Tris Pharma Inc. has emerged as a significant player in the pharmaceutical industry, particularly in the development and commercialization of innovative medicines for unmet patient needs. This analysis delves into Tris Pharma's market position, strengths, and strategic insights, providing a comprehensive overview of the company's competitive landscape.
Company Overview
Tris Pharma is a privately-owned biopharmaceutical company based in the United States. The company focuses on developing and commercializing innovative medicines that address unmet patient needs, particularly in neuroscience and other therapeutic categories[6]. With a robust portfolio of FDA-approved products and a strong pipeline of innovative candidates, Tris Pharma has established itself as a leader in the pharmaceutical industry.
Key Facts
- Founded: Not specified in the search results
- Headquarters: Monmouth Junction, New Jersey
- Employees: Approximately 500
- Focus Areas: ADHD and related disorders, neuroscience, and other therapeutic categories
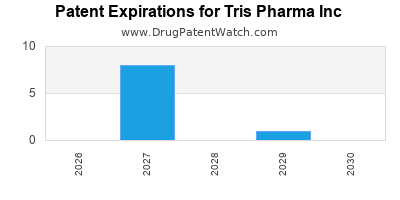
- Patents: More than 150 U.S. and International patents including applications[6]
Product Portfolio and Pipeline
Tris Pharma's product portfolio includes both branded and generic products, with a particular focus on ADHD treatments. The company has successfully developed and commercialized several innovative medications that offer unique benefits to patients.
ADHD Medications
Tris Pharma has established a strong presence in the ADHD market with its FDA-approved products:
- Quillivant XR: A long-acting, once-daily liquid methylphenidate formulation
- Quillichew ER: A long-acting, once-daily chewable methylphenidate formulation[7]
These products address specific challenges faced by ADHD patients, particularly younger individuals who may have difficulty swallowing pills or require customized dosing options.
Generic Products
In addition to its branded portfolio, Tris Pharma has a robust generics division focused on creating high-quality, patient-friendly products. The company is a market leader in many of its product categories and has a pipeline of 20+ generic products in development[2].
Pipeline and Innovation
Tris Pharma's commitment to innovation is evident in its robust pipeline of products. The company has several ANDAs and two NDAs pending FDA approval[1]. This pipeline positions Tris Pharma for future growth and demonstrates its ongoing commitment to addressing unmet medical needs.
Manufacturing Capabilities and Expansion
Tris Pharma has been actively expanding its manufacturing capabilities to support its growing product portfolio and pipeline.
Recent Expansion
In July 2023, Tris Pharma announced a significant expansion of its manufacturing capacity:
- Initiated a 15-month expansion project
- Current footprint: 90,000 sq. ft.
- Projected footprint after expansion: Approximately 200,000 sq. ft.
- First phase expected to double cGMP capacity[1]
This expansion is crucial for Tris Pharma to meet the increasing demand for its products and support its growth trajectory.
Strategic Partnerships and Collaborations
Tris Pharma has demonstrated a strategic approach to partnerships and collaborations, which has been instrumental in expanding its market reach and enhancing its product offerings.
Neuraxpharm Group Partnership
In December 2020, Tris Pharma announced a partnership with Neuraxpharm Group, a leading European specialty pharmaceutical company focused on CNS:
- Neuraxpharm received exclusive rights to commercialize Quillivant XR and Quillichew ER in Europe
- This partnership expands Tris Pharma's reach into the European market, one of the largest pharmaceutical markets globally[7]
"As one of the largest pharmaceutical markets in the world, Europe has been a high priority for Tris as we continue to strive to make our differentiated product portfolio available to patients globally," said Ketan Mehta, Tris' Founder and CEO[7].
This partnership highlights Tris Pharma's strategy of leveraging strategic collaborations to expand its global footprint and maximize the value of its innovative products.
Leadership and Management
Tris Pharma has been strengthening its leadership team to support its growth and expansion strategies.
Recent Appointments
In May 2023, Tris Pharma announced two key appointments to its leadership team:
- Thomas Englese: Appointed as Chief Commercial Officer
- Marc Lesnick, Ph.D.: Appointed as Chief Development Officer[4][8]
These appointments reflect Tris Pharma's commitment to enhancing its commercial success and advancing its clinical pipeline.
Scientific Advisory Board
In October 2020, Tris Pharma launched its Scientific Advisory Board (SAB). The purpose of the SAB is to help the company achieve its emerging clinical research and product development pipeline goals[9]. This initiative demonstrates Tris Pharma's commitment to scientific excellence and its focus on advancing innovative therapies.
Market Position and Competitive Landscape
Tris Pharma operates in a highly competitive pharmaceutical landscape, particularly in the generics and specialty pharmaceutical sectors. While specific market share data for Tris Pharma is not provided in the search results, we can infer its competitive position based on available information.
