Last updated: July 27, 2025
Introduction
Capecitabine, branded commercially as XELODA, is an oral chemotherapeutic agent primarily used in treating various cancers, including colorectal, breast, and gastric cancers. As a prodrug of 5-fluorouracil (5-FU), capecitabine offers targeted delivery and improved patient compliance, factors that have significantly influenced its market positioning. This report explores the evolving market dynamics and financial trajectory of capecitabine, considering factors such as competitive landscape, regulatory environments, pipeline developments, and healthcare trends that shape its commercial outlook.
Market Overview
The global pharmaceutical market for capecitabine is characterized by a mature yet dynamic landscape. The increasing prevalence of targeted cancer therapies and rising cancer incidence worldwide propel the demand for capecitabine. According to the Global Oncology Drugs Market report, the market was valued at approximately USD 16.4 billion in 2021 and is projected to grow at a CAGR of 7.2% through 2028[1].
Key market drivers include:
- Growing prevalence of colorectal and breast cancers: The World Health Organization reports over 2 million new cases of colorectal cancer annually, emphasizing the ongoing need for efficacious chemotherapeutics[2].
- Shift towards oral chemotherapy: Capecitabine’s oral administration offers convenience, enhancing patient adherence and quality of life, thereby boosting its adoption.
- Advancements in combination therapies: Integration with targeted agents and immunotherapies enhances efficacy, extending its clinical utility.
However, challenges like side effects (hand-foot syndrome, diarrhea), patent expiry, and competition from other chemotherapeutic agents limit market growth trajectory, emphasizing the importance of innovation and strategic positioning.
Competitive Landscape
Major pharmaceutical companies dominate the capecitabine market:
- Roche/Genentech: Originator of XELODA, with significant market share owing to established brand recognition.
- Sun Pharmaceutical Industries: Generic version manufacturer, capturing substantial market share due to cost advantages.
- Mylan/NOVARTIS: Other key players producing authorized generics.
The expiration of key patents in the early 2010s triggered a surge in generics, intensifying price competition and pressure on revenue streams for originators. Nonetheless, branded formulations benefit from clinical brand loyalty and perceived efficacy.
Emerging biosimilars and novel oral chemotherapeutic agents threaten incumbent dominance. The competitive landscape's evolution underscores the necessity for strategic alliances, lifecycle management, and clinical differentiation.
Regulatory and Patent Landscape
Patent expiries significantly influence the financial outlook of capecitabine. Roche’s original patents for XELODA expired around 2014, leading to a proliferation of generic products. Regulatory agencies such as the FDA and EMA continue to evaluate these generics, ensuring quality and bioequivalence.
Additionally, regulatory approval for new indications or formulations can bolster revenue streams. Recent filings exploring combination regimens and expanded indications may reignite growth potential.
Pipeline Developments and Innovation
While capecitabine itself is a mature product, ongoing clinical trials and formulational innovations impact its market trajectory:
- Combination therapies: Trials combining capecitabine with targeted agents (e.g., trastuzumab, bevacizumab) bolster its role in personalized medicine.
- Novel formulations: Liposomal versions and controlled-release preparations aim to enhance efficacy and reduce adverse effects.
- Biomarker-driven approaches: Precision oncology targeting specific genetic profiles can improve outcomes, potentially expanding capecitabine's use.
Investments in such innovations provide opportunities to mitigate the impact of generic competition and sustain revenues.
Market Penetration and Geographical Trends
Emerging markets are pivotal in future growth:
- Asia-Pacific: Countries like China and India report high cancer burdens and expanding healthcare infrastructure, leading to increased adoption of affordable generics.
- Latin America and Africa: Growing healthcare access and cancer awareness underpin market potential, although price sensitivity remains a barrier.
In contrast, mature markets emphasize clinical differentiation, combination therapies, and biosimilars to maintain market share.
Financial Trajectory Analysis
Expenditure on capecitabine reflects its transition from innovative drug to generic staple:
- Revenue Trends: Initial peak during exclusivity, followed by a decline post-patent expiry. Nonetheless, global sales remain robust due to volume-driven markets and expanding indications.
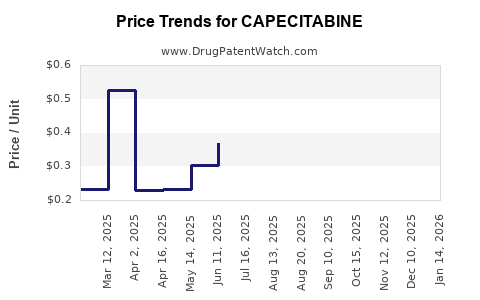
- Pricing Dynamics: Generic entry led to precipitous price reductions—averaging 30-50%—but strategic branding and new regimens have stabilized revenue streams.
- Profit Margins: Historically high for branded drugs; however, margins have compressed amid competitive pricing and increased manufacturing costs.
Projected revenue for leading manufacturers suggests stabilization with potential growth in emerging markets and pipeline-driven indications. A compounded annual decline of approximately 2-3% is expected in developed markets, while growth in emerging regions could offset declines.
Healthcare Economics and Policy Impact
Healthcare policies substantially influence capecitabine's market:
- Cost-effectiveness evaluations: Articles underscore capecitabine’s favorable profile versus infusion therapies, supporting reimbursement and formulary inclusion.
- Government procurement and pricing controls: Especially prevalent in emerging markets, influencing sales volume and revenue.
- Patent laws and exclusivity periods: Variability across jurisdictions impacts timing of generic entry and associated revenue.
Future Outlook
Capecitabine’s market is poised for moderate growth driven by:
- Expanding indications in gastric and esophageal cancers.
- Integration into combination regimens with immunotherapies.
- Ongoing pharmaceutical innovations.
However, generic competition, biosimilar threats, and pricing pressures necessitate strategic marketing, pipeline expansion, and cost optimization to preserve financial momentum.
Key Takeaways
- The global capecitabine market transitioned from rapid growth to maturity, with revenue stabilization post-patent expiry.
- Generic competition has driven prices down; however, strategic use in combination therapies and niche indications sustain revenues.
- Emerging markets offer growth opportunities owing to rising cancer incidences and healthcare infrastructure development.
- Innovation in formulations and identified biomarkers can extend capecitabine’s clinical utility, supporting future revenue streams.
- Regulatory and healthcare policy environments profoundly influence market access, pricing, and profitability.
FAQs
1. How has patent expiry impacted capecitabine’s market?
Patent expiry around 2014 led to the proliferation of generics, significantly reducing prices and market share for original branded formulations. However, volume-based sales and new indications have helped recover some revenue.
2. What are the primary drivers of demand for capecitabine?
Rising cancer incidences globally, favorable oral administration, and its integration into combination therapies primarily drive demand.
3. Which regions present the most growth opportunities for capecitabine?
Emerging markets in Asia-Pacific, Latin America, and Africa offer significant growth due to increasing cancer prevalence and expanding healthcare access.
4. How do combination therapies influence capecitabine’s market?
Synergistic regimens with targeted therapies improve treatment efficacy, extend indications, and support market demand despite generic competition.
5. What is the future outlook for capecitabine’s financial trajectory?
Moderate growth is anticipated, driven by pipeline innovations and expanding indications, balanced against competitive pressures and pricing dynamics.
References
[1] Global Oncology Drugs Market Report, 2022.
[2] WHO Cancer Statistics, 2022.