ALUNBRIG Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Alunbrig, and when can generic versions of Alunbrig launch?
Alunbrig is a drug marketed by Takeda Pharms Usa and is included in one NDA. There are four patents protecting this drug.
This drug has ninety patent family members in thirty-nine countries.
The generic ingredient in ALUNBRIG is brigatinib. One supplier is listed for this compound. Additional details are available on the brigatinib profile page.
DrugPatentWatch® Generic Entry Outlook for Alunbrig
Alunbrig was eligible for patent challenges on April 28, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 10, 2035. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ALUNBRIG?
- What are the global sales for ALUNBRIG?
- What is Average Wholesale Price for ALUNBRIG?
Summary for ALUNBRIG
| International Patents: | 90 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 61 |
| Clinical Trials: | 10 |
| Patent Applications: | 416 |
| Drug Prices: | Drug price information for ALUNBRIG |
| What excipients (inactive ingredients) are in ALUNBRIG? | ALUNBRIG excipients list |
| DailyMed Link: | ALUNBRIG at DailyMed |
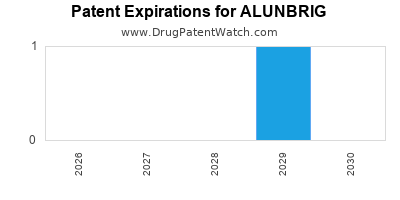

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ALUNBRIG
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ALUNBRIG
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Intergroupe Francophone de Cancerologie Thoracique | Phase 2 |
| Takeda | Phase 1/Phase 2 |
| Princess Maxima Center for Pediatric Oncology | Phase 1/Phase 2 |
Pharmacology for ALUNBRIG
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 3A Inducers Tyrosine Kinase Inhibitors |
US Patents and Regulatory Information for ALUNBRIG
ALUNBRIG is protected by four US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ALUNBRIG is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting ALUNBRIG
Crystalline forms of 5-chloro-N4-[-2-(dimethylphosphoryl)phenyl]-N2-{2-methoxy-4-[4-(4-methylp- iperazin-1-YL) piperidin-1-YL]phenyl}pyrimidine-2,4-diamine
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ANAPLASTIC LYMPHOMA KINASE (ALK)-POSITIVE METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC)
Phosphorous derivatives as kinase inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Phosphorus derivatives as kinase inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ANAPLASTIC LYMPHOMA KINASE (ALK)-POSITIVE METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC)
Methods for inhibiting cell proliferation in ALK-driven cancers
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ANAPLASTIC LYMPHOMA KINASE (ALK)-POSITIVE METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC)
FDA Regulatory Exclusivity protecting ALUNBRIG
TREATMENT OF PATIENTS WITH ANAPLASTIC LYMPHOMA KINASE (ALK)-POSITIVE METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC) WHO HAVE PROGRESSED ON OR ARE INTOLERANT TO CRIZOTINIB
Exclusivity Expiration: ⤷ Sign Up
FOR THE TREATMENT OF ADULT PATIENTS WITH ANAPLASTIC LYMPHOMA KINASE (ALK)-POSITIVE METASTATIC NON-SMALL CELL LUNG CANCER (NSCLC) AS DETECTED BY AN FDA-APPROVED TEST, NOT INCLUDING PATIENTS WHO HAVE PROGRESSED ON OR ARE INTOLERANT TO CRIZOTINIB
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-001 | Apr 28, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-003 | Oct 2, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-002 | Apr 28, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Takeda Pharms Usa | ALUNBRIG | brigatinib | TABLET;ORAL | 208772-001 | Apr 28, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ALUNBRIG
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharma A/S | Alunbrig | brigatinib | EMEA/H/C/004248 Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase (ALK)‑positive advanced non‑small cell lung cancer (NSCLC) previously not treated with an ALK inhibitor.Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase ALKpositive advanced NSCLC previously treated with crizotinib. |
Authorised | no | no | no | 2018-11-22 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ALUNBRIG
When does loss-of-exclusivity occur for ALUNBRIG?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 15335950
Patent: Crystalline forms of 5-chloro-N4-[-2-(dimethylphosphoryl) phenyl]-N2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl) piperidin-1-yl] pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 65169
Patent: FORMES CRISTALLINES DE 5-CHLORO-N4-[2-(DIMETHYLPHOSPHORYL)PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL)PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N4-[2-(DIMETHYLPHOSPHORYL)PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL)PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 17000979
