ENDO OPERATIONS Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ENDO OPERATIONS, and when can generic versions of ENDO OPERATIONS drugs launch?
ENDO OPERATIONS has two hundred and twenty-one approved drugs.
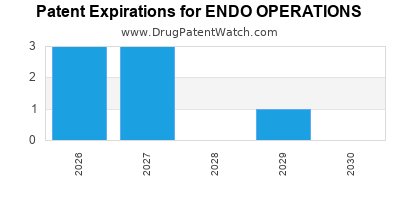
There are forty-two US patents protecting ENDO OPERATIONS drugs. There is one tentative approval on ENDO OPERATIONS drugs.
There are one hundred and nineteen patent family members on ENDO OPERATIONS drugs in forty countries and three hundred and fifty-three supplementary protection certificates in seventeen countries.
Summary for ENDO OPERATIONS
| International Patents: | 119 |
| US Patents: | 42 |
| Tradenames: | 176 |
| Ingredients: | 155 |
| NDAs: | 221 |
Drugs and US Patents for ENDO OPERATIONS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Endo Operations | DANTRIUM | dantrolene sodium | INJECTABLE;INJECTION | 018264-001 | Approved Prior to Jan 1, 1982 | AP | RX | Yes | Yes | ⤷ Subscribe | ⤷ Subscribe | ||||
| Endo Operations | DICLOFENAC POTASSIUM | diclofenac potassium | FOR SOLUTION;ORAL | 202964-001 | May 2, 2016 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Endo Operations | CALCITONIN-SALMON | calcitonin salmon | INJECTABLE;INJECTION | 209358-001 | Nov 10, 2021 | AP | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Endo Operations | GLIPIZIDE | glipizide | TABLET, EXTENDED RELEASE;ORAL | 076159-002 | Sep 20, 2013 | DISCN | No | No | ⤷ Subscribe | ⤷ Subscribe | |||||
| Endo Operations | HYDROXYUREA | hydroxyurea | CAPSULE;ORAL | 075340-001 | Feb 24, 1999 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Endo Operations | TRAMADOL HYDROCHLORIDE | tramadol hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 200491-003 | Jun 27, 2012 | DISCN | No | No | ⤷ Subscribe | ⤷ Subscribe | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ENDO OPERATIONS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Endo Operations | SUMAVEL DOSEPRO | sumatriptan succinate | INJECTABLE;SUBCUTANEOUS | 022239-001 | Jul 15, 2009 | 8,267,903 | ⤷ Subscribe |
| Endo Operations | MEGACE ES | megestrol acetate | SUSPENSION;ORAL | 021778-001 | Jul 5, 2005 | 5,145,684 | ⤷ Subscribe |
| Endo Operations | OPANA ER | oxymorphone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 021610-007 | Feb 29, 2008 | 8,309,112 | ⤷ Subscribe |
| Endo Operations | NASCOBAL | cyanocobalamin | SPRAY, METERED;NASAL | 021642-001 | Jan 31, 2005 | 7,229,636 | ⤷ Subscribe |
| Endo Operations | FROVA | frovatriptan succinate | TABLET;ORAL | 021006-001 | Nov 8, 2001 | 5,464,864 | ⤷ Subscribe |
| Endo Operations | OPANA ER | oxymorphone hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 021610-003 | Jun 22, 2006 | 5,662,933 | ⤷ Subscribe |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ENDO OPERATIONS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 20 units/mL, 1 mL | ➤ Subscribe | 2018-03-23 |
| ➤ Subscribe | Extended-release Tablets | 7.5 mg and 15 mg | ➤ Subscribe | 2008-05-29 |
| ➤ Subscribe | Tablets | 2.5 mg | ➤ Subscribe | 2011-03-09 |
| ➤ Subscribe | Nasal Spray | 500 mcg/spray | ➤ Subscribe | 2017-04-28 |
| ➤ Subscribe | Gel | 1% | ➤ Subscribe | 2008-08-21 |
| ➤ Subscribe | Injection | 1 mg/mL, 50 mL vials | ➤ Subscribe | 2011-12-16 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Injection | 1 mg/mL ampules | ➤ Subscribe | 2016-03-09 |
| ➤ Subscribe | Extended-release Tablets | 5 mg, 10 mg, 20 mg and 40 mg | ➤ Subscribe | 2007-11-23 |
| ➤ Subscribe | Extended-release Tablets | 30 mg | ➤ Subscribe | 2008-06-12 |
| ➤ Subscribe | Gel | 10 mg/actuation | ➤ Subscribe | 2012-08-14 |
| ➤ Subscribe | Injection | 100 mg/mL, 2.5 mL vials | ➤ Subscribe | 2007-09-24 |
| ➤ Subscribe | Injection | 250 mg/mL | ➤ Subscribe | 2014-06-11 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
International Patents for ENDO OPERATIONS Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| South Korea | 20050109561 | ⤷ Subscribe |
| Jordan | 2505 | ⤷ Subscribe |
| Portugal | 1494649 | ⤷ Subscribe |
| Brazil | PI0400214 | ⤷ Subscribe |
| Israel | 188322 | ⤷ Subscribe |
| Slovenia | 1457208 | ⤷ Subscribe |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for ENDO OPERATIONS Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1507558 | 2012/018 | Ireland | ⤷ Subscribe | PRODUCT NAME: ALISKIREN OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, AMLODIPINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF AND HYDROCHLOROTHIAZIDE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; NAT REGISTRATION NO/DATE: EU/1/11/730/001-060 20111122; FIRST REGISTRATION NO/DATE: SWITZERLAND 6167801-6167805 20110705 |
| 0285237 | 95C0008 | Belgium | ⤷ Subscribe | PRODUCT NAME: ESTRADIOL, HEMIHYDRATE; NAT. REGISTRATION NO/DATE: NL 19489 19941107; FIRST REGISTRATION: FR - NL 19489 19941107 |
| 0678503 | C00678503/04 | Switzerland | ⤷ Subscribe | PRODUCT NAME: ALISKIREN + AMLODIPIN; REGISTRATION NO/DATE: SWISSMEDIC 61454 05.07.2011 |
| 3342411 | LUC00148 | Luxembourg | ⤷ Subscribe | PRODUCT NAME: EVEROLIMUS; AUTHORISATION NUMBER AND DATE: EU/1/09/538/001-008 20110830 |
| 1441735 | SPC/GB08/020 | United Kingdom | ⤷ Subscribe | PRODUCT NAME: RALTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, ESPECIALLY THE POTASSIUM SALT; REGISTERED: UK EU/1/07/436/001 20080102 |
| 2465580 | SPC/GB21/030 | United Kingdom | ⤷ Subscribe | PRODUCT NAME: CABOTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF, INCLUDING CABOTEGRAVIR SODIUM.; REGISTERED: UK EU/1/20/1481 (NI) 20201221; UK PLGB 35728/0055-57 20201221 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.