CORLANOR Drug Patent Profile
✉ Email this page to a colleague
When do Corlanor patents expire, and what generic alternatives are available?
Corlanor is a drug marketed by Amgen Inc and is included in two NDAs. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has ninety-seven patent family members in forty-two countries.
The generic ingredient in CORLANOR is ivabradine hydrochloride. There are nine drug master file entries for this compound. Six suppliers are listed for this compound. Additional details are available on the ivabradine hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Corlanor
A generic version of CORLANOR was approved as ivabradine hydrochloride by INGENUS PHARMS LLC on December 30th, 2021.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for CORLANOR?
- What are the global sales for CORLANOR?
- What is Average Wholesale Price for CORLANOR?
Summary for CORLANOR
| International Patents: | 97 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 105 |
| Clinical Trials: | 6 |
| Patent Applications: | 215 |
| Drug Prices: | Drug price information for CORLANOR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CORLANOR |
| What excipients (inactive ingredients) are in CORLANOR? | CORLANOR excipients list |
| DailyMed Link: | CORLANOR at DailyMed |
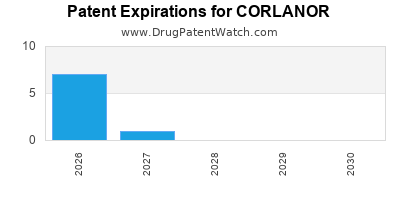
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for CORLANOR
Generic Entry Date for CORLANOR*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for CORLANOR
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Amgen | Phase 3 |
| Phillip Levy | Phase 4 |
| iRhythm Technologies, Inc. | Phase 4 |
Pharmacology for CORLANOR
Paragraph IV (Patent) Challenges for CORLANOR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CORLANOR | Tablets | ivabradine hydrochloride | 5 mg and 7.5 mg | 206143 | 6 | 2019-10-15 |
US Patents and Regulatory Information for CORLANOR
CORLANOR is protected by eight US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of CORLANOR is ⤷ Subscribe.
This potential generic entry date is based on patent 7,867,996.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amgen Inc | CORLANOR | ivabradine | SOLUTION;ORAL | 209964-001 | Apr 22, 2019 | RX | Yes | Yes | 7,867,996 | ⤷ Subscribe | Y | Y | ⤷ Subscribe | ||
| Amgen Inc | CORLANOR | ivabradine hydrochloride | TABLET;ORAL | 206143-001 | Apr 15, 2015 | AB | RX | Yes | No | 7,879,842*PED | ⤷ Subscribe | Y | ⤷ Subscribe | ||
| Amgen Inc | CORLANOR | ivabradine | SOLUTION;ORAL | 209964-001 | Apr 22, 2019 | RX | Yes | Yes | 7,879,842 | ⤷ Subscribe | Y | Y | ⤷ Subscribe | ||
| Amgen Inc | CORLANOR | ivabradine hydrochloride | TABLET;ORAL | 206143-002 | Apr 15, 2015 | AB | RX | Yes | Yes | 7,867,996*PED | ⤷ Subscribe | Y | ⤷ Subscribe | ||
| Amgen Inc | CORLANOR | ivabradine hydrochloride | TABLET;ORAL | 206143-001 | Apr 15, 2015 | AB | RX | Yes | No | 7,361,649*PED | ⤷ Subscribe | Y | ⤷ Subscribe | ||
| Amgen Inc | CORLANOR | ivabradine hydrochloride | TABLET;ORAL | 206143-002 | Apr 15, 2015 | AB | RX | Yes | Yes | 7,361,649*PED | ⤷ Subscribe | Y | ⤷ Subscribe | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for CORLANOR
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Accord Healthcare S.L.U. | Ivabradine Accord | ivabradine | EMEA/H/C/004241 Symptomatic treatment of chronic stable angina pectorisIvabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm.Ivabradine is indicated :- in adults unable to tolerate or with a contra-indication to the use of beta-blockers- or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.Treatment of chronic heart failureIvabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. (see section 5.1) |
Authorised | yes | no | no | 2017-05-22 | |
| Zentiva, k.s. | Ivabradine Zentiva | ivabradine | EMEA/H/C/004117 Symptomatic treatment of chronic stable angina pectoris Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm. Ivabradine is indicated:in adults unable to tolerate or with a contra-indication to the use of beta-blockersorin combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose. Treatment of chronic heart failure Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. |