Patent: Formas cristalinas de 5-cloro-n4-[2-(dimetilfosforil)fenil]-n2-{2-metoxi-4-[4-(4-metilpiperazin-1-il)piperidin-1-il]pirimidina-2,4-diamina
Estimated Expiration: ⤷ Sign Up
China
Patent: 7108559
Patent: 5‑氯‑N4‑[2‑(二甲基磷酰基)苯基]‑N2‑{2‑甲氧基‑4‑[4‑(4‑甲基哌嗪‑1‑基)哌啶‑1‑基]嘧啶‑2,4‑二胺的晶形 (Crystalline forms of 5-chloro-n4-[-2-(dimethylphosphoryl) phenyl]-n2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl) piperidin-1-yl] pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Sign Up
Patent: 1825717
Patent: 5-氯-N4-[2-(二甲基磷酰基)苯基]-N2-{2-甲氧基-4-[4-(4-甲基哌嗪-1-基)哌啶-1-基]苯基}嘧啶-2,4-二胺的晶形 (Crystalline forms of 5-chloro-N4-[-2-(dimethylphosphoryl) phenyl]-N2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl) piperidin-1-yl] pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Sign Up
Patent: 1888368
Patent: 5-氯-N4-[2-(二甲基磷酰基)苯基]-N2-{2-甲氧基-4-[4-(4-甲基哌嗪-1-基)哌啶-1-基]苯基}嘧啶-2,4-二胺的晶形 (Crystalline forms of 5-chloro-N4-[-2-(dimethylphosphoryl) phenyl]-N2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl) piperidin-1-yl] pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Sign Up
Colombia
Patent: 17004714
Patent: Formas cristalinas de 5-cloro-n4-[2-(dimetilfosforil)fenil]-n2-{2-metoxi-4-[4-(4-metilpiperazin-1-il)piperidin-1-il]pirimidina-2,4-diamina
Estimated Expiration: ⤷ Sign Up
Costa Rica
Patent: 170146
Patent: FORMAS CRISTALINAS DE 5-CLORO-N4-[-2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-YL)PIPERIDIN-1-IL]PIRIMIDINA-2,4-DIAMINA
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0201343
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 23295
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 09647
Estimated Expiration: ⤷ Sign Up
Dominican Republic
Patent: 017000101
Patent: FORMAS CRISTALINAS DE 5-CLORO-N4-[2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL]PIRIMIDINA-2,4-DIAMINA
Estimated Expiration: ⤷ Sign Up
Ecuador
Patent: 17030878
Patent: FORMAS CRISTALINAS DE 5-CLORO-N4-[2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL]PIRIMIDINA-2,4-DIAMINA
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 5145
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N4-[2-(ДИМЕТИЛФОСФОРИЛ)ФЕНИЛ]-N2-{2-МЕТОКСИ-4-[4-(4-МЕТИЛПИПЕРАЗИН-1-ИЛ)ПИПЕРИДИН-1-ИЛ]ФЕНИЛ}ПИРИМИДИН-2,4-ДИАМИНА (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL)PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL)PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Patent: 1790892
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N4-[2-(ДИМЕТИЛФОСФОРИЛ)ФЕНИЛ]-N2-{2-МЕТОКСИ-4-[4-(4-МЕТИЛПИПЕРАЗИН-1-ИЛ)ПИПЕРИДИН-1-ИЛ]}ПИРИМИДИН-2,4-ДИАМИНА
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 09647
Patent: FORMES CRISTALLINES DE 5-CHLORO-N4- [-2- (DIMÉTHYLPHOSPHORYL) PHÉNYL]-N2- {2-MÉTHOXY-4- [4- (4-MÉTHYLPIPÉRAZIN-1-YL) PIPÉRIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Patent: 60618
Patent: FORMES CRISTALLINES DE 5-CHLORO-N4-[-2-(DIMÉTHYLPHOSPHORYL) PHÉNYL]-N2-{2-MÉTHOXY-4-[4-(4-MÉTHYLPIPERAZINE-1-YL) PIPÉRIDINE-1-YL]PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 51693
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 1818
Patent: צורות גבישיות של 5- כלורו-אן 4- [2-(דימתילפוספוריל)פניל]-אן2-{2-מתוקסי-4-[4-(4-מתילפיפראזין-1-איל) פיפרידין-1-איל]פירימידין-2-4-דיאמין (Crystalline forms of 5-chloro-n4-[2-(dimethylphosphoryl)phenyl]-n2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Sign Up
Patent: 9910
Patent: צורות גבישיות של 5- כלורו-אן 4- [2-(דימתילפוספוריל)פניל]-אן2-{2-מתוקסי-4-[4-(4-מתילפיפראזין-1-איל) פיפרידין-1-איל]פירימידין-2-4-דיאמין (Crystalline forms of 5-chloro-n4-[2-(dimethylphosphoryl)phenyl]-n2- {2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 33072
Estimated Expiration: ⤷ Sign Up
Patent: 17535538
Patent: 5−クロロ−N4−[2−(ジメチルホスホリル)フェニル]−N2−{2−メトキシ−4−[4−(4−メチルピペラジン−1−イル)ピペリジン−1−イル]ピリミジン−2,4−ジアミンの結晶形態
Estimated Expiration: ⤷ Sign Up
Patent: 20063276
Patent: 5−クロロ−N4−[2−(ジメチルホスホリル)フェニル]−N2−{2−メトキシ−4−[4−(4−メチルピペラジン−1−イル)ピペリジン−1−イル]フェニル}ピリミジン−2,4−ジアミンの結晶形態 (CRYSTAL FORM OF 5-CHLORO-N4-[2-(DIMETHYL PHOSPHORYL)PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZINE-1-YL)PIPERIDINE-1-YL]PHENYL}PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Lithuania
Patent: 09647
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 2216
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 17005120
Patent: FORMAS CRISTALINAS DE 5-CLORO-N4-[-2-(DIMETILFOSFORIL) FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL] PIRIMIDIN-2,4-DIAMINA. (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE.)
