nextstellis Drug Patent Profile
✉ Email this page to a colleague
When do Nextstellis patents expire, and what generic alternatives are available?
Nextstellis is a drug marketed by Mayne Pharma and is included in one NDA. There are four patents protecting this drug.
This drug has two hundred and two patent family members in forty-seven countries.
The generic ingredient in NEXTSTELLIS is drospirenone; estetrol. There are eleven drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the drospirenone; estetrol profile page.
DrugPatentWatch® Generic Entry Outlook for Nextstellis
Nextstellis will be eligible for patent challenges on April 15, 2025. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 15, 2026. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for nextstellis?
- What are the global sales for nextstellis?
- What is Average Wholesale Price for nextstellis?
Summary for nextstellis
| International Patents: | 202 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 32 |
| Clinical Trials: | 1 |
| Drug Prices: | Drug price information for nextstellis |
| What excipients (inactive ingredients) are in nextstellis? | nextstellis excipients list |
| DailyMed Link: | nextstellis at DailyMed |
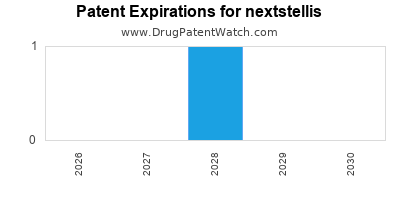
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for nextstellis
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for nextstellis
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| McGill University Health Centre/Research Institute of the McGill University Health Centre | Phase 4 |
US Patents and Regulatory Information for nextstellis
nextstellis is protected by four US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of nextstellis is ⤷ Sign Up.
This potential generic entry date is based on NEW CHEMICAL ENTITY.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting nextstellis
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Drug delivery system comprising a tetrahydroxilated estrogen for use in hormonal contraception
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: USE BY FEMALES OF REPRODUCTIVE POTENTIAL TO PREVENT PREGNANCY
FDA Regulatory Exclusivity protecting nextstellis
NEW CHEMICAL ENTITY
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for nextstellis
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Estetra SPRL | Lydisilka | estetrol, drospirenone | EMEA/H/C/005382 Oral contraception.The decision to prescribe Lydisilka should take into consideration the individual woman’s current risk factors, particularly those for venous thromboembolism (VTE), and how the risk of VTE with Lydisilka compares with other combined hormonal contraceptives (CHCs) (see sections 4.3 and 4.4). |
Authorised | no | no | no | 2021-05-19 | |
| Gedeon Richter Plc. | Drovelis | estetrol, drospirenone | EMEA/H/C/005336 oral contraceptive |
Authorised | no | no | no | 2021-05-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for nextstellis
See the table below for patents covering nextstellis around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Denmark | 1390040 | ⤷ Sign Up | |
| Lithuania | PA2022508 | ⤷ Sign Up | |
| Hungary | E037383 | ⤷ Sign Up | |
| Ecuador | SP17085669 | UNIDAD DE DOSIFICACIÓN ORODISPERSABLE QUE CONTIENE UN COMPONENTE ESTETROL | ⤷ Sign Up |
| European Patent Office | 1390042 | SYSTEME D'ADMINISTRATION DE MEDICAMENT COMPRENANT UN OESTROGENE TETRAHYDROXYLE DESTINE A UNE CONTRACEPTION HORMONALE (DRUG DELIVERY SYSTEM COMPRISING A TETRAHYDROXYLATED ESTROGEN FOR USE IN HORMONAL CONTRACEPTION) | ⤷ Sign Up |
| Denmark | 1446128 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for nextstellis
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3632448 | LUC00266 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: DROSPIRENONE; AUTHORISATION NUMBER AND DATE: 61678, 20210401 |
| 0398460 | 04C0022 | France | ⤷ Sign Up | PRODUCT NAME: ESTRADIOL ANHYDRE DROSPIRENONE; REGISTRATION NO/DATE IN FRANCE: NL 28661 DU 20040316; REGISTRATION NO/DATE AT EEC: RVG 27505 DU 20021211 |
| 3632448 | 22C1031 | France | ⤷ Sign Up | PRODUCT NAME: DROSPIRENONE; NAT. REGISTRATION NO/DATE: NL49691 20191121; FIRST REGISTRATION: DK - 61678 20191016 |
| 3701944 | PA2022508,C3701944 | Lithuania | ⤷ Sign Up | PRODUCT NAME: DROSPIRENONAS DERINYJE SU ESTETROLIU; REGISTRATION NO/DATE: EU/1/21/1547 20210519 |
| 2588114 | LUC00227 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: DROSPIRENONE; AUTHORISATION NUMBER AND DATE: 31332 20191022 |
| 0398460 | SPC/GB04/032 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: ESTRADIOL, OPTIONALLY IN THE FORM OF A HYDRATE, TOGETHER WITH DROSPIRENONE; REGISTERED: NL RVG 27505 20021211; UK PL 00053/0341 20040310 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


