Market Dynamics and Financial Trajectory for Metrogel (Metronidazole)
Introduction to Metrogel
Metrogel, a formulation of metronidazole, is a widely used antibiotic and antiprotozoal medication. It is available in various forms, including vaginal gel, tablets, capsules, and injections. Here, we will delve into the market dynamics and financial trajectory of Metrogel, focusing on its global market size, growth trends, key players, and financial highlights.
Global Market Size and Growth Trends
The global metronidazole market, which includes Metrogel, has seen mixed trends in recent years. As of 2021, the global metronidazole market size was estimated to be worth approximately $113 million. However, it is forecast to decline to $109.1 million by 2028, with a Compound Annual Growth Rate (CAGR) of -0.5% during the forecast period of 2022-2030[1].
In contrast, a more expansive view of the metronidazole market suggests a different trajectory. By 2032, the global metronidazole market is projected to reach $168.41 billion, exhibiting a CAGR of 5.96%[4]. This discrepancy highlights the complexity and varying forecasts within the market.
Market Segments and Applications
The metronidazole market is segmented based on type and application. Key segments include:
- Type: Glyoxal Process, EthideneDiamine Process, and Others[1].
- Application: Metronidazole Tablets, Metronidazole Capsule, Metronidazole Injection, and Others[1].
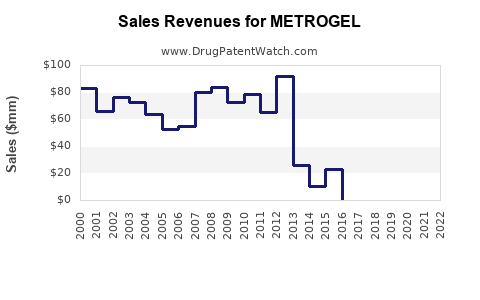
Metrogel, specifically the vaginal gel formulation, is a significant segment within the metronidazole market. In the U.S., the annual sales for metronidazole gel totaled approximately $62 million for the 12 months ended June 2021[3].
Key Players and Competitive Landscape
The metronidazole market is competitive, with several key players:
- Hongyuan Pharmaceutical: A major player in the global metronidazole market[1][4].
- Yinhe Aarti Pharmaceutical: Another significant player contributing to the market's growth[4].
- Bausch Health: The original manufacturer of Metrogel-Vaginal, which has seen generic competition[3].
The approval of generic versions, such as the one by Solaris Pharma, has introduced new competitors into the market, potentially affecting the financial trajectory of branded products like Metrogel-Vaginal[3].
Financial Highlights
Revenue and Sales
- The U.S. annual sales for metronidazole gel, specifically Metrogel-Vaginal, were approximately $62 million for the 12 months ended June 2021[3].
- The broader metronidazole market's revenue figures are expected to vary, with some forecasts indicating a decline in the short term but significant growth in the long term[1][4].
Generic Competition Impact
The approval of generic versions of Metrogel-Vaginal, such as the one by Solaris Pharma, can significantly impact the revenue of the original branded product. Generic competition often leads to price reductions and market share shifts, affecting the financial performance of the original manufacturer[3].
Research and Development
Research and development expenses are crucial for pharmaceutical companies. For example, Aclaris Therapeutics, though not directly involved in Metrogel, spent $48.0 million on research and development in 2018, highlighting the importance of R&D in the pharmaceutical sector[2].
Market Drivers and Challenges
Drivers
- Increasing Demand: The rise in bacterial and protozoal infections drives the demand for metronidazole products like Metrogel[1].
- Animal Health: The increasing use of metronidazole in veterinary medicine is another driving factor[4].
Challenges
- Generic Competition: The entry of generic versions can reduce the market share and revenue of branded products like Metrogel-Vaginal[3].
- Regulatory Approvals: Obtaining and maintaining regulatory approvals is a significant challenge in the pharmaceutical industry[3].
Regional Market Insights
The metronidazole market, including Metrogel, has a global presence but varies by region. For instance, the U.S. market for metronidazole gel is substantial, with annual sales of $62 million. Emerging markets, such as China, India, and Japan, also play a crucial role in the overall market dynamics[5].
Conclusion
The market dynamics and financial trajectory of Metrogel are influenced by various factors, including generic competition, regulatory approvals, and global demand. While the short-term forecast indicates a decline, long-term projections suggest significant growth driven by increasing demand and expanding applications.
Key Takeaways
- Global Market Size: The global metronidazole market was valued at $113 million in 2021 but is forecast to decline in the short term.
- Generic Competition: The approval of generic versions of Metrogel-Vaginal impacts the revenue of the original branded product.
- Long-term Growth: The market is expected to grow significantly by 2032, driven by increasing demand and veterinary applications.
- Key Players: Hongyuan Pharmaceutical, Yinhe Aarti Pharmaceutical, and Bausch Health are significant players in the market.
FAQs
-
What is the current market size of the global metronidazole market?
- The global metronidazole market size was estimated to be worth $113 million in 2021[1].
-
What is the forecasted growth rate of the metronidazole market by 2032?
- The metronidazole market is expected to exhibit a CAGR of 5.96% by 2032[4].
-
Who are the key players in the metronidazole market?
- Key players include Hongyuan Pharmaceutical, Yinhe Aarti Pharmaceutical, and Bausch Health[1][4].
-
What is the impact of generic competition on Metrogel-Vaginal?
- Generic competition can reduce the market share and revenue of the original branded product, Metrogel-Vaginal[3].
-
What are the driving factors for the metronidazole market?
- The increase in bacterial and protozoal infections and the use of metronidazole in veterinary medicine are key driving factors[1][4].
Sources
- Biospace: Metronidazole Market Size, Growth, Trends, Report 2022-2030
- Aclaris Therapeutics: Aclaris Therapeutics Reports Fourth Quarter and Full Year 2018
- Biospace: Solaris Pharma Receives FDA Approval for Generic Metrogel-Vaginal
- Business Research Insights: Metronidazole Market Report | Global Forecast To 2032
- Bausch Health: 2018 Annual Report