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 0940
Patent: Crystalline forms of 5-chloro-n4-[-2-(dimethylphosphoryl) phenyl]-n2-{ 2-methoxy-4-[4-(4-methylpiperazin-1-yl) piperidin-1-yl] pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Sign Up
Peru
Patent: 171344
Patent: FORMAS CRISTALINAS DE 5-CLORO-N4-[2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL]PIRIMIDINA-2,4-DIAMINA
Estimated Expiration: ⤷ Sign Up
Philippines
Patent: 017500732
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 09647
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 737
Patent: KRISTALNI OBLICI 5-HLORO-N4-[-2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOKSI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL]PIRIMIDIN-2,4-DIAMINA (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL]PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 201702980Q
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 09647
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 1702737
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PHENYL}PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 2331856
Estimated Expiration: ⤷ Sign Up
Patent: 170072905
Patent: 결정 형태의 5-클로로-N4-[2-페닐]-N2-{2-메톡시-4-[4-피페리딘-1-일]피리미딘-2,4-디아민 (5--N4-[2-]-N2-2--4-[4-4--1--1-]-24-CRYSTALLINE FORMS OF 5-CHLORO-N4-[2-DIMETHYLPHOSPHORYLPHENYL]-N2-2-METHOXY-4-[4-4-METHYLPIPERAZIN-1-YLPIPERIDIN-1-YL]PYRIMIDINE-24-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Patent: 210142781
Patent: 결정 형태의 5-클로로-N4-[2-페닐]-N2-{2-메톡시-4-[4-피페리딘-1-일]피리미딘-2,4-디아민 (5--N4-[2-]-N2-2--4-[4-4--1--1-]-24-CRYSTALLINE FORMS OF 5-CHLORO-N4-[2-DIMETHYLPHOSPHORYLPHENYL]-N2-2-METHOXY-4-[4-4-METHYLPIPERAZIN-1-YLPIPERIDIN-1-YL]PYRIMIDINE-24-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 13726
Estimated Expiration: ⤷ Sign Up
Tunisia
Patent: 17000157
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 9794
Patent: КРИСТАЛІЧНА ФОРМА 5-ХЛОР-N4-[2-(ДИМЕТИЛФОСФОРИЛ)ФЕНІЛ]-N2-{2-МЕТОКСИ-4-[4-(4-МЕТИЛПІПЕРАЗИН-1-ІЛ)ПІПЕРИДИН-1-ІЛ]}ПІРИМІДИН-2,4-ДІАМІНУ (CRYSTALLINE FORMS OF 5-CHLORO-N4-[-2-(DIMETHYLPHOSPHORYL) PHENYL]-N2-{2-METHOXY-4-[4-(4-METHYLPIPERAZIN-1-YL) PIPERIDIN-1-YL] PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ALUNBRIG around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Peru | 20171344 | FORMAS CRISTALINAS DE 5-CLORO-N4-[2-(DIMETILFOSFORIL)FENIL]-N2-{2-METOXI-4-[4-(4-METILPIPERAZIN-1-IL)PIPERIDIN-1-IL]PIRIMIDINA-2,4-DIAMINA | ⤷ Sign Up |
| Denmark | 2300013 | ⤷ Sign Up | |
| Japan | 2020125308 | キナーゼ阻害剤としてのリン誘導体 (PHOSPHOROUS DERIVATIVES AS KINASE INHIBITORS) | ⤷ Sign Up |
| Australia | 2009248923 | Phosphorous derivatives as kinase inhibitors | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ALUNBRIG
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2300013 | 2019/031 | Ireland | ⤷ Sign Up | PRODUCT NAME: BRIGATINIB, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; REGISTRATION NO/DATE: EU/1/18/1264 20181122 |
| 2300013 | 2019024 | Norway | ⤷ Sign Up | PRODUCT NAME: BRIGATINIB ELLER ET FARMASOEYTISK AKSEPTABELT SALT DERAV; REG. NO/DATE: EU/1/18/1264 20181218 |
| 2300013 | 31/2019 | Austria | ⤷ Sign Up | PRODUCT NAME: BRIGATINIB ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/18/1264 (MITTEILUNG) 20181126 |
| 2300013 | SPC/GB19/036 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: BRIGATINIB, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: UK EU/1/18/1264 (NI) 20181126; UK PLGB 15475/0037 20181126; UK PLGB 15475/0038 20181126; UK PLGB 15475/0039 20181126; UK PLGB 15475/0075 20181126 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


