FARXIGA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Farxiga, and when can generic versions of Farxiga launch?
Farxiga is a drug marketed by Astrazeneca Ab and is included in one NDA. There are eighteen patents protecting this drug and one Paragraph IV challenge.
This drug has four hundred and forty patent family members in fifty-one countries.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this compound. Five suppliers are listed for this compound. Additional details are available on the dapagliflozin profile page.
DrugPatentWatch® Generic Entry Outlook for Farxiga
Farxiga was eligible for patent challenges on January 8, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 16, 2030. This may change due to patent challenges or generic licensing.
Annual sales in 2021 were $2.7bn, indicating a strong incentive for generic entry.
There have been eleven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are twelve tentative approvals for the generic drug (dapagliflozin), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for FARXIGA?
- What are the global sales for FARXIGA?
- What is Average Wholesale Price for FARXIGA?
Summary for FARXIGA
| International Patents: | 440 |
| US Patents: | 18 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 5 |
| Raw Ingredient (Bulk) Api Vendors: | 41 |
| Clinical Trials: | 49 |
| Patent Applications: | 148 |
| Drug Prices: | Drug price information for FARXIGA |
| Drug Sales Revenues: | Drug sales revenues for FARXIGA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for FARXIGA |
| What excipients (inactive ingredients) are in FARXIGA? | FARXIGA excipients list |
| DailyMed Link: | FARXIGA at DailyMed |
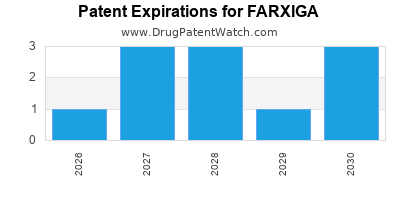
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for FARXIGA
Generic Entry Date for FARXIGA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for FARXIGA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| The University of Hong Kong | Phase 2 |
| University Health Network, Toronto | Phase 2 |
| ZonMw: The Netherlands Organisation for Health Research and Development | Phase 4 |
Pharmacology for FARXIGA
| Drug Class | Sodium-Glucose Cotransporter 2 Inhibitor |
| Mechanism of Action | Sodium-Glucose Transporter 2 Inhibitors |
Paragraph IV (Patent) Challenges for FARXIGA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| FARXIGA | Tablets | dapagliflozin | 5 mg and 10 mg | 202293 | 20 | 2018-01-08 |
US Patents and Regulatory Information for FARXIGA
FARXIGA is protected by eighteen US patents and five FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of FARXIGA is ⤷ Subscribe.
This potential generic entry date is based on patent 7,919,598.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | RX | Yes | Yes | ⤷ Subscribe | ⤷ Subscribe | ⤷ Subscribe | ||||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | RX | Yes | Yes | 8,431,685*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | RX | Yes | Yes | 8,716,251*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for FARXIGA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | 7,563,871 | ⤷ Subscribe |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | 6,936,590 | ⤷ Subscribe |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 6,936,590 | ⤷ Subscribe |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for FARXIGA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Forxiga | dapagliflozin | EMEA/H/C/002322 Type 2 diabetes mellitusForxiga is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureForxiga is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseForxiga is indicated in adults for the treatment of chronic kidney disease. |
Authorised | no | no | no | 2012-11-11 | |
| AstraZeneca AB | Edistride | dapagliflozin | EMEA/H/C/004161 Type 2 diabetes mellitusEdistride is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureEdistride is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseEdistride is indicated in adults for the treatment of chronic kidney disease. |
Authorised | no | no | no | 2015-11-09 | |
| Viatris Limited | Dapagliflozin Viatris | dapagliflozin | EMEA/H/C/006006 Type 2 diabetes mellitusDapagliflozin Viatris is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise- as monotherapy when metformin is considered inappropriate due to intolerance.- in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureDapagliflozin Viatris is indicated in adults for the treatment of symptomatic chronic heart failure with reduced ejection fraction.Chronic kidney diseaseDapagliflozin Viatris is indicated in adults for the treatment of chronic kidney disease. |
Authorised | yes | no | no | 2023-03-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for FARXIGA
When does loss-of-exclusivity occur for FARXIGA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1730
Patent: ESTRUCTURAS DE CRISTAL DE INHIBIDORES SGLT2 Y PROCESOS PARA PREPARAR LOS MISMOS
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 07265246
Patent: Crystalline solvates and complexes of (IS) -1, 5-anhydro-L-C- (3- ( (phenyl) methyl) phenyl) -D-glucitol derivatives with amino acids as SGLT2 inhibitors for the treatment of diabetes
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 0713544
Patent: solvatos cristalinos complexos de derivados de (1s)-1,5-anidro-1-c-(3-((fenil) metil)fenil)-d-glucitol com aminoÁcidos como inibidores de sglt2 para o tratamento de diabetes
Estimated Expiration: ⤷ Subscribe
Patent: 2017015106
Patent: estrutura de cristal de formula 1b e composição farmacêutica
Estimated Expiration: ⤷ Subscribe
Patent: 2017021516
Patent: processo de preparação de uma estrutura de cristal de solvato de (s)-propileno glicol de fórmula ia
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 53344
Patent: SOLVATES CRISTALLINS ET COMPLEXES DE DERIVES DE (IS)-1,5-ANHYDRO-L-C-{3-[(PHENYL)METHYL]PHENYL}-D-GLUCITOL AVEC DES ACIDES AMINES EN TANT QU'INHIBITEURS DE SGLT2 POUR LE TRAITEMENT DU DIABETE (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-1-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS ASSGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 24318
Patent: SOLVATES CRISTALLINS ET COMPLEXES DE DERIVES DE (IS)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL-D-GLUCITOL AVEC DES ACIDES AMINES EN TANT QU'INH IBITEURS DE SGLT2 POUR LE TRAITEMENT DU DIABETE (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 85797
Patent: SOLVATES CRISTALLINS ET COMPLEXES DE DERIVES DE (IS)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL-D-GLUCITOL AVEC DES ACIDES AMINES EN TANT QU'INH IBITEURS DE SGLT2 POUR LE TRAITEMENT DU DIABETE (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Chile
Patent: 07001915
Patent: Compuesto cristalino de 6-[4-cloro-3-(4-etoxibencil)fenil]tetrahidropiran-2,3,4,5,-tetraol; procedimiento de preparacion; composicion farmaceutica que comprende a dicho compuesto; y su uso para preparar un medicamento para tratar diabetes, dislipidemia, obesidad, entre otras.
Estimated Expiration: ⤷ Subscribe
China
Patent: 1479287
Patent: Crystalline solvates and complexes of (is) -1, 5-anhydro-l-c- (3- ( (phenyl) methyl) phenyl) -d-glucitol derivatives with amino acids as sglt2 inhibitors for the treatment of diabetes
Estimated Expiration: ⤷ Subscribe
Patent: 3145773
Patent: Crystalline solvates and complexes of (IS) -1, 5-anhydro-L-C- (3- ( (phenyl) methyl) phenyl) -D-glucitol derivatives with amino acids as SGLT2 inhibitors for the treatment of diabetes
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 60299
Patent: SOLVATOS CRISTALINOS Y COMPLEJOS DE DERIVADOS DE (1S)-1,5-ANHIDRO-1-C-(3-((FENIL)METIL)FENIL)-D-GLUCITOL CON AMINOÁCIDOS COMO INHIBIDORES SGLT2 PARA EL TRATAMIENTO DE DIABETES.
