TASIGNA Drug Patent Profile
✉ Email this page to a colleague
When do Tasigna patents expire, and what generic alternatives are available?
Tasigna is a drug marketed by Novartis and is included in one NDA. There are seven patents protecting this drug and two Paragraph IV challenges.
This drug has two hundred and ninety patent family members in fifty-two countries.
The generic ingredient in TASIGNA is nilotinib hydrochloride. There are eleven drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the nilotinib hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Tasigna
Tasigna was eligible for patent challenges on October 29, 2011.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (nilotinib hydrochloride), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for TASIGNA?
- What are the global sales for TASIGNA?
- What is Average Wholesale Price for TASIGNA?
Summary for TASIGNA
| International Patents: | 290 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 133 |
| Clinical Trials: | 67 |
| Patent Applications: | 4,662 |
| Drug Prices: | Drug price information for TASIGNA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TASIGNA |
| What excipients (inactive ingredients) are in TASIGNA? | TASIGNA excipients list |
| DailyMed Link: | TASIGNA at DailyMed |
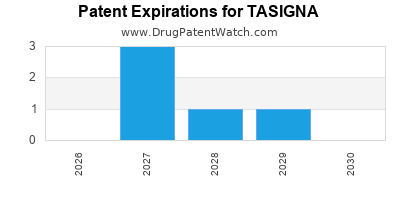

Paragraph IV (Patent) Challenges for TASIGNA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TASIGNA | Capsules | nilotinib hydrochloride | 50 mg | 022068 | 1 | 2019-10-17 |
| TASIGNA | Capsules | nilotinib hydrochloride | 150 mg and 200 mg | 022068 | 1 | 2013-11-08 |
US Patents and Regulatory Information for TASIGNA
TASIGNA is protected by seven US patents and five FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | RX | Yes | Yes | 8,293,756*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | RX | Yes | Yes | 8,163,904*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | RX | Yes | Yes | 9,061,029*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | RX | Yes | Yes | 8,389,537*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-003 | Mar 22, 2018 | RX | Yes | No | 8,163,904*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-002 | Jun 17, 2010 | RX | Yes | No | 8,501,760*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-002 | Jun 17, 2010 | RX | Yes | No | ⤷ Subscribe | ⤷ Subscribe | ⤷ Subscribe | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TASIGNA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-003 | Mar 22, 2018 | 7,169,791*PED | ⤷ Subscribe |
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-002 | Jun 17, 2010 | 7,169,791*PED | ⤷ Subscribe |
| Novartis | TASIGNA | nilotinib hydrochloride | CAPSULE;ORAL | 022068-001 | Oct 29, 2007 | 7,169,791*PED | ⤷ Subscribe |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TASIGNA
When does loss-of-exclusivity occur for TASIGNA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9029
Patent: DERIVADOS DE PIRIMIDIL-AMINO-BENZAMIDA PARA TRATAR TRASTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLOGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE BCR-ABL, C-KIT, DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 10322102
Patent: Method of treating proliferative disorders and other pathological conditions mediated by Bcr-Abl, c-Kit, DDR1, DDR2 or PDGF-R kinase activity
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 2012011693
Patent: método de tratamento de doenças proliferativas e de outras condições patológicas mediadas por atividade de bcr-abl, c-kit, ddr1, ddr2 ou pdgf-r quinase
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 79490
Patent: METHODE DE TRAITEMENT DE TROUBLES PROLIFERATIFS ET D'AUTRES AFFECTIONS PATHOLOGIQUES MEDIES PAR BCR-ABL, C-KIT, DDR1, DDR2 OU L'ACTIVITE KINASE DU PDGF-R (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Chile
Patent: 12001270
Patent: Compuesto 4-metil-n-[3-(4-metil-1h-imidazol-1-il)-5-(trifluorometil)fenil]-3-[(4-piridin-3-ilpirimidin-2-il)amino] benzamida (nilotinib) para el tratamiento de un trastorno proliferativo u otras condiciones mediadas por quinasa bcr-abl, c-kit, ddr1, ddr2 o pdgf-r, administrado oralmente dispersado en una compota de manzana.
