Market Dynamics and Financial Trajectory for Azor (Amlodipine and Olmesartan)
Introduction to Azor
Azor, a combination medication of amlodipine and olmesartan, is widely used to treat hypertension (high blood pressure). Amlodipine, a calcium channel blocker, relaxes blood vessels to improve blood flow, while olmesartan, an angiotensin II receptor blocker, prevents blood vessel constriction. This dual-action approach makes Azor an effective treatment for patients requiring multiple therapies to manage their blood pressure.
Market Size and Forecast
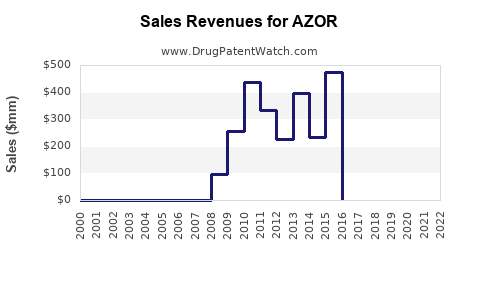