Estimated Expiration: ⤷ Subscribe
Croatia
Patent: 0141007
Estimated Expiration: ⤷ Subscribe
Cyprus
Patent: 15738
Estimated Expiration: ⤷ Subscribe
Denmark
Patent: 69374
Estimated Expiration: ⤷ Subscribe
Eurasian Patent Organization
Patent: 8229
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 0428
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-С-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА (CRYSTALLINE SOLVATES OF (IS)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 8259
Patent: КРИСТАЛЛИЧЕСКИЙ СОЛЬВАТ ПРОИЗВОДНОГО (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТОЙ И СПОСОБ ЕГО ПОЛУЧЕНИЯ (CRYSTALLINE SOLVATE OF (1S)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVE WITH AMINO ACID AND PROCESS FOR PREPARING THE SAME)
Estimated Expiration: ⤷ Subscribe
Patent: 5999
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 0900066
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ) -D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА
Estimated Expiration: ⤷ Subscribe
Patent: 1171333
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-С-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА
Estimated Expiration: ⤷ Subscribe
Patent: 1490902
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА
Estimated Expiration: ⤷ Subscribe
Patent: 1791254
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА
Estimated Expiration: ⤷ Subscribe
Patent: 2091391
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (1S)-1,5-АНГИДРО-1-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ В КАЧЕСТВЕ ИНГИБИТОРОВ SGLT2 ДЛЯ ЛЕЧЕНИЯ ДИАБЕТА
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 69374
Patent: SOLVATES CRISTALLINS DE DÉRIVÉS DE (1S)-1,5-ANHYDRO-1-C-(3-((PHÉNYL)MÉTHYL)PHÉNYL)-D-GLUCITOL AVEC DES ALCOHOLS EN TANT QU'INHIBITEURS DE SGLT2 POUR LE TRAITEMENT DU DIABÈTE (CRYSTALLINE SOLVATES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH ALCOHOLS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 57918
Patent: Solvates cristallins et complexes de dérivés de (1S)-1,5-anhydro-1-C-((phényl)méthyl) phényl)-d-glucitol avec acides aminés en tant qu'inhibiteurs SGLT2 pour le traitement des diabètes (Crystalline solvates and complexes of (1s)-1,5-anhydro-1-c-(3-((phenyl) methyl) phenyl)-d-glucitol derivatives with amino acids as SGLT2 inhibitors for the treatment of diabetes)
Estimated Expiration: ⤷ Subscribe
Patent: 45466
Patent: SOLVATE DE PROPYLENE GLYCOLE DE (2S,3R,4S,5S,6R)-2-[4-CHLORO-3-(4-ÉTHOXY-BENZYL)-PHÉNYL]-6-HYDROXYMÉTHYL-2-MÉTHOXY-TETRAHYDRO-PYRAN-3,4,5-TRIOL EN TANT QU'INHIBITEURS DU SGLT2 POUR LE TRAITEMENT DU DIABÈTE ((2S,3R,4S,5S,6R)-2-[4-CHLORO-3-(4-ETHOXY-BENZYL)-PHENYL]-6-HYDROXYMETHYL-2-METHOXY-TETRAHYDRO-PYRAN-3,4,5-TRIOL PROPYLENE GLYCOL SOLVATE AS SGT2 INHIBITOR FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 63807
Patent: COMPOSITION PHARMACEUTIQUE COMPRENANT SOLVATE DE (S)-PROPYLENE GLYCOL DE (2S,3R,4S,5S,6R)-2-[4-CHLORO-3-(4-ÉTHOXY-BENZYL)-PHÉNYL]-6-HYDROXYMÉTHYL-2-MÉTHOXY-TETRAHYDRO-PYRAN-3,4,5-TRIOL CRISTALLINE (PHARMACEUTICAL COMPOSITION COMPRISING CRYSTALLINE (2S,3R,4S,5S,6R)-2-[4-CHLORO-3-(4-ETHOXY-BENZYL)-PHENYL]-6-HYDROXYMETHYL-2-METHOXY-TETRAHYDRO-PYRAN-3,4,5-TRIOL (S)-PROPYLENE GLYCOL SOLVATE)
Estimated Expiration: ⤷ Subscribe
Hong Kong
Patent: 27359
Patent: 用於治療糖尿病的 抑製劑的 -脫水- 苯基 甲基 苯基 -葡萄糖醇衍生物與醇的結晶溶劑合物 (CRYSTALLINE SOLVATES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)- D-GLUCITOL DERIVATIVES WITH ALCOHOLS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES SGLT2 (1S)-15--1-C-(3 -(()))-D-)
Estimated Expiration: ⤷ Subscribe
Israel
Patent: 5882
Patent: צורות גבישיות של נגזרות של (1 s)-1, 5-אנהידרו-1- c -(3-((פניל)מתיל)פניל)-d-גלוציטול עם חומצות אמיניות ושימוש בהן לטיפול בסכרת (Crystalline forms of (1s)-1,5-anhydro-1-c-(3-(phenyl)methyl)phenyl)-d-glucitol derivatives with amino acids and use thereof for the treatment of diabetes)
Estimated Expiration: ⤷ Subscribe
Patent: 4180
Patent: סולבטים גבישיים וקומפלקסים של נגזרות של (s1) -1, 5 אנהידרו- 1 - c - ( 3- ((פניל(מתיל(פניל)-d- גלוציטול עם חומצות אמינו המשמשים כמעכבי sglt2 עבור הטיפול בסוכרת (Crystalline solvates and complexes of (1s)-1,5-anhydro-1-c-(3-((phenyl)methyl)phenyl)-d-glucitol derivatives with amino acids as sglt2 inhibitors for the treatment of diabetes)
Estimated Expiration: ⤷ Subscribe
Patent: 4181
Patent: סולבטים גבישיים וקומפלקסים של נגזרות -1,51)(s-אנהידרו(3--1-c-((פניל)מתיל)פניל)-d-גלוציטול עם חומצות אמינו המשמשים כמעכבי 2sglt עבור הטיפול בסוכרת (Crystalline solvates and complexes of (1s)-1,5-anhydro-1-c-(3-((phenyl)methyl)phenyl)-d-glucitol derivatives with amino acids as sglt2 inhibitors for the treatment of diabetes)
Estimated Expiration: ⤷ Subscribe
Patent: 4182
Patent: סולבטים גבישיים וקומפלקסים של נגזרות 1s))-5,1-אנהידרו-1-c-(3-((פניל)מתיל)פניל)-d-גלוציטול עם חומצות אמינו המשמשים כמעכבי 2sglt עבור הטיפול בסוכרת (Crystalline solvates and complexes of (1s)-1,5-anhydro-1-c-(3-((phenyl)methyl)phenyl)-d-glucitol derivatives with amino acids as sglt2 inhibitors for the treatment of diabetes)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 13889
Estimated Expiration: ⤷ Subscribe
Patent: 66651
Estimated Expiration: ⤷ Subscribe
Patent: 37187
Estimated Expiration: ⤷ Subscribe
Patent: 09545525
Estimated Expiration: ⤷ Subscribe
Patent: 13209394
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 15071636
Patent: 糖尿病の治療用SGLT2阻害剤としてのアミノ酸を有する(1S)−1,5−アンヒドロ−1−C−(3−((フェニル)メチル)フェニル)−D−グルシトール誘導体の結晶性溶媒和物および複合体 (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 16172758
Patent: 糖尿病の治療用SGLT2阻害剤としてのアミノ酸を有する(1S)−1,5−アンヒドロ−1−C−(3−((フェニル)メチル)フェニル)−D−グルシトール誘導体の結晶性溶媒和物および複合体 (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 17222681
Patent: 糖尿病の治療用SGLT2阻害剤としてのアミノ酸を有する(1S)−1,5−アンヒドロ−1−C−(3−((フェニル)メチル)フェニル)−D−グルシトール誘導体の結晶性溶媒和物および複合体 (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Patent: 19059779
Patent: 糖尿病の治療用SGLT2阻害剤としてのアミノ酸を有する(1S)−1,5−アンヒドロ−1−C−(3−((フェニル)メチル)フェニル)−D−グルシトール誘導体の結晶性溶媒和物および複合体 (CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES HAVING AMINO ACIDS AS SGLT2 INHIBITORS FOR TREATING DIABETES)
Estimated Expiration: ⤷ Subscribe
Malaysia
Patent: 8566
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5- ANHYDRO-1 -C-(3-( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 3930
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 9143
Patent: SOLVATOS Y COMPLEJOS CRISTALINOS DE LOS DERIVADOS DE (1S)-1,5-ANHIDRO-1-C-(3-((FENIL)METIL)FENIL)-D-GLUCITOL CON AMINOACIDOS COMO INHIBIDORES DE SGLT2 PARA EL TRATAMIENTO DE LA DIABETES. (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES.)
Estimated Expiration: ⤷ Subscribe
Patent: 7155
Patent: SOLVATOS Y COMPLEJOS CRISTALINOS DE LOS DERIVADOS DE (1S)-1, 5-ANHIDRO-1-C- (3-((FENIL) METIL) FENIL) -D-GLUCITOL CON AMINOÁCIDOS COMO INHIBIDORES DE SGLT2 PARA EL TRATAMIENTO DE LA DIABETES. (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES.)
Estimated Expiration: ⤷ Subscribe
Patent: 08015377
Patent: SOLVATOS Y COMPLEJOS CRISTALINOS DE LOS DERIVADOS DE (1S) - 1,5-ANHIDRO-1-C-(3-((FENIL)METIL)FENIL)-D-GLUCITOL CON AMINOACIDOS COMO INHIBIDORES DE SGLT2 PARA EL TRATAMIENTO DE LA DIABETES. (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES.)