Authorised | yes | no | no | 2016-11-11 | |
| Les Laboratoires Servier | Corlentor | ivabradine | EMEA/H/C/000598 Symptomatic treatment of chronic stable angina pectorisIvabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm. Ivabradine is indicated:in adults unable to tolerate or with a contraindication to the use of beta-blockersor in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.Treatment of chronic heart failureIvabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. |
Authorised | no | no | no | 2005-10-25 | |
| Les Laboratoires Servier | Procoralan | ivabradine | EMEA/H/C/000597 Symptomatic treatment of chronic stable angina pectoris Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm. Ivabradine is indicated :in adults unable to tolerate or with a contraindication to the use of beta-blockersor in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.Treatment of chronic heart failure Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. |
Authorised | no | no | no | 2005-10-25 | |
| "Anpharm" Przedsiębiorstwo Farmaceutyczne S.A. | Ivabradine Anpharm | ivabradine | EMEA/H/C/004187 Symptomatic treatment of chronic stable angina pectorisIvabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm. Ivabradine is indicated:in adults unable to tolerate or with a contra-indication to the use of beta-blockersor in combination with beta-blockers in patients inadequately controlled with an optimal betablocker dose.Treatment of chronic heart failureIvabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. |
Authorised | no | no | no | 2015-09-08 | |
| JensonR+ Limited | Ivabradine JensonR | ivabradine | EMEA/H/C/004217 Symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate ≥ 70 bpm. Ivabradine is indicated: - in adults unable to tolerate or with a contra-indication to the use of beta-blockers - or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose. Treatment of chronic heart failure Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is ≥ 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated., |
Withdrawn | yes | no | no | 2016-11-11 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for CORLANOR
When does loss-of-exclusivity occur for CORLANOR?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 07
Patent: Beta-crystalline form of ivabradine hydrochloride,a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Patent: 56
Patent: y-Crystalline form of Ivabradine Hydrochloride, a process for its preperation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Argentina
Patent: 2926
Patent: FORMA CRISTALINA GAMA DEL CLORHIDRATO DE IVABRADINA, UN PROCEDIMIENTO PARA SU PREPARACION Y COMPOSICIONES QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 3147
Patent: FORMA CRISTALINA DEL CLORHIDRATO DE IVABRADINA, UN PROCEDIMIENTO PARA SU PREPARACION Y COMPOSICIONES FARMACEUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 06200856
Patent: beta-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Patent: 06200857
Patent: y-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Austria
Patent: 96974
Estimated Expiration: ⤷ Subscribe
Patent: 07926
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 0600623
Patent: forma beta-cristalina de cloridrato de ivabradina, processo para preparação do mesmo e composições farmacêuticas contendo o mesmo
Estimated Expiration: ⤷ Subscribe
Patent: 0600796
Patent: forma gama-cristalina de cloridrato de ivabradina, processo para preparação do mesmo e composições farmacêuticas contendo o mesmo
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 37400
Patent: FORME CRISTALLINE Y DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT (Y CRYSTALLINE FORM OF IVABRADINE CHLORHYDRATE, PROCESS FOR THE PREPARATION THEREOF AND PHARMACEUTICAL COMPOUNDS CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Patent: 37414
Patent: FORME CRISTALLINE B DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT (B CRYSTALLINE FORM OF IVABRADINE CHLORHYDRATE, PROCESS FOR THE PREPARATION THEREOF AND PHARMACEUTICAL COMPOUNDS CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
China
Patent: 27600
