profile
✉ Email this page to a colleague
When do Oxycontin patents expire, and what generic alternatives are available?
Oxycontin is a drug marketed by Purdue Pharma Lp and is included in one NDA. There are eighteen patents protecting this drug and five Paragraph IV challenges.
This drug has three hundred and seventy-two patent family members in forty-eight countries.
The generic ingredient in OXYCONTIN is oxycodone hydrochloride. There are nineteen drug master file entries for this compound. Forty-five suppliers are listed for this compound. Additional details are available on the oxycodone hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Oxycontin
A generic version of OXYCONTIN was approved as oxycodone hydrochloride by SPECGX LLC on June 30th, 2004.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for OXYCONTIN?
- What are the global sales for OXYCONTIN?
- What is Average Wholesale Price for OXYCONTIN?
Summary for OXYCONTIN
| International Patents: | 372 |
| US Patents: | 18 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 3 |
| Raw Ingredient (Bulk) Api Vendors: | 24 |
| Clinical Trials: | 61 |
| Patent Applications: | 3,901 |
| Drug Prices: | Drug price information for OXYCONTIN |
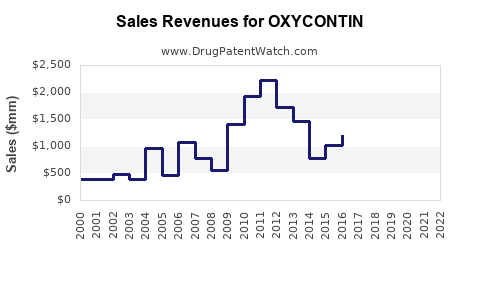
| Drug Sales Revenues: | Drug sales revenues for OXYCONTIN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OXYCONTIN |
| What excipients (inactive ingredients) are in OXYCONTIN? | OXYCONTIN excipients list |
| DailyMed Link: | OXYCONTIN at DailyMed |

Recent Clinical Trials for OXYCONTIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Miami | Phase 2 |
| 3D Medicines | Phase 2 |
| Medical College of Wisconsin | Phase 2 |
Pharmacology for OXYCONTIN
| Drug Class | Opioid Agonist |
| Mechanism of Action | Full Opioid Agonists |
Paragraph IV (Patent) Challenges for OXYCONTIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OXYCONTIN | Extended-release Tablets | oxycodone hydrochloride | 20 mg | 022272 | 2 | 2010-10-29 |
| OXYCONTIN | Extended-release Tablets | oxycodone hydrochloride | 15 mg | 022272 | 1 | 2010-10-28 |
| OXYCONTIN | Extended-release Tablets | oxycodone hydrochloride | 10 mg | 022272 | 1 | 2010-10-25 |
| OXYCONTIN | Extended-release Tablets | oxycodone hydrochloride | 30 mg, 60 mg and 80 mg | 022272 | 1 | 2010-10-18 |
| OXYCONTIN | Extended-release Tablets | oxycodone hydrochloride | 40 mg | 022272 | 1 | 2010-10-04 |
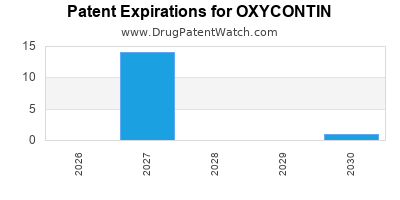
US Patents and Regulatory Information for OXYCONTIN
OXYCONTIN is protected by eighteen US patents.
Patents protecting OXYCONTIN
Process for preparing oxycodone compositions
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Process for preparing oxycodone hydrochloride having less than 25 PPM 14-hydroxycodeinone
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MANAGEMENT OF PAIN SEVERE ENOUGH TO REQUIRE DAILY, AROUND-THE-CLOCK, LONG-TERM OPIOID TREATMENT AND FOR WHICH ALTERNATIVE TREATMENT OPTIONS ARE INADEQUATE
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MANAGEMENT OF PAIN SEVERE ENOUGH TO REQUIRE DAILY, AROUND-THE-CLOCK, LONG-TERM OPIOID TREATMENT AND FOR WHICH ALTERNATIVE TREATMENT OPTIONS ARE INADEQUATE
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MANAGEMENT OF PAIN SEVERE ENOUGH TO REQUIRE DAILY, AROUND-THE-CLOCK, LONG-TERM OPIOID TREATMENT AND FOR WHICH ALTERNATIVE TREATMENT OPTIONS ARE INADEQUATE
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Oxycodone hydrochloride having less than 25 PPM 14-hydroxycodeinone
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MANAGEMENT OF PAIN SEVERE ENOUGH TO REQUIRE DAILY, AROUND-THE-CLOCK, LONG-TERM OPIOID TREATMENT AND FOR WHICH ALTERNATIVE TREATMENT OPTIONS ARE INADEQUATE
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MANAGEMENT OF PAIN SEVERE ENOUGH TO REQUIRE DAILY, AROUND-THE-CLOCK, LONG-TERM OPIOID TREATMENT AND FOR WHICH ALTERNATIVE TREATMENT OPTIONS ARE INADEQUATE
Oxycodone compositions
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Tamper resistant dosage forms
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-004 | Apr 5, 2010 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-006 | Apr 5, 2010 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-003 | Apr 5, 2010 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-002 | Apr 5, 2010 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-005 | Apr 5, 2010 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OXYCONTIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-003 | Apr 5, 2010 | ⤷ Sign Up | ⤷ Sign Up |
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-005 | Apr 5, 2010 | ⤷ Sign Up | ⤷ Sign Up |
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-002 | Apr 5, 2010 | ⤷ Sign Up | ⤷ Sign Up |
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-003 | Apr 5, 2010 | ⤷ Sign Up | ⤷ Sign Up |
| Purdue Pharma Lp | OXYCONTIN | oxycodone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022272-002 | Apr 5, 2010 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for OXYCONTIN
See the table below for patents covering OXYCONTIN around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Germany | 602007010974 | ⤷ Sign Up | |
| Russian Federation | 2007132975 | ⤷ Sign Up | |
| Canada | 2551231 | ⤷ Sign Up | |
| Israel | 178787 | תהליך ליצור צורת מתן מוצקה חסינה בפני התמכרות המכילה אופיואיד וכורכן פוליאתילן אוקסיד בעל חוזק לשבירה של לפחות 500 ניוטון ושלב של חשיפה לאולטרסאונד ולכוח (Process for the production of an abuse proofed solid dosage form, comprising opioid and polyethylene oxide binder having a breaking srtength of at least 500n and a step of exposition to ultrasound and force) | ⤷ Sign Up |
| European Patent Office | 1258246 | Compositions d'oxycodone à libération controlée (Controlled release oxycodone compositions) | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OXYCONTIN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1685839 | 92292 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: COMBINAISON D OXYCODONE EN TANT QUE COMPOSANT A ET DE NALOXONE EN TANT QUE COMPOSANT B SOUS TOUTES LES FORMES PROTEGES PAR LE BREVET DE BASE |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


