In the ever-evolving pharmaceutical industry, AstraZeneca stands as a formidable player, consistently pushing the boundaries of innovation and growth. As we delve into the competitive landscape surrounding this pharmaceutical giant, we'll explore its market position, strengths, and strategic insights that have propelled it to the forefront of the industry.
AstraZeneca's Market Position
AstraZeneca has solidified its position as a leading global biopharmaceutical company, known for its innovative approach to drug discovery and development[2]. With operations spanning over 100 countries, the company has established a strong global presence, allowing it to tap into diverse markets and cater to a broad range of patient needs[2].
Revenue Growth and Future Ambitions
AstraZeneca's market position is further strengthened by its impressive revenue growth. In 2023, the company reported total revenue of $45.8 billion, up from $44.4 billion in the previous year[1]. This growth trajectory has set the stage for even more ambitious goals. AstraZeneca has unveiled its bold vision to achieve $80 billion in total revenue by 2030, nearly doubling its current figures[1].
"Today AstraZeneca announces a new era of growth. In 2023 we delivered the ambitious $45 billion revenue goal set a decade ago. With the exciting growth of our innovative pipeline, which has the potential to transform millions of lives, we are now aiming for $80 billion by 2030." - Pascal Soriot, Chief Executive Officer, AstraZeneca[1]
This ambitious target underscores AstraZeneca's confidence in its pipeline and strategic direction, positioning it as a company with significant growth potential in the coming years.
Global Market Share
AstraZeneca's global market share is substantial, with the company ranking 8th globally based on prescription drug sales in 2023[5]. This positioning places AstraZeneca among the top-tier pharmaceutical companies worldwide, competing with industry giants like Pfizer, Merck, and GlaxoSmithKline.
AstraZeneca's Key Strengths
AstraZeneca's competitive edge in the pharmaceutical landscape is built on several key strengths that set it apart from its rivals.
Strong Product Portfolio
One of AstraZeneca's primary strengths lies in its robust and diverse product portfolio. The company boasts several blockbuster drugs across various therapeutic areas, including oncology, cardiovascular, renal, metabolism, respiratory, and immunology[3]. This diversification not only contributes to the company's financial stability but also positions it to address a wide range of medical needs.
Robust Research and Development Pipeline
AstraZeneca's commitment to innovation is evident in its strong R&D pipeline. The company invests heavily in research and development, with spending well above 20% of its revenues on R&D projects[5]. This focus on innovation has resulted in a pipeline of over 60 molecules in clinical development[3], promising a steady stream of new products in the coming years.
Global Presence and Market Reach
With operations in over 100 countries, AstraZeneca's global footprint is a significant strength[2]. This extensive reach allows the company to navigate diverse regulatory environments, tap into emerging markets, and maintain a strong presence in established pharmaceutical markets.
Strategic Partnerships and Collaborations
AstraZeneca has fostered strategic partnerships with academic institutions, research organizations, and other pharmaceutical companies[2]. These collaborations enhance the company's capabilities, accelerate drug development processes, and provide access to cutting-edge technologies and expertise.
Strategic Insights: AstraZeneca's Path Forward
AstraZeneca's strategic direction is shaped by several key insights and initiatives that are likely to drive its future growth and competitive position.
Focus on High-Growth Therapeutic Areas
AstraZeneca has strategically focused on high-growth therapeutic areas, particularly oncology. The company's oncology portfolio has shown impressive growth, with sales increasing by 20% in 2023[9]. This focus on areas with significant unmet medical needs positions AstraZeneca to capture growing market segments.
Emphasis on Personalized Medicine
AstraZeneca is at the forefront of personalized medicine, developing treatments tailored to individual patient profiles[2]. This approach not only improves patient outcomes but also differentiates AstraZeneca in an increasingly competitive market.
Expansion into Rare Diseases
The $39 billion acquisition of Alexion in 2021 marked AstraZeneca's strategic entry into the rare disease market[9]. This move has diversified the company's portfolio and opened up new growth opportunities in a specialized and potentially lucrative segment of the pharmaceutical industry.
Commitment to Sustainability
AstraZeneca's focus on sustainability and corporate social responsibility resonates with consumers and investors alike[2]. This commitment not only enhances the company's reputation but also aligns with growing global emphasis on sustainable business practices.
Navigating Challenges in the Competitive Landscape
While AstraZeneca's position is strong, the company faces several challenges in the competitive pharmaceutical landscape.
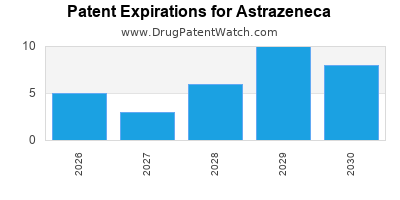
Patent Expirations
Like many pharmaceutical companies, AstraZeneca must navigate the challenge of patent expirations for key products. The upcoming patent expirations for blockbuster drugs like Farxiga and Tagrisso present a significant hurdle in maintaining revenue growth[9].
Intense Competition in Key Therapeutic Areas
AstraZeneca faces stiff competition in several of its key therapeutic areas, particularly in oncology[8]. As a relatively late entrant in some fields, the company must continually innovate to differentiate its offerings and capture market share.
Regulatory and Pricing Pressures
The pharmaceutical industry as a whole faces increasing regulatory scrutiny and pressure to control drug prices. AstraZeneca must navigate these challenges while maintaining profitability and funding its extensive R&D efforts.
AstraZeneca's Competitive Advantages
Several factors contribute to AstraZeneca's competitive edge in the pharmaceutical market:
Strong Brand Reputation
AstraZeneca has built a strong brand reputation over the years, known for delivering high-quality, innovative medicines[2]. This reputation instills trust among healthcare professionals, patients, and investors.
Diverse Geographic Revenue Stream
AstraZeneca's revenue is well-distributed geographically, with significant contributions from various regions. In 2023, the US market accounted for 44% of the company's total revenue, highlighting the importance of this key market while also demonstrating the company's global reach[10].
Effective Corporate Strategy
AstraZeneca's corporate strategy effectively aligns with changing market dynamics, focusing on areas with high unmet medical needs[7]. This strategic alignment positions the company to capitalize on emerging opportunities in the pharmaceutical landscape.
Future Outlook and Growth Strategies
Looking ahead, AstraZeneca's growth strategy centers on several key initiatives:
Ambitious Product Launch Plans
AstraZeneca plans to launch 20 new medicines by 2030, many with the potential to generate more than $5 billion in peak year revenues[1]. This aggressive pipeline strategy is central to the company's ambitious revenue growth targets.
Continued Investment in R&D
AstraZeneca remains committed to investing heavily in research and development, maintaining its position at the forefront of pharmaceutical innovation[5].
Strategic M&A and Partnerships
While organic growth is a priority, AstraZeneca's history suggests that strategic mergers, acquisitions, and partnerships will continue to play a role in expanding its capabilities and market reach[9].
Expansion in Emerging Markets
AstraZeneca is likely to continue its focus on expanding its presence in emerging markets, capitalizing on growing healthcare needs and increasing access to medicines in these regions.
Key Takeaways
- AstraZeneca holds a strong market position, ranking 8th globally in prescription drug sales.
- The company aims to achieve $80 billion in total revenue by 2030, driven by a robust pipeline and strategic focus on high-growth therapeutic areas.
- Key strengths include a diverse product portfolio, strong R&D capabilities, global presence, and strategic partnerships.
- Challenges include patent expirations, intense competition, and regulatory pressures.
- Future growth strategies center on ambitious product launches, continued R&D investment, strategic M&A, and expansion in emerging markets.
FAQs
-
What is AstraZeneca's current market position in the pharmaceutical industry?
AstraZeneca ranks 8th globally based on prescription drug sales as of 2023, with a strong presence in key therapeutic areas such as oncology, cardiovascular, and respiratory diseases.
-
How does AstraZeneca plan to achieve its $80 billion revenue target by 2030?
AstraZeneca plans to launch 20 new medicines by 2030, many with potential for over $5 billion in peak year revenues. The company will also continue investing in R&D and focus on high-growth therapeutic areas.
-
What are some of AstraZeneca's key competitive advantages?
AstraZeneca's competitive advantages include a strong product portfolio, robust R&D pipeline, global presence, strategic partnerships, and a focus on personalized medicine and rare diseases.
-
How is AstraZeneca addressing the challenge of patent expirations?
While specific strategies aren't detailed in the provided information, pharmaceutical companies typically address patent expirations through developing new drugs, extending existing patents through new formulations or indications, and diversifying their product portfolio.
-
What role does sustainability play in AstraZeneca's strategy?
AstraZeneca has a strong commitment to sustainability and corporate social responsibility, which enhances its reputation and aligns with growing global emphasis on sustainable business practices.
Sources cited:
[1] https://www.astrazeneca.com/media-centre/press-releases/2024/astrazeneca-to-deliver-80bn-revenue-by-2030.html
[2] https://canvasbusinessmodel.com/blogs/competitors/astrazeneca-competitive-landscape
[3] https://www.studentsassignmenthelp.co.uk/answers/astrazeneca-uk-swot-analysis/
[5] https://www.statista.com/topics/7584/astrazeneca/
[7] https://thestrategystory.com/blog/astrazeneca-swot-analysis/
[8] https://www.investing.com/news/swot-analysis/astrazenecas-swot-analysis-pharma-giants-stock-poised-for-growth-amid-challenges-93CH-3838445
[9] https://www.fiercepharma.com/pharma/astrazeneca-fast-track-growth-sets-target-revenue-80b-2030
[10] https://www.astrazeneca.com/media-centre/press-releases/2024/astrazeneca-invests-3bn-500mn-in-us.html