twyneo Drug Patent Profile
✉ Email this page to a colleague
When do Twyneo patents expire, and when can generic versions of Twyneo launch?
Twyneo is a drug marketed by Galderma Labs Lp and is included in one NDA. There are seven patents protecting this drug.
This drug has sixty-three patent family members in thirteen countries.
The generic ingredient in TWYNEO is benzoyl peroxide; tretinoin. There are seventeen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the benzoyl peroxide; tretinoin profile page.
DrugPatentWatch® Generic Entry Outlook for Twyneo
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 30, 2030. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for twyneo?
- What are the global sales for twyneo?
- What is Average Wholesale Price for twyneo?
Summary for twyneo
| International Patents: | 63 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Drug Prices: | Drug price information for twyneo |
| What excipients (inactive ingredients) are in twyneo? | twyneo excipients list |
| DailyMed Link: | twyneo at DailyMed |
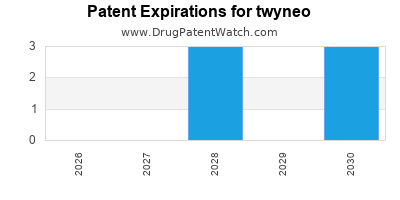

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for twyneo
Generic Entry Date for twyneo*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CREAM;TOPICAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for twyneo
| Drug Class | Retinoid |
US Patents and Regulatory Information for twyneo
twyneo is protected by seven US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of twyneo is ⤷ Subscribe.
This potential generic entry date is based on patent ⤷ Subscribe.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | TWYNEO | benzoyl peroxide; tretinoin | CREAM;TOPICAL | 214902-001 | Jul 26, 2021 | RX | Yes | Yes | ⤷ Subscribe | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Galderma Labs Lp | TWYNEO | benzoyl peroxide; tretinoin | CREAM;TOPICAL | 214902-001 | Jul 26, 2021 | RX | Yes | Yes | ⤷ Subscribe | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Galderma Labs Lp | TWYNEO | benzoyl peroxide; tretinoin | CREAM;TOPICAL | 214902-001 | Jul 26, 2021 | RX | Yes | Yes | ⤷ Subscribe | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for twyneo
When does loss-of-exclusivity occur for twyneo?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 10337830
Patent: Core stabilized microcapsules, method of their preparation and uses thereof
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 2012012023
Patent: processo para preparar microcápsulas, microcápsulas, composição, método para tratar uma condição superficial em um indivíduo, e, uso das microcápsulas
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 73544
Patent: MICROCAPSULES A NOYAU STABILISE, PROCEDE DE LEUR PREPARATION ET UTILISATIONS DE CELLES-CI (CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF)
Estimated Expiration: ⤷ Subscribe
China
Patent: 2596186
Patent: Core stabilized microcapsules, method of their preparation and uses thereof
Estimated Expiration: ⤷ Subscribe
Eurasian Patent Organization
Patent: 1200347
Patent: МИКРОКАПСУЛЫ СО СТАБИЛИЗИРОВАННЫМ ЯДРОМ, СПОСОБ ИХ ПОЛУЧЕНИЯ И ИХ ПРИМЕНЕНИЕ
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 67132
Patent: MICROCAPSULES À NOYAU STABILISÉ, PROCÉDÉ DE LEUR PRÉPARATION ET UTILISATIONS DE CELLES-CI (CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 34107
Estimated Expiration: ⤷ Subscribe
Patent: 13516404
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 4128
Patent: MICROCAPSULAS ESTABILIZADAS CON NUCLEO, METODO PARA SU PREPARACION Y USOS DE LAS MISMAS. (CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF.)
Estimated Expiration: ⤷ Subscribe
Patent: 12006636
Patent: MICROCAPSULAS ESTABILIZADAS CON NUCLEO, METODO PARA SU PREPARACION Y USOS DE LAS MISMA. (CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF.)
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 1203582
Patent: CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 130008002
Patent: CORE STABILIZED MICROCAPSULES, METHOD OF THEIR PREPARATION AND USES THEREOF
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 11499
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering twyneo around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Eurasian Patent Organization | 201200347 | ⤷ Subscribe | |
| Australia | 2006274541 | Metal oxide coating of water insoluble ingredients | ⤷ Subscribe |
| Eurasian Patent Organization | 200970724 | КОМПОЗИЦИИ ДЛЯ МЕСТНОГО ПРИМЕНЕНИЯ, СОДЕРЖАЩИЕ ПЕРОКСИД И РЕТИНОИД | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for twyneo
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1586316 | SPC/GB11/054 | United Kingdom | ⤷ Subscribe | PRODUCT NAME: BROMFENAC 2-AMINO-3-(4-BROMOBENZOYL)PHENYLACETIC ACID OR A PHARMACOLOGICALLY ACCEPTABLE SALT THEREOF OR A HYDRATE THEREOF; REGISTERED: UK EU/1/11/692/001 20110523 |
| 0591275 | SPC/GB05/030 | United Kingdom | ⤷ Subscribe | PRODUCT NAME: NITISINONE (2-(2-NITRO-4-TRIFLUOROMETHYLBENZOYL)-1,3-CYCLOHEXANEDIONE) OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: UK EU/1/04/303/001 20050221; UK EU/1/04/303/002 20050221; UK EU/1/04/303/003 20050221 |
| 1458369 | CA 2008 00029 | Denmark | ⤷ Subscribe | PRODUCT NAME: ADAPALEN, BENZOYLPEROXID |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Twyneo Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