Strengths
-
Innovative ADHD Therapies: Tris Pharma's unique formulations of methylphenidate (Quillivant XR and Quillichew ER) provide a competitive edge in the ADHD market[7].
-
Robust Generic Portfolio: The company is a market leader in many of its product categories within the generics space[2].
-
Strong Pipeline: With several ANDAs and NDAs pending approval, Tris Pharma is well-positioned for future growth[1].
-
Manufacturing Expansion: The ongoing expansion of manufacturing capacity demonstrates Tris Pharma's commitment to meeting market demand and supporting growth[1].
-
Strategic Partnerships: Collaborations like the one with Neuraxpharm Group enable Tris Pharma to expand its global reach[7].
Competitive Landscape
While Tris Pharma has established a strong position in certain niche markets, it faces competition from larger pharmaceutical companies, particularly in the generics space. Some key competitors in the generics market include:
- Teva
- Mylan (Viatris)
- Sandoz (Novartis)
- Sun Pharma[10]
These companies have significant market presence and resources, which could pose challenges for Tris Pharma in certain market segments.
Strategic Insights and Future Outlook
Based on the available information, we can identify several key strategic insights for Tris Pharma:
-
Focus on Differentiated Products: Tris Pharma's success with innovative ADHD formulations suggests that continuing to develop unique, patient-friendly products could be a key growth driver.
-
Expansion of Manufacturing Capacity: The ongoing expansion project indicates that Tris Pharma is preparing for increased demand and potential new product launches.
-
Strategic Partnerships: Collaborations like the one with Neuraxpharm Group can help Tris Pharma expand its global reach and maximize the value of its product portfolio.
-
Investment in R&D: The robust pipeline and establishment of a Scientific Advisory Board demonstrate Tris Pharma's commitment to ongoing innovation.
-
Strengthening Leadership: Recent appointments to the leadership team suggest that Tris Pharma is positioning itself for growth and expansion.
Key Takeaways
- Tris Pharma has established a strong position in the ADHD market with innovative formulations.
- The company's generics division is a market leader in several product categories.
- Ongoing manufacturing expansion and a robust pipeline position Tris Pharma for future growth.
- Strategic partnerships are enabling global expansion and market penetration.
- Investment in R&D and leadership strengthening demonstrate a commitment to long-term growth and innovation.
While Tris Pharma faces competition from larger pharmaceutical companies, its focus on innovation, strategic partnerships, and manufacturing expansion provide a solid foundation for continued growth and market presence.
FAQs
-
What are Tris Pharma's main product offerings?
Tris Pharma's main product offerings include ADHD medications such as Quillivant XR and Quillichew ER, as well as a range of generic products across various therapeutic categories.
-
How is Tris Pharma expanding its manufacturing capabilities?
Tris Pharma is undergoing a 15-month expansion project that will increase its manufacturing footprint from 90,000 sq. ft. to approximately 200,000 sq. ft., with the first phase expected to double cGMP capacity.
-
What strategic partnerships has Tris Pharma formed recently?
One significant partnership is with Neuraxpharm Group, which gives Neuraxpharm exclusive rights to commercialize Tris Pharma's ADHD medications in Europe.
-
How does Tris Pharma support innovation in product development?
Tris Pharma supports innovation through ongoing R&D efforts, a robust product pipeline, and the establishment of a Scientific Advisory Board to guide clinical research and product development.
-
What are the key competitive advantages of Tris Pharma?
Key competitive advantages include innovative ADHD formulations, a strong generics portfolio, expanding manufacturing capabilities, strategic partnerships for global reach, and a commitment to ongoing R&D and innovation.
Sources cited:
- https://www.trispharma.com/tris-pharma-expands-manufacturing-capacity-and-strengthens-supply-chain-to-support-new-and-pending-approvals/
- https://staging.trispharma.com/our-focus/generics/
- https://www.biospace.com/tris-pharma-expands-leadership-team-as-company-broadens-commercial-portfolio-and-progresses-clinical-pipeline
- https://leadiq.com/c/tris-pharma/5a1d8a5e240000240063a46c
- https://www.trispharma.com/tris-pharma-and-neuraxpharm-group-announce-partnership-to-commercialize-tris-quillivant-xr-and-quillichew-er-in-europe/
- https://www.businesswire.com/news/home/20230502005475/en/Tris-Pharma-Expands-Leadership-Team-as-Company-Broadens-Commercial-Portfolio-and-Progresses-Clinical-Pipeline
- https://www.trispharma.com/tris-pharma-launches-its-scientific-advisory-board/
- https://umbrex.com/resources/industry-analyses/how-to-analyze-a-pharmaceutical-company/generics-portfolio-strength-and-market-share/