Patent: Crystalline form beta of the chlorhydrate of ivabradine, process for its preparation and pharamcetuical composition containing it
Estimated Expiration: ⤷ Subscribe
Patent: 27602
Patent: Crystalline form gamma of the chlorhydrate of ivabradine, process for its preparation and phamaceutical composition containing it
Estimated Expiration: ⤷ Subscribe
Patent: 0402502
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 70096
Patent: FORMA CRISTALINA BETA DE CLORHITRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y LAS COMPOSICIONES FARMACEUTICAS QUE LAS CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 70097
Patent: FORMA CRISTALINA GAMMA DEL CLORHITRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y LAS COMPOSICIONES FARMACEUTICAS QUE LAS CONTIENEN
Estimated Expiration: ⤷ Subscribe
Costa Rica
Patent: 48
Patent: FORMA CRISTALINA BETA DEL CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y COMPOSICIONES FARMACEUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 49
Patent: FORMA CRISTALINA GAMMA DEL CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y COMPOSICIONES FARMACEUTICAS QUE LO CONTIENEN
Estimated Expiration: ⤷ Subscribe
Croatia
Patent: 0080406
Estimated Expiration: ⤷ Subscribe
Patent: 0080520
Estimated Expiration: ⤷ Subscribe
Cuba
Patent: 614
Patent: FORMA CRISTALINA BETA DEL CLORHIDRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y LAS COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 616
Patent: FORMA CRISTALINA GAMMA DEL CLORHIDRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y LAS COMPOSICIONES FARMACÉUTICAS QUE LAS CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 060037
Patent: FORMA CRISTALINA GAMMA DEL CLORHIDRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y LAS COMPOSICIONES FARMACÉUTICAS QUE LAS CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 060039
Patent: FORMA CRISTALINA BETA DEL CLORHIDRATO DE LA IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y LAS COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Cyprus
Patent: 09010
Estimated Expiration: ⤷ Subscribe
Patent: 09072
Estimated Expiration: ⤷ Subscribe
Denmark
Patent: 95965
Estimated Expiration: ⤷ Subscribe
Patent: 07562
Estimated Expiration: ⤷ Subscribe
Ecuador
Patent: 066373
Patent: FORMA CRISTALINA BETA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 066375
Patent: FORMA CRISTALINA GAMMA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Eurasian Patent Organization
Patent: 8464
Patent: β-КРИСТАЛЛИЧЕСКАЯ ФОРМА ГИДРОХЛОРИДА ИВАБРАДИНА, СПОСОБ ЕЁ ПОЛУЧЕНИЯ И ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, КОТОРЫЕ ЕЁ СОДЕРЖАТ (BETA-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Patent: 8465
Patent: γ-КРИСТАЛЛИЧЕСКАЯ ФОРМА ГИДРОХЛОРИДА ИВАБРАДИНА, СПОСОБ ЕЁ ПОЛУЧЕНИЯ И ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, КОТОРЫЕ ЕЁ СОДЕРЖАТ (γ-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITION CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Patent: 0600320
Patent: γ-КРИСТАЛЛИЧЕСКАЯ ФОРМА ГИДРОХЛОРИДА ИВАБРАДИНА, СПОСОБ ЕЁ ПОЛУЧЕНИЯ И ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, КОТОРЫЕ ЕЁ СОДЕРЖАТ
Estimated Expiration: ⤷ Subscribe
Patent: 0600322
Patent: β-КРИСТАЛЛИЧЕСКАЯ ФОРМА ГИДРОХЛОРИДА ИВАБРАДИНА, СПОСОБ ЕЁ ПОЛУЧЕНИЯ И ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, КОТОРЫЕ ЕЁ СОДЕРЖАТ
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 95965
Patent: Forme cristalline beta du chlorhydrate de l'ivabradine, son procédé de préparation, et les compositions pharmaceutiques qui la contiennent (Crystalline form beta of the chlorhydrate of ivabradine, process for its preparation and pharamcetuical composition containing it)
Estimated Expiration: ⤷ Subscribe
Patent: 07562
Patent: Forme cristalline gamma du chlorhydrate de l'ivabradine, son procede de preparation, et les compositions pharmaceutique qui la contiennent (Crystalline form gamma of the chlorhydrate of ivabradine, process for its preparation and pharamcetuical composition containing it)
Estimated Expiration: ⤷ Subscribe
France
Patent: 82553
Patent: FORME CRISTALLINE BETA DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT
Estimated Expiration: ⤷ Subscribe
Patent: 82555
Patent: FORME CRISTALLINE GAMMA DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT (New gamma crystalline form of ivabradine hydrochloride having specific X-ray diffraction profile)
Estimated Expiration: ⤷ Subscribe
Georgia, Republic of
Patent: 0084465
Patent: BETA-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Patent: 0084467
Patent: GAMMA CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Germany
Patent: 2006001312
Estimated Expiration: ⤷ Subscribe
Patent: 2006002624
Estimated Expiration: ⤷ Subscribe
Guatemala
Patent: 0600084
Patent: FORMA CRISTALINA BETA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 0600088
Patent: FORMA CRISTALINA GAMMA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y COMPOSICIONES FARMACEUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Hong Kong
Patent: 96387
Patent: ß-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Patent: 96659
Patent: Crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 25776
Estimated Expiration: ⤷ Subscribe
Patent: 28974
Estimated Expiration: ⤷ Subscribe
Patent: 06241154
Patent: beta-CRYSTAL FORM OF IVABRADINE HYDROCHLORIDE, METHOD FOR PRODUCING THE SAME AND MEDICINAL COMPOSITION CONTAINING THE SAME
Estimated Expiration: ⤷ Subscribe
Patent: 06241156
Patent: gamma-CRYSTAL FORM OF IVABRADINE HYDROCHLORIDE, METHOD FOR PRODUCING THE SAME AND MEDICINAL COMPOSITION CONTAINING THE SAME
Estimated Expiration: ⤷ Subscribe
Jordan
Patent: 15
Patent: الشكل البلوري جاما لهيدروكلوريد الايفابراداين وعملية تحضيره والمكونات الصيدلانية المحتوية عليه (Gamma-Crystalline form of ivabradine hydrochloride,a process for its preparation and pharmaceutical composition containing it)
Estimated Expiration: ⤷ Subscribe
Patent: 48
Patent: الشكل البلوري بيتا لهيدروكلوريد الايفابراداين وعملية تحضيره والمكونات الصيدلانية المحتوية علية (Beta Crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it)
Estimated Expiration: ⤷ Subscribe
Malaysia
Patent: 8128
Patent: B-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Patent: 8129
Patent: Y-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Montenegro
Patent: 408
Patent: BETA KRISTALNI OBLIK IVABRADIN HIDROHLORIDA, POSTUPAK NJEGOVE IZRADE I FARMACEUTSKE SMEŠE KOJE GA SADRŽE (CRYSTALLINE FORM BETA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARAMCETUICAL COMPOSITION CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Patent: 747
Patent: KRISTALNI OBLIK GAMA IVABRADIN HLORHIDRATA, POSTUPAK NJEGOVE IZRADE I FARMACEUTSKE SMEŠE KOJE GA SADRŽE (CRYSTALLINE FORM GAMMA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARAMCETUICAL COMPOSITION CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Morocco
Patent: 131
Patent: FORME CRISTALLINE ß DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT.
Estimated Expiration: ⤷ Subscribe
Patent: 134
Patent: FORME CRISTALLINE GAMMA DU CHLORHYDRATE DE L'IVABRADINE, SON PROCEDE DE PREPARATION, ET LES COMPOSITIONS PHARMACEUTIQUES QUI LA CONTIENNENT.
Estimated Expiration: ⤷ Subscribe
New Zealand
Patent: 5576
Patent: Beta-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Patent: 5578
Patent: Gamma-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Norway
Patent: 8370
Estimated Expiration: ⤷ Subscribe
Patent: 8481
Estimated Expiration: ⤷ Subscribe
Patent: 060946
Estimated Expiration: ⤷ Subscribe
Patent: 060948
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 061009
Patent: FORMA CRISTALINA BETA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACION, Y COMPOSICIONES FARMACEUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Patent: 061142
Patent: FORMA CRISTALINA GAMMA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACION Y COMPOSICIONES FARMACEUTICAS QUE LA CONTIENEN
Estimated Expiration: ⤷ Subscribe
Poland
Patent: 95965
Estimated Expiration: ⤷ Subscribe
Patent: 07562
Estimated Expiration: ⤷ Subscribe
Portugal
Patent: 95965
Estimated Expiration: ⤷ Subscribe
Patent: 07562
Estimated Expiration: ⤷ Subscribe
Saudi Arabia
Patent: 270038
Patent: الصورة المتبلرة- β لهيدروكلوريد إيفابرادين ، وعملية لتحضيرها، (�-Crystalline Form of Ivabradine Hydrochloride, a Process for its Preparation and Pharmaceutical Compositions Containing It)
