FOLIC Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Folic, and what generic alternatives are available?
Folic is a drug marketed by Ben Venue, Fresenius Kabi Usa, Xgen Pharms, Amneal Pharm, Athem, Barr, Cadila Pharms Ltd, Chartwell Molecular, Contract Pharmacal, Endo Operations, Everylife, Halsey, Hikma Pharms, Impax Labs, Ivax Sub Teva Pharms, Jubilant Cadista, Lannett, Leading, Lilly, Mk Labs, Nexgen Pharma Inc, Nuvo Pharms Inc, Pharmeral, Pioneer Pharms, Purepac Pharm, Qingdao Baheal Pharm, Sandoz, Sun Pharm Industries, Tablicaps, UDL, Usl Pharma, Valeant Pharm Intl, Vangard, Vintage Pharms, Watson Labs, Whiteworth Town Plsn, and Mission Pharma. and is included in thirty-nine NDAs.
The generic ingredient in FOLIC is folic acid. There are ten drug master file entries for this compound. Twenty-eight suppliers are listed for this compound. Additional details are available on the folic acid profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Folic
A generic version of FOLIC was approved as folic acid by WATSON LABS on December 31st, 1969.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for FOLIC?
- What are the global sales for FOLIC?
- What is Average Wholesale Price for FOLIC?
Summary for FOLIC
| US Patents: | 0 |
| Applicants: | 37 |
| NDAs: | 39 |
| Drug Prices: | Drug price information for FOLIC |
| Drug Sales Revenues: | Drug sales revenues for FOLIC |
| DailyMed Link: | FOLIC at DailyMed |
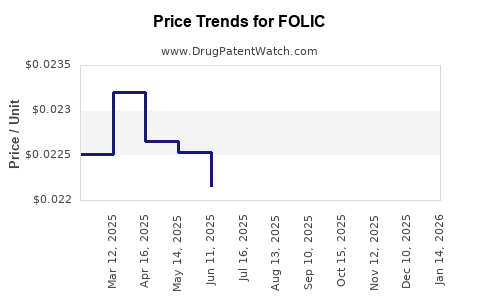
US Patents and Regulatory Information for FOLIC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Valeant Pharm Intl | FOLIC ACID | folic acid | TABLET;ORAL | 080903-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Sun Pharm Industries | FOLIC ACID | folic acid | TABLET;ORAL | 040582-001 | Jul 18, 2005 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Vangard | FOLIC ACID | folic acid | TABLET;ORAL | 088730-001 | Mar 23, 1984 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Tablicaps | FOLIC ACID | folic acid | TABLET;ORAL | 083133-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Ben Venue | FOLIC ACID | folic acid | INJECTABLE;INJECTION | 081066-001 | Dec 29, 1993 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