Estimated Expiration: ⤷ Subscribe
China
Patent: 2612368
Patent: Method of treating proliferative disorders and other pathological conditions mediated by Bcr-Abl, c-Kit, DDR1, DDR2 or PDGF-R kinase activity
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 51690
Patent: MÉTODO PARA EL TRATAMIENTO DE TRANSTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLÓGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE BCR -ABL, C-KIT,DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
Croatia
Patent: 0160472
Estimated Expiration: ⤷ Subscribe
Cyprus
Patent: 17519
Estimated Expiration: ⤷ Subscribe
Denmark
Patent: 01384
Estimated Expiration: ⤷ Subscribe
Ecuador
Patent: 12011903
Patent: COMPOSICIONES ADECUADAS PARA EL TRATAMIENTO DE TRASTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLÓGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE BCR-ABL, C-KIT, DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 01384
Patent: MÉTHODE DE TRAITEMENT DE TROUBLES PROLIFÉRATIFS ET D'AUTRES AFFECTIONS PATHOLOGIQUES MÉDIÉS PAR BCR-ABL, C-KIT, DDR1, DDR2 OU L'ACTIVITÉ KINASE DU PDGF-R (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Finland
Patent: 01384
Estimated Expiration: ⤷ Subscribe
Guatemala
Patent: 1200150
Patent: MÉTODO PARA EL TRATAMIENTO DE TRASTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLÓGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE BRC-ABL,C-KIT, DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
Hong Kong
Patent: 69950
Patent: 治療增殖性障礙和其它由 激酶活性介導的病理學病症的方法 (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY BCR-ABLC-KITDDR1DDR2 PDGF-R)
Estimated Expiration: ⤷ Subscribe
Hungary
Patent: 27307
Estimated Expiration: ⤷ Subscribe
Israel
Patent: 9727
Patent: פירימידינילאמינובנזאמידים לטיפול בהפרעות התרבות מהירה ומצבים פתולוגיים אחרים המתווכים על ידי פעילות קינאז 2ddr, 1ddr, kit-c, abl-bcrאו r-pdgf (Pyrimidiylaminobenzamides for treating proliferative disorders and other pathological conditions mediated by bcr-abl, c-kit, ddr1, ddr2 or pdgf-r kinase activity)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 29615
Estimated Expiration: ⤷ Subscribe
Patent: 13511524
Estimated Expiration: ⤷ Subscribe
Patent: 15180636
Patent: 増殖性障害およびBCR−ABL、C−KIT、DDR1、DDR2またはPDGF−Rキナーゼ活性によりもたらされる他の病態の治療方法 (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Jordan
Patent: 34
Patent: طريقة لعلاج اضطرابات تكاثرية وحالات مرضية أخرى متوسطة بنشاط كيناز BCR-ABL، C-KIT، DDR1، DDR2، أو PDGF-R (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Malaysia
Patent: 9956
Patent: METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 12005694
Patent: METODO PARA EL TRATAMIENTO DE TRANSTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLOGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE CBR-ABL, C-KIT, DDR1, DDR2 O PDGF-R. (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY.)
