In the ever-evolving pharmaceutical industry, Alkermes Inc. has carved out a unique position as a specialized neuroscience company focused on developing innovative treatments for central nervous system (CNS) disorders and addiction. This comprehensive analysis delves into Alkermes' market position, strengths, and strategic insights, providing a detailed look at how the company navigates the competitive landscape of the pharmaceutical sector.
Company Overview
Alkermes Inc. is a biopharmaceutical company headquartered in Dublin, Ireland, with significant operations in the United States. The company's primary focus is on developing and commercializing therapies for CNS disorders, including schizophrenia, bipolar disorder, and addiction[10]. With a history dating back to 1987, Alkermes has established itself as a key player in the neuroscience space, leveraging its expertise in drug development and delivery technologies.
Market Position
Current Standing
As of 2024, Alkermes has solidified its position as a mid-sized pharmaceutical company with a specialized focus. The company's market capitalization stood at approximately $2.1 billion in Q4 2023, placing it in the small-cap category compared to pharmaceutical giants[8]. However, Alkermes' targeted approach and focus on CNS disorders have allowed it to compete effectively in its chosen therapeutic areas.
Key Products and Revenue Streams
Alkermes' revenue is primarily driven by three main products:
- VIVITROL: For opioid and alcohol addiction treatment
- ARISTADA: For schizophrenia treatment
- LYBALVI: For bipolar disorder and schizophrenia[7]
In 2024, Alkermes reported total revenues of $1.56 billion, with net sales of proprietary products increasing by approximately 18% year-over-year[7]. This growth demonstrates the company's ability to expand its market share in key therapeutic areas.
Market Share in Target Segments
While specific market share data for individual products is not provided in the search results, we can infer from the revenue growth and product performance that Alkermes has been gaining traction in its target segments. For instance, LYBALVI, a relatively new addition to the company's portfolio, achieved sales of $56.2 million in Q4 2024, showing a 61% year-over-year increase[1].
Competitive Strengths
Specialized Expertise in CNS Disorders
Alkermes' primary strength lies in its deep neuroscience expertise and focus on CNS and addiction treatment medications. This specialization allows the company to develop targeted therapies for complex and often underserved patient populations[10].
Diverse Product Portfolio
The company boasts a diverse portfolio of both commercial and development-stage pharmaceutical products. As of 2024, Alkermes has:
- 7 commercial-stage products
- 4 clinical-stage development programs
- Multiple preclinical stage candidates[8]
This diverse pipeline helps mitigate risks associated with product failures and provides multiple avenues for growth.
Robust R&D Capabilities
Alkermes' commitment to innovation is evident in its substantial R&D investments. In 2023, the company spent $303.7 million on R&D, employing approximately 500 researchers[8]. This focus on research and development enables Alkermes to maintain a competitive edge in drug discovery and development.
Strategic Partnerships
Collaborations with major pharmaceutical companies, including Johnson & Johnson, AstraZeneca, and Merck, enhance Alkermes' capabilities and market reach[8]. These partnerships provide access to additional resources, expertise, and potential revenue streams.
Manufacturing Capabilities
Alkermes operates state-of-the-art manufacturing facilities that adhere to stringent regulatory standards. This in-house manufacturing capability ensures quality control and supply chain reliability, giving the company a competitive advantage in product delivery and consistency[2].
Strategic Insights
Focus on High-Growth Therapeutic Areas
Alkermes' strategic focus on CNS disorders and addiction treatment aligns with growing market demand. The global CNS therapeutics market is projected to reach $150.2 billion by 2030, growing at a CAGR of 4.6%[8]. By concentrating on these high-growth areas, Alkermes positions itself to capture a significant share of this expanding market.
Emphasis on Innovation and Pipeline Development
The company's ongoing investment in R&D and its diverse pipeline demonstrate a commitment to long-term growth. Alkermes is currently conducting Phase 2 studies for ALKS 2680, a potential treatment for narcolepsy, with data expected in H2 2025[7]. This focus on pipeline development ensures a steady stream of potential new products to drive future growth.
Strategic Repositioning
Alkermes has recently completed a multi-year effort to transition into a pure-play neuroscience company. This strategic repositioning, including the divestiture of its manufacturing business in Ireland, allows the company to focus on high-margin proprietary products and streamline operations[5].
Financial Strategy
The company's debt-free status and robust cash position provide financial flexibility for both organic growth and potential strategic opportunities in the neuroscience space[5]. This strong financial foundation enables Alkermes to invest in R&D, pursue strategic partnerships, and navigate market challenges effectively.
Competitive Challenges
Market Competition
Alkermes faces significant competition in its target therapeutic areas. For example:
- In schizophrenia treatment, the company competes with established players like Janssen and Otsuka Pharmaceutical
- In addiction treatment, key competitors include Indivior and Braeburn Pharmaceuticals[8]
This intense competition necessitates continuous innovation and effective marketing strategies to maintain and grow market share.
