Introduction
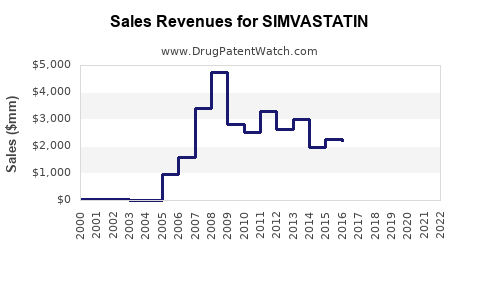
Simvastatin, a widely used statin, has been a cornerstone in the management of hypercholesterolemia and cardiovascular diseases for decades. The market dynamics and financial trajectory of simvastatin are influenced by several key factors, including generic competition, regional demand, and healthcare infrastructure.
Global Market Size and Growth
The global simvastatin drug market is projected to expand at a compound annual growth rate (CAGR) of 5.20% from 2023 to 2030. This growth is driven by increasing demand for cholesterol management solutions, particularly in regions with high prevalence of lifestyle-related diseases[1].
Regional Market Analysis
North America
North America emerges as the largest region for simvastatin, commanding a substantial 40% market share. The United States dominates this region, driven by the high prevalence of hypercholesterolemia and cardiovascular disorders. Robust healthcare infrastructure and advanced diagnostic capabilities further contribute to the widespread usage of simvastatin in this region[1].
Asia Pacific
The Asia Pacific region stands out as the fastest-growing market for simvastatin, representing 24% of the global market share and growing at a CAGR of 6.6%. This growth is propelled by rising awareness about heart health, lifestyle changes leading to increased cardiovascular diseases, and improving healthcare infrastructure across the region[1].
Europe, South America, and Middle East & Africa
Europe, South America, and the Middle East & Africa also show significant growth, with CAGRs of 4.5%, 5%, and 5.1%, respectively. These regions are driven by similar factors, including increasing awareness of heart health and expanding healthcare infrastructure[1].
Impact of Generic Competition
The expiration of simvastatin's patent has led to the entry of numerous generic versions into the market. This has significantly reduced the cost of the drug, making it more affordable for consumers. However, it poses challenges for original manufacturers, leading to reduced profit margins and intensified pricing pressures.
Cost Savings
A study using data from the Medical Expenditure Panel Survey found that the introduction of generic statins, including simvastatin, resulted in significant cost savings. On average, individuals saved $925.60 annually, translating to $11.9 billion in savings for the US healthcare system[2].
Market Share and Revenue
The entry of generic simvastatin has led to a decline in brand-name statin purchases. For instance, the number of brand-name statin purchases decreased by 90.9% nationally and 27.4% individually after the end of market exclusivity. This shift has resulted in reduced revenue for original manufacturers, while generic versions have seen a surge in prescriptions and spending[2].
Therapeutic and Economic Impact
Cost-Effectiveness
Studies have shown that simvastatin is cost-effective in reducing vascular events among individuals with vascular disease or diabetes. A study involving 20,536 adults found that simvastatin allocation was associated with a 22% reduction in hospitalization costs for vascular events, highlighting its economic benefits[5].
Healthcare Expenditures
The introduction of generic simvastatin has significantly impacted healthcare expenditures. For example, private insurance spending on Lipitor (a brand-name statin) dropped from $4.72 billion to $0.01 billion after the end of market exclusivity, while spending on generic atorvastatin and rosuvastatin increased substantially[2].
Market Segments and End Users
Drug Class
Simvastatin is part of the broader statin market, which includes other drugs like atorvastatin, rosuvastatin, and lovastatin. The statin market is segmented by drug class, with simvastatin being one of the key players[4].
Therapeutic Treatment
Simvastatin is primarily used for treating cardiovascular disorders, but it also has applications in managing obesity and inflammatory disorders. The demand for simvastatin is driven by its efficacy in these therapeutic areas[4].
End Users
The primary end users of simvastatin include hospitals and clinics. The drug is widely prescribed in these settings due to its effectiveness in managing cholesterol levels and reducing the risk of cardiovascular events[4].
Future Outlook
Market Size and CAGR
The global statin market, which includes simvastatin, is expected to grow from $15.39 billion in 2023 to $18.93 billion by 2030, with a CAGR of 3% during the forecast period. This growth is driven by increasing prevalence of cholesterol-related diseases and the expanding use of statins in various therapeutic areas[4].
Regional Growth
The Asia Pacific region is expected to continue its rapid growth, driven by increasing awareness of heart health and improving healthcare infrastructure. North America will remain a significant market, while other regions will also contribute to the overall growth of the simvastatin market[1].
Key Takeaways
- The global simvastatin market is growing at a CAGR of 5.20% from 2023 to 2030.
- North America and Asia Pacific are the largest and fastest-growing regions, respectively.
- Generic competition has significantly reduced the cost of simvastatin, leading to cost savings for consumers and healthcare systems.
- Simvastatin is cost-effective in reducing vascular events and is widely used in managing cardiovascular disorders.
- The market is segmented by drug class, therapeutic treatment, and end users, with hospitals and clinics being primary end users.
FAQs
What is the projected CAGR for the global simvastatin market from 2023 to 2030?
The global simvastatin market is projected to expand at a CAGR of 5.20% from 2023 to 2030[1].
How has generic competition affected the simvastatin market?
Generic competition has led to significant cost savings, reduced brand-name statin purchases, and increased prescriptions for generic versions, posing challenges for original manufacturers in terms of revenue and market share[2].
Which regions are the largest and fastest-growing markets for simvastatin?
North America is the largest region, commanding a 40% market share, while the Asia Pacific region is the fastest-growing, with a CAGR of 6.6%[1].
What are the primary therapeutic uses of simvastatin?
Simvastatin is primarily used for treating cardiovascular disorders but also has applications in managing obesity and inflammatory disorders[4].
How does simvastatin impact healthcare expenditures?
Simvastatin, especially in its generic form, has significantly reduced healthcare expenditures by decreasing the cost of statin therapy and reducing hospitalization costs for vascular events[2][5].
Sources
- Cognitive Market Research, "Simvastatin Drug Market Report 2024 (Global Edition)".
- JAMA Network Open, "Use and Expenditures for Brand-name Statins After Introduction of Generic Competition".
- Market Research Intellect, "Ezetimibe and Simvastatin Sales Market Size, Scope And Forecast".
- Maximize Market Research, "Statin Market: Global Industry Analysis and Forecast (2024-2030)".
- PubMed, "Cost-effectiveness of simvastatin in people at different levels of vascular disease risk".