Introduction to Carbamazepine
Carbamazepine is a widely used antiepileptic drug, also employed in the treatment of bipolar disorder, trigeminal neuralgia, and neuropathic pain. Understanding its market dynamics and financial trajectory is crucial for stakeholders in the pharmaceutical industry.
Current Market Size and Projections
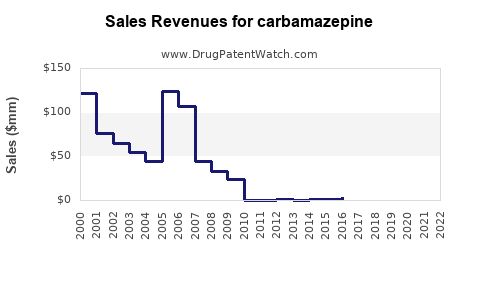
As of 2023, the global carbamazepine market was valued at USD 713.4 million. It is projected to grow to USD 958.77 million by 2031, with a compound annual growth rate (CAGR) of 3.3% from 2024 to 2031[3].
Market Segmentation
The carbamazepine market is segmented based on type (tablets, capsules, liquid) and application (epilepsy, bipolar disorder, trigeminal neuralgia, neuropathic pain, alcohol withdrawal). Geographical regions such as North America, Europe, Asia-Pacific, South America, and the Middle East and Africa are also key segments[3][4].
Drivers of Market Growth
Several factors are driving the growth of the carbamazepine market:
- Increasing Incidence of Neurological Conditions: The rising incidence of epilepsy and bipolar disorder globally is a significant driver. Improved diagnostic methods and growing awareness contribute to higher demand for carbamazepine[3].
- Advancements in Medication Formulations: Improvements in drug formulations, such as prolonged-release versions, enhance patient compliance and treatment efficacy, further propelling market growth[3].
- Generic Availability: The availability of generic carbamazepine provides affordable alternatives, expanding the market reach[3].
- Research into Novel Therapeutic Uses: Continuous research into new therapeutic applications and combinations of carbamazepine enhances its utility and market potential[3].
Challenges in Q4 2024
The carbamazepine API market is expected to face significant challenges in Q4 2024:
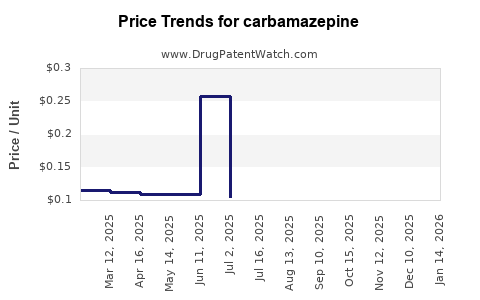
- Demand Contraction: Pharmaceutical manufacturers are likely to exhibit caution in inventory management due to saturated market conditions, leading to a contraction in demand and pricing pressures[1].
- Economic Uncertainties: Global economic uncertainties, including persistent inflation and constrained healthcare budgets, will compel buyers to adopt conservative purchasing strategies. This will result in reduced volume-based procurement and a shift towards just-in-time inventory models to mitigate financial risks[1].
- Currency Volatility: Depreciating local currencies in key importing regions will erode purchasing power, forcing manufacturers to recalibrate their procurement strategies and contributing to reduced demand and more conservative inventory management approaches[1].
Impact of Economic Factors
Economic factors play a critical role in shaping the carbamazepine market:
- Pricing Pressures: The competitive landscape will allow buyers to negotiate more favorable terms, further pressuring API pricing and market dynamics. Volatile pricing mechanisms will trigger a cautious "wait-and-watch" approach among downstream buyers, contributing to market contraction[1].
- Inventory Management: The strategic shift towards more targeted neurological and psychiatric treatments, coupled with inventory overhang, will dampen carbamazepine API procurement. This will result in a self-reinforcing cycle of reduced demand and price pressures[1].
Regional Variations
Regional variations will significantly influence the market:
- Geographical Segmentation: The market is segmented into regions such as North America, Europe, Asia-Pacific, South America, and the Middle East and Africa. Each region has its own set of economic, healthcare, and regulatory dynamics that will impact the demand and pricing of carbamazepine[3][4].
- Country-Specific Trends: Country-level trends, including GDP, healthcare budgets, and regulatory environments, will also play a crucial role in shaping the market. For instance, regions with stronger healthcare systems and higher GDP are likely to have more stable demand for carbamazepine[4].
Competitive Landscape
The competitive landscape of the carbamazepine market is characterized by:
- Key Players: Companies like Novartis and Teva are focusing on strategy building to strengthen their product portfolios and expand their business globally. These companies are investing in research and development to enhance the therapeutic uses of carbamazepine[4].
- Market Strategies: Stakeholders are advised to maintain agile strategies, closely monitor regional variations, and remain adaptable to the rapidly transforming pharmaceutical API landscape. This includes leveraging competitive pricing mechanisms and negotiating favorable terms[1].
Economic Appraisal and Cost-Effectiveness
An economic appraisal of carbamazepine highlights its cost-effectiveness:
- Cost-Minimization Model: Studies have shown that carbamazepine is cost-effective compared to other antiepileptic drugs like lamotrigine, phenytoin, and valproate. The direct medical costs of 2-year therapy for carbamazepine are relatively lower, making it a preferred option for many patients[2].
Future Prospects
Despite the challenges in Q4 2024, the long-term prospects for the carbamazepine market remain positive:
- Growing Demand: The increasing incidence of neurological conditions and advancements in medication formulations will continue to drive demand for carbamazepine.
- Innovative Therapeutic Uses: Continuous research into novel therapeutic applications will enhance the market potential of carbamazepine.
- Generic and Branded Options: The availability of both generic and branded carbamazepine will provide a range of options for patients, contributing to market growth[3].
Key Takeaways
- The carbamazepine market is expected to grow from USD 713.4 million in 2023 to USD 958.77 million by 2031.
- Economic uncertainties and demand contraction are expected to impact the market in Q4 2024.
- Regional variations and competitive strategies will play a crucial role in shaping the market.
- Carbamazepine remains a cost-effective option for treating neurological conditions.
- Continuous research and advancements in medication formulations will drive long-term market growth.
Frequently Asked Questions (FAQs)
Q: What is the current market size of the carbamazepine market?
A: The global carbamazepine market was valued at USD 713.4 million in 2023[3].
Q: What is the projected growth rate of the carbamazepine market from 2024 to 2031?
A: The market is expected to grow at a CAGR of 3.3% from 2024 to 2031[3].
Q: What are the primary drivers of the carbamazepine market growth?
A: The primary drivers include the increasing incidence of neurological conditions, advancements in medication formulations, and the availability of generic carbamazepine[3].
Q: How will economic uncertainties impact the carbamazepine API market in Q4 2024?
A: Economic uncertainties will lead to demand contraction, pricing pressures, and more conservative inventory management approaches, resulting in reduced demand and price pressures[1].
Q: Which regions are expected to be most lucrative for the carbamazepine market in the near future?
A: Regions with strong healthcare systems and higher GDP, such as North America and Europe, are likely to be more lucrative due to stable demand and better economic conditions[4].
Cited Sources
- ChemAnalyst: Q4-2024 Carbamazepine API Market Forecast - ChemAnalyst.
- PubMed: An economic appraisal of carbamazepine, lamotrigine, phenytoin ...
- Market Research Intellect: Carbamazepine Market Size and Projections.
- Cognitive Market Research: Carbamazepine Market Report 2024 (Global Edition).