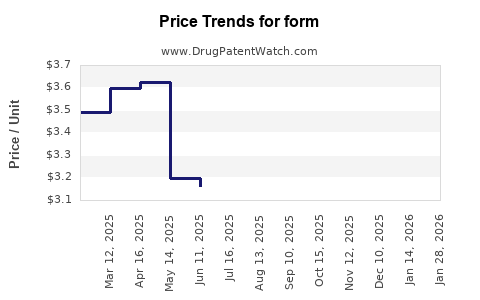
Form - Generic Drug Details
✉ Email this page to a colleague
US Patents and Regulatory Information for form
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Usa Inc | FORMOTEROL FUMARATE | formoterol fumarate | SOLUTION;INHALATION | 091141-001 | Jun 22, 2021 | AN | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Astrazeneca | BEVESPI AEROSPHERE | formoterol fumarate; glycopyrrolate | AEROSOL, METERED;INHALATION | 208294-001 | Apr 25, 2016 | RX | Yes | Yes | 8,324,266 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Astrazeneca | BEVESPI AEROSPHERE | formoterol fumarate; glycopyrrolate | AEROSOL, METERED;INHALATION | 208294-001 | Apr 25, 2016 | RX | Yes | Yes | 9,463,161 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |