ASMANEX HFA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Asmanex Hfa, and what generic alternatives are available?
Asmanex Hfa is a drug marketed by Organon Llc and is included in one NDA.
The generic ingredient in ASMANEX HFA is mometasone furoate. There are thirty drug master file entries for this compound. Twenty-seven suppliers are listed for this compound. Additional details are available on the mometasone furoate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Asmanex Hfa
A generic version of ASMANEX HFA was approved as mometasone furoate by PADAGIS US on March 18th, 2002.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ASMANEX HFA?
- What are the global sales for ASMANEX HFA?
- What is Average Wholesale Price for ASMANEX HFA?
Summary for ASMANEX HFA
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 79 |
| Clinical Trials: | 12 |
| Patent Applications: | 4,474 |
| Drug Prices: | Drug price information for ASMANEX HFA |
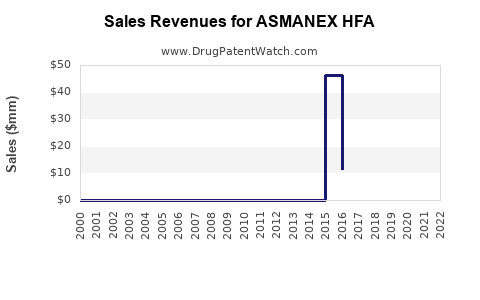
| Drug Sales Revenues: | Drug sales revenues for ASMANEX HFA |
| What excipients (inactive ingredients) are in ASMANEX HFA? | ASMANEX HFA excipients list |
| DailyMed Link: | ASMANEX HFA at DailyMed |
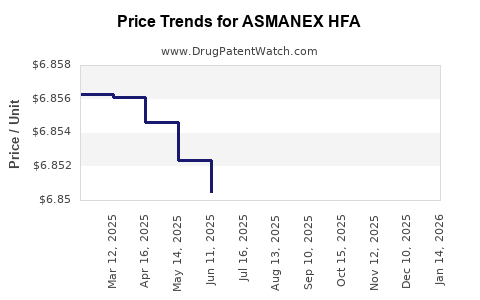
See drug prices for ASMANEX HFA
Recent Clinical Trials for ASMANEX HFA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Milton S. Hershey Medical Center | Phase 3 |
| National Heart, Lung, and Blood Institute (NHLBI) | Phase 3 |
| University of Dundee | Phase 4 |
Pharmacology for ASMANEX HFA
| Drug Class | Corticosteroid |
| Mechanism of Action | Corticosteroid Hormone Receptor Agonists |
US Patents and Regulatory Information for ASMANEX HFA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-001 | Apr 25, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-002 | Apr 25, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-003 | Aug 12, 2019 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ASMANEX HFA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-002 | Apr 25, 2014 | ⤷ Try for Free | ⤷ Try for Free |
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-001 | Apr 25, 2014 | ⤷ Try for Free | ⤷ Try for Free |
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-001 | Apr 25, 2014 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ASMANEX HFA
See the table below for patents covering ASMANEX HFA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 100358528 | ⤷ Try for Free | |
| European Patent Office | 1174139 | Utilisation du furoate de mometasone pour le traitement des affections pulmonaires et des voies respiratoires (Use of mometasone furoate for treating airway passage and lung diseases) | ⤷ Try for Free |
| Germany | 69526425 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ASMANEX HFA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3043773 | 132021000000191 | Italy | ⤷ Try for Free | PRODUCT NAME: MOMETASONE O UN SUO SALE E OLOPATADINA O UN SUO SALE(RYALTRIS); AUTHORISATION NUMBER(S) AND DATE(S): 048499014, 048499026, 048499038, 20210726;140638, 20210426 |
| 3043773 | 21C1057 | France | ⤷ Try for Free | PRODUCT NAME: MOMETASONE OU L'UN DE SES SELS AVEC OLOPATADINE OU L'UN DE SES SELS; NAT. REGISTRATION NO/DATE: NL52121 20211026; FIRST REGISTRATION: AT - 140638 20210426 |
| 0112669 | 96C0002 | Belgium | ⤷ Try for Free | PRODUCT NAME: MOMETASONE FUROATE; NAT. REGISTRATION NO/DATE: NL 19601 19950919; FIRST REGISTRATION: GR - 10681/89 19900622 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ASMANEX HFA
More… ↓