Regulatory Hurdles
The pharmaceutical industry is subject to stringent regulatory approval processes, which can impact drug development timelines and costs. With an average regulatory review time of 18-24 months and estimated compliance costs of $36.2 million annually, navigating these regulatory challenges is crucial for Alkermes' success[8].
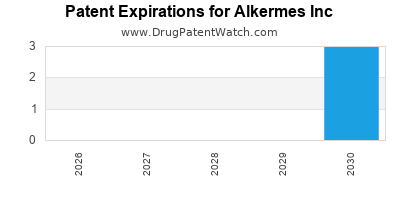
Patent Expirations
Like all pharmaceutical companies, Alkermes faces the threat of patent expirations and generic competition. Key patents for VIVITROL and ARISTADA are set to expire in 2028 and 2030, respectively, potentially impacting future revenues[8].
Future Outlook and Growth Opportunities
Expansion into New Therapeutic Areas
Alkermes' existing drug delivery platforms present opportunities for expansion into additional therapeutic domains, including:
- Neurological disorders
- Psychiatric conditions
- Chronic pain management
- Oncology supportive care[8]
This diversification could help mitigate risks associated with the company's current focus on CNS disorders and addiction treatment.
Emerging Markets
The pharmaceutical industry is experiencing significant growth in emerging markets, with regions like Asia-Pacific, the Middle East and Africa, and Latin America showing strong growth potential[8]. Alkermes could leverage these opportunities to expand its global footprint and drive future growth.
Strategic Partnerships and Collaborations
Continuing to pursue strategic partnerships and collaborations could provide Alkermes with access to new technologies, markets, and resources. Potential areas for collaboration include:
- Joint research and development initiatives
- Licensing agreements for novel compounds
- Co-marketing arrangements for existing products
Digital Health Integration
As the healthcare industry increasingly embraces digital technologies, Alkermes has the opportunity to integrate digital health solutions into its product offerings. This could include developing companion apps for medication adherence, leveraging AI for drug discovery, or exploring telemedicine options for patient support.
"2024 marked the completion of a multi-year effort to transition the business into a highly profitable, pure-play neuroscience company. We enter 2025 with a diversified portfolio of proprietary commercial products generating substantial profitability and an advancing development pipeline that represents a significant value creation opportunity in one of the most exciting potential new therapeutic categories in neuroscience," said Richard Pops, Chief Executive Officer of Alkermes[7].
Key Takeaways
- Alkermes has successfully positioned itself as a specialized neuroscience company with a focus on CNS disorders and addiction treatment.
- The company's diverse product portfolio and robust R&D capabilities provide a strong foundation for future growth.
- Strategic repositioning as a pure-play neuroscience company has enhanced profitability and operational focus.
- Alkermes faces competitive challenges but has opportunities for expansion in emerging markets and new therapeutic areas.
- The company's financial strength and strategic partnerships provide flexibility for future growth initiatives.
FAQs
-
Q: What are Alkermes' main revenue-generating products?
A: Alkermes' main revenue-generating products are VIVITROL (for addiction treatment), ARISTADA (for schizophrenia), and LYBALVI (for bipolar disorder and schizophrenia).
-
Q: How does Alkermes' market capitalization compare to larger pharmaceutical companies?
A: As of Q4 2023, Alkermes' market capitalization was approximately $2.1 billion, significantly smaller than pharmaceutical giants like Pfizer ($270 billion) and Johnson & Johnson ($430 billion).
-
Q: What is Alkermes' approach to research and development?
A: Alkermes invests heavily in R&D, spending $303.7 million in 2023 and employing approximately 500 researchers to maintain its competitive edge in drug discovery and development.
-
Q: How has Alkermes strategically repositioned itself in recent years?
A: Alkermes has completed a multi-year effort to transition into a pure-play neuroscience company, divesting its manufacturing business in Ireland to focus on high-margin proprietary products.
-
Q: What are some potential growth opportunities for Alkermes in the coming years?
A: Potential growth opportunities for Alkermes include expansion into new therapeutic areas, penetration of emerging markets, strategic partnerships and collaborations, and integration of digital health solutions.
Sources cited:
[1] https://www.fiercepharma.com/pharma/good-news-lybalvi-alkermes-sees-10-stock-price-increase
[2] https://pitchgrade.com/companies/alkermes-plc
[5] https://www.stocktitan.net/news/ALKS/alkermes-plc-reports-financial-results-for-the-fourth-quarter-and-c2259z6z116k.html
[7] https://investor.alkermes.com/news-releases/news-release-details/alkermes-plc-reports-financial-results-fourth-quarter-and-year-4
[8] https://dcf.fm/products/alks-swot-analysis
[10] https://www.alkermes.com/about-us