ALUNBRIG Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Alunbrig, and what generic alternatives are available?
Alunbrig is a drug marketed by Takeda Pharms Usa and is included in one NDA. There are four patents protecting this drug.
This drug has ninety patent family members in thirty-nine countries.
The generic ingredient in ALUNBRIG is brigatinib. One supplier is listed for this compound. Additional details are available on the brigatinib profile page.
DrugPatentWatch® Generic Entry Outlook for Alunbrig
Alunbrig was eligible for patent challenges on April 28, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 10, 2035. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ALUNBRIG?
- What are the global sales for ALUNBRIG?
- What is Average Wholesale Price for ALUNBRIG?
Summary for ALUNBRIG
| International Patents: | 90 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 61 |
| Clinical Trials: | 10 |
| Patent Applications: | 419 |
| Drug Prices: | Drug price information for ALUNBRIG |
| What excipients (inactive ingredients) are in ALUNBRIG? | ALUNBRIG excipients list |
| DailyMed Link: | ALUNBRIG at DailyMed |
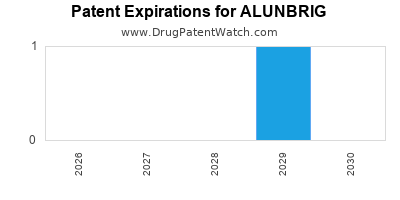

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ALUNBRIG
Generic Entry Date for ALUNBRIG*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ALUNBRIG
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Intergroupe Francophone de Cancerologie Thoracique | Phase 2 |
| Princess Maxima Center for Pediatric Oncology | Phase 1/Phase 2 |
| Takeda | Phase 1/Phase 2 |
Pharmacology for ALUNBRIG
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 3A Inducers Tyrosine Kinase Inhibitors |
US Patents and Regulatory Information for ALUNBRIG
ALUNBRIG is protected by four US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ALUNBRIG is ⤷ Try for Free.
This potential generic entry date is based on patent 10,385,078.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-001 | Apr 28, 2017 | RX | Yes | No | 9,611,283 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-001 | Apr 28, 2017 | RX | Yes | No | 9,012,462 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-002 | Apr 28, 2017 | RX | Yes | No | 9,273,077 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-002 | Apr 28, 2017 | RX | Yes | No | 10,385,078 | ⤷ Try for Free | Y | Y | ⤷ Try for Free | ||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-003 | Oct 2, 2017 | RX | Yes | Yes | 9,273,077 | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ALUNBRIG
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharma A/S | Alunbrig | brigatinib | EMEA/H/C/004248 Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase (ALK)‑positive advanced non‑small cell lung cancer (NSCLC) previously not treated with an ALK inhibitor.Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase ALKpositive advanced NSCLC previously treated with crizotinib. |
Authorised | no | no | no | 2018-11-22 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ALUNBRIG
When does loss-of-exclusivity occur for ALUNBRIG?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 15335950
Estimated Expiration: ⤷ Try for Free
Canada
Patent: 65169
Estimated Expiration: ⤷ Try for Free
Chile
Patent: 17000979
Estimated Expiration: ⤷ Try for Free
China
Patent: 7108559
Estimated Expiration: ⤷ Try for Free
Patent: 1825717
Estimated Expiration: ⤷ Try for Free
Patent: 1888368
Estimated Expiration: ⤷ Try for Free
Colombia
Patent: 17004714
Estimated Expiration: ⤷ Try for Free
Costa Rica
Patent: 170146
Estimated Expiration: ⤷ Try for Free
Croatia
Patent: 0201343
Estimated Expiration: ⤷ Try for Free
Cyprus
Patent: 23295
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 09647
Estimated Expiration: ⤷ Try for Free
Dominican Republic
Patent: 017000101
Estimated Expiration: ⤷ Try for Free
Ecuador
Patent: 17030878
Estimated Expiration: ⤷ Try for Free
Eurasian Patent Organization
Patent: 5145
Estimated Expiration: ⤷ Try for Free
Patent: 1790892
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 09647
Estimated Expiration: ⤷ Try for Free
Patent: 60618
Estimated Expiration: ⤷ Try for Free
Hungary
Patent: 51693
Estimated Expiration: ⤷ Try for Free
Israel
Patent: 1818
Estimated Expiration: ⤷ Try for Free
Patent: 9910
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 33072
Estimated Expiration: ⤷ Try for Free
Patent: 17535538
Estimated Expiration: ⤷ Try for Free
Patent: 20063276
Estimated Expiration: ⤷ Try for Free
Lithuania
Patent: 09647
Estimated Expiration: ⤷ Try for Free
Malaysia
Patent: 2216
Estimated Expiration: ⤷ Try for Free
Mexico
Patent: 17005120
Estimated Expiration: ⤷ Try for Free
New Zealand
Patent: 0940
Estimated Expiration: ⤷ Try for Free
Peru
Patent: 171344
Estimated Expiration: ⤷ Try for Free
Philippines
Patent: 017500732
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 09647
Estimated Expiration: ⤷ Try for Free
Serbia
Patent: 737
Estimated Expiration: ⤷ Try for Free
Singapore
Patent: 201702980Q
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 09647
Estimated Expiration: ⤷ Try for Free
South Africa
Patent: 1702737
Estimated Expiration: ⤷ Try for Free
South Korea
Patent: 2331856
Estimated Expiration: ⤷ Try for Free
Patent: 170072905
Estimated Expiration: ⤷ Try for Free
Patent: 210142781
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 13726
Estimated Expiration: ⤷ Try for Free
Tunisia
Patent: 17000157
Estimated Expiration: ⤷ Try for Free
Ukraine
Patent: 9794
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ALUNBRIG around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 102105150 | Phosphorous derivatives as kinase inhibitors | ⤷ Try for Free |
| European Patent Office | 3209647 | ⤷ Try for Free | |
| Japan | 2022116057 | キナーゼ阻害剤としてのリン誘導体 | ⤷ Try for Free |
| Netherlands | 300990 | ⤷ Try for Free | |
| South Korea | 101781605 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ALUNBRIG
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2300013 | 122019000046 | Germany | ⤷ Try for Free | PRODUCT NAME: BRIGATINIB, ODER EIN PHARMAZEUTISCH UNBEDENKLICHES SALZ DAVON; REGISTRATION NO/DATE: EU/1/18/1264 20181122 |
| 2300013 | 2019C/525 | Belgium | ⤷ Try for Free | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), ASSIGNMENT |
| 2300013 | C20190026 00287 | Estonia | ⤷ Try for Free | PRODUCT NAME: BRIGATINIIB;REG NO/DATE: EU/1/18/1264 26.11.2018 |
| 2300013 | PA2019510,C2300013 | Lithuania | ⤷ Try for Free | PRODUCT NAME: BRIGATINIBAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/18/1264 20181122 |
| 2300013 | SPC/GB19/036 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: BRIGATINIB, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: UK EU/1/18/1264 (NI) 20181126; UK PLGB 15475/0037 20181126; UK PLGB 15475/0038 20181126; UK PLGB 15475/0039 20181126; UK PLGB 15475/0075 20181126 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ALUNBRIG
More… ↓