Estimated Expiration: ⤷ Subscribe
New Zealand
Patent: 4346
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 9190
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 9195
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 9202
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Norway
Patent: 6828
Estimated Expiration: ⤷ Subscribe
Patent: 085169
Estimated Expiration: ⤷ Subscribe
Patent: 221233
Patent: Krystallinske solvater og komplekser av (1S)-1,5-anhydro-1-C-(3-((fenyl)metyl)fenyl)-D-glucitol-derivater med aminosyrer som SGLT2-inhibitorer for behandling av diabetes
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 080349
Patent: ESTRUCTURAS DE CRISTAL DE INHIBIDORES SGLT2 Y PROCESOS PARA PREPARAR LOS MISMOS
Estimated Expiration: ⤷ Subscribe
Patent: 120776
Patent: ESTRUCTURAS CRISTALINAS DE DAPAGLIFLOZIN Y SUS PROCESOS DE PREPARACION
Estimated Expiration: ⤷ Subscribe
Philippines
Patent: 012500168
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Poland
Patent: 69374
Estimated Expiration: ⤷ Subscribe
Portugal
Patent: 69374
Estimated Expiration: ⤷ Subscribe
Serbia
Patent: 638
Patent: KRISTALNI SOLVATI DERIVATA (1S)-1,5-ANHIDRO-1-C-(3-((FENIL) METIL) FENIL)-D-GLUCITOLA SA ALKOHOLIMA KAO INHIBITORI SGLT2 ZA TRETMAN DIJABETESA (CRYSTALLINE SOLVATES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH ALCOHOLS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 2741
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S) -1, 5-ANHYDRO-1-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Patent: 201402181S
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S) -1, 5-ANHYDRO-1-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Slovenia
Patent: 69374
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 0810475
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 1493102
Estimated Expiration: ⤷ Subscribe
Patent: 090023643
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 21665
Estimated Expiration: ⤷ Subscribe
Patent: 59862
Estimated Expiration: ⤷ Subscribe
Patent: 69130
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 21245
Estimated Expiration: ⤷ Subscribe
Patent: 66876
Estimated Expiration: ⤷ Subscribe
Patent: 19528
Estimated Expiration: ⤷ Subscribe
Patent: 0811127
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Subscribe
Patent: 1406743
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Subscribe
Patent: 1509927
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Subscribe
Patent: 1546054
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Subscribe
Ukraine
Patent: 765
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (IS)-1,5-АНГИДРО-L-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ КАК ИНГИБИТОРЫ БЕЛКА SGLT2, ПРИГОДНЫЕ В ЛЕЧЕНИИ ДИАБЕТА;КРИСТАЛІЧНІ СОЛЬВАТИ І КОМПЛЕКСИ ПОХІДНИХ (IS)-1,5-АНГІДРО-L-C-(3-((ФЕНІЛ)МЕТИЛ)ФЕНІЛ)-D-ГЛЮЦИТОЛУ З АМІНОКИСЛОТАМИ ЯК ІНГІБІТОРИ БІЛКА SGLT2, ПРИДАТНІ У ЛІКУВАННІ ДІАБЕТУ (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ((PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering FARXIGA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2356331 | METHODES DE SUPPRESSION DU GLUCAGON (METHODS FOR GLUCAGON SUPPRESSION) | ⤷ Subscribe |
| Serbia | 53638 | KRISTALNI SOLVATI DERIVATA (1S)-1,5-ANHIDRO-1-C-(3-((FENIL) METIL) FENIL)-D-GLUCITOLA SA ALKOHOLIMA KAO INHIBITORI SGLT2 ZA TRETMAN DIJABETESA (CRYSTALLINE SOLVATES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH ALCOHOLS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES) | ⤷ Subscribe |
| World Intellectual Property Organization (WIPO) | 2021037400 | ⤷ Subscribe | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for FARXIGA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1506211 | 2013/013 | Ireland | ⤷ Subscribe | PRODUCT NAME: DAPAGLIFLOZIN AND PHARACEUTICALLY ACCEPTABLE SALTS THREOF; REGISTRATION NO/DATE: EU/1/12/795/001-010 20121112 |
| 1734971 | 91989 | Luxembourg | ⤷ Subscribe | 91989, EXPIRES: 20260617 |
| 1506211 | 132014902277722 | Italy | ⤷ Subscribe | PRODUCT NAME: UNA COMBINAZIONE DI DAPAGLIFLOZIN O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE E METFORMINA O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE COME PROTETTI DAL BREVETTO DI BASE EP1506211(XIGDUO); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/13/900, 20140116 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
FARXIGA Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