Estimated Expiration: ⤷ Subscribe
Montenegro
Patent: 413
Patent: POSTUPAK LEČENJA PROLIFERATIVNIH OBOLJENJA I DRUGIH PATOLOŠKIH STANJA POSREDOVANIH AKTIVNOSCU BCR-ABL, C-KIT, DDR1, DDR2 ILI PDGF-R KINAZE (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Morocco
Patent: 738
Patent: عملية معالجة الاضطرابات التكاثري والحالات الطبية الأخرى ب par bcr ـ abl, cـ kit, ddr1, ddr2 أو نشاط الكيناز pdgf-r
Estimated Expiration: ⤷ Subscribe
New Zealand
Patent: 9968
Patent: Method of treating proliferative disorders and other pathological conditions mediated by bcr-abl, c-kit, ddr1, ddr2 or pdgf-r kinase activity
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 121476
Patent: COMPOSICION PARA EL TRATAMIENTO DE TRASTORNOS PROLIFERATIVOS Y OTRAS CONDICIONES PATOLOGICAS MEDIADAS POR LA ACTIVIDAD DE CINASA DE BCR-ABL, C-KIT, DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
Poland
Patent: 01384
Estimated Expiration: ⤷ Subscribe
Russian Federation
Patent: 25835
Patent: СПОСОБ ЛЕЧЕНИЯ ПРОЛИФЕРАТИВНЫХ НАРУШЕНИЙ И ДРУГИХ ПАТОЛОГИЧЕСКИХ СОСТОЯНИЙ, ОПОСРЕДОВАННЫХ АКТИВНОСТЬЮ КИНАЗ BCR-ABL, C-KIT, DDR1, DDR2 ИЛИ PDGF-R (METHOD FOR TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY ACTIVITY OF KINASE BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R)
Estimated Expiration: ⤷ Subscribe
Patent: 12124811
Patent: СПОСОБ ЛЕЧЕНИЯ ПРОЛИФЕРАТИВНЫХ НАРУШЕНИЙ И ДРУГИХ ПАТОЛОГИЧЕСКИХ СОСТОЯНИЙ, ОПОСРЕДОВАННЫХ АКТИВНОСТЬЮ КИНАЗ Bcr-Abl, c-Kit, DDR1, DDR2, ИЛИ PDGF-R
Estimated Expiration: ⤷ Subscribe
San Marino
Patent: 01600143
Patent: METODO DI TRATTAMENTO DI DISTURBI PROLIFERATIVI E ALTRE CONDIZIONI PATOLOGICHE MEDIATE DALLA ATTIVITÀ CHINASICA DI BCR-ABL, C-KIT, DDR1, DDR2 O PDGF-R
Estimated Expiration: ⤷ Subscribe
Serbia
Patent: 747
Patent: POSTUPAK LEČENJA PROLIFERATIVNIH OBOLJENJA I DRUGIH PATOLOŠKIH STANJA POSREDOVANIM KINAZNOM AKTIVNOŠĆU BCR-ABL, C-KIT, DDR1, DDR2 ILI PDGF-R (METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY)
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 201501169V
Patent: METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY
Estimated Expiration: ⤷ Subscribe
Slovenia
Patent: 01384
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 1203328
Patent: METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 1743315
Estimated Expiration: ⤷ Subscribe
Patent: 120102635
Patent: METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 72128
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 98116
Estimated Expiration: ⤷ Subscribe
Patent: 1141481
Patent: Method of treating proliferative disorders and other pathological conditions mediated by Bcr-Abl, c-Kit, DDR1, DDR2 or PDGF-R kinase activity
Estimated Expiration: ⤷ Subscribe
Tunisia
Patent: 12000206
Patent: METHOD OF TREATING PROLIFERATIVE DISORDERS AND OTHER PATHOLOGICAL CONDITIONS MEDIATED BY BCR-ABL, C-KIT, DDR1, DDR2 OR PDGF-R KINASE ACTIVITY
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TASIGNA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Finland | 2068839 | ⤷ Subscribe | |
| Japan | 2013511524 | ⤷ Subscribe | |
| European Patent Office | 1912973 | FORMES CRISTALLINES DE 4-METHYL-N-[3-(4-METHYL-IMIDAZOL-1-YL)-5-TRIFLUOROMETHYL-PHENYL]-3-(4-PYRIDIN-3-YL-PYRIMIDIN-2-YLAMINO)-BENZAMIDE (CRYSTALLINE FORMS OF 4-METHYL-N-[3-(4-METHYL-IMIDAZOL-1-YL)-5-TRIFLUOROMETHYL-PHENYL]3-(4-PYRIDIN-3-YL-PYRIMIDIN-2-YLAMINO)-BENZAMIDE) | ⤷ Subscribe |
| Japan | 4110140 | ⤷ Subscribe | |
| Norway | 20080897 | ⤷ Subscribe | |
| Mexico | PA05000328 | INHIBIDORES DE CINASAS DE TIROSINA. (INHIBITORS OF TYROSINE KINASES.) | ⤷ Subscribe |
| South Africa | 200901511 | PHARMACEUTICAL COMPOSITIONS COMPRISING NILOTINIB OR ITS SALT | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
TASIGNA Market Analysis and Financial Projection
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