Estimated Expiration: ⤷ Subscribe
Patent: 270040
Patent: الصورة المتبلرة - γ لهيدروكلوريد إيڤابرادين، وعملية لتحضيرها، وتركيبات صيدلانية تحتوي عليها، (γ -Crystalline Form of Ivabradine Hydrochloride, a Process for its Preparation and Pharmaceutical Compositions Containing It)
Estimated Expiration: ⤷ Subscribe
Serbia
Patent: 598
Patent: KRISTALNI OBLIK GAMA IVABRADIN HLORHIDRATA, POSTUPAK NJEGOVE IZRADE I FARMACEUTSKE SMEŠE KOJE GA SADRŽE (CRYSTALLINE FORM GAMMA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPRATION AND PHARMACEUTICAL COMPOSITION CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Patent: 661
Patent: BETA KRISTALNI OBLIK IVABRADIN HIDROHLORIDA, POSTUPAK NJEGOVE IZRADE I FARMACEUTSKE SMEŠE KOJE GA SADRŽE (CRYSTALLINE FORM BETA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITION CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 5228
Patent: Beta-crystalline form of ivabradine hydrochloride,a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Patent: 5229
Patent: Gamma-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceuticalcompositions containing it
Estimated Expiration: ⤷ Subscribe
Slovenia
Patent: 95965
Estimated Expiration: ⤷ Subscribe
Patent: 07562
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 0601762
Patent: ß-crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
Patent: 0601764
Patent: ?-Crystalline form of ivabradine hydrochloride, a process for its preparation and pharmaceutical compositions containing it
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 0827502
Estimated Expiration: ⤷ Subscribe
Patent: 0835447
Estimated Expiration: ⤷ Subscribe
Patent: 060095499
Patent: gamma;-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Patent: 060095501
Patent: beta;-CRYSTALLINE FORM OF IVABRADINE HYDROCHLORIDE, A PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 08689
Estimated Expiration: ⤷ Subscribe
Patent: 13581
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 14143
Estimated Expiration: ⤷ Subscribe
Patent: 14144
Estimated Expiration: ⤷ Subscribe
Patent: 0640872
Estimated Expiration: ⤷ Subscribe
Patent: 0640873
Estimated Expiration: ⤷ Subscribe
Ukraine
Patent: 904
Patent: CRYSTALLINE FORM BETA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARAMCETUICAL COMPOSITION CONTAINING IT
Estimated Expiration: ⤷ Subscribe
Patent: 595
Patent: ГАММА-КРИСТАЛЛИЧЕСКАЯ ФОРМА ГИДРОХЛОРИДА ИВАБРАДИНА, СПОСОБ ЕЕ ПОЛУЧЕНИЯ И ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, КОТОРАЯ ЕЕ СОДЕРЖИТ;ГАММА-КРИСТАЛІЧНА ФОРМА ГІДРОХЛОРИДУ ІВАБРАДИНУ, СПОСІБ ЇЇ ОДЕРЖАННЯ І ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ, ЯКА ЇЇ МІСТИТЬ (GAMMA-CRYSTALLINE FORM OF THE CHLOROHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING IT)
Estimated Expiration: ⤷ Subscribe
Uruguay
Patent: 405
Patent: FORMA CRISTALINA BETA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y COMPOSICIONES FARMACÉUTICAS QUE LA CONTINEN.
Estimated Expiration: ⤷ Subscribe
Patent: 406
Patent: FORMA CRISTALINA GAMMA DE CLORHIDRATO DE IVABRADINA, SU PROCEDIMIENTO DE PREPARACIÓN Y COMPOSICIONES FARMACÉUTICAS QUE LA CONTIENEN.
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering CORLANOR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Germany | 602006002624 | ⤷ Subscribe | |
| Slovenia | 1707562 | ⤷ Subscribe | |
| Montenegro | 02747 | KRISTALNI OBLIK GAMA IVABRADIN HLORHIDRATA, POSTUPAK NJEGOVE IZRADE I FARMACEUTSKE SMEŠE KOJE GA SADRŽE (CRYSTALLINE FORM GAMMA OF THE CHLORHYDRATE OF IVABRADINE, PROCESS FOR ITS PREPARATION AND PHARAMCETUICAL COMPOSITION CONTAINING IT) | ⤷ Subscribe |
| Germany | 602006001312 | ⤷ Subscribe | |
| China | 1827602 | Crystalline form gamma of the chlorhydrate of ivabradine, process for its preparation and phamaceutical composition containing it | ⤷ Subscribe |
| Saudi Arabia | 06270040 | الصورة المتبلرة - γ لهيدروكلوريد إيڤابرادين، وعملية لتحضيرها، وتركيبات صيدلانية تحتوي عليها، (γ -Crystalline Form of Ivabradine Hydrochloride, a Process for its Preparation and Pharmaceutical Compositions Containing It) | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
CORLANOR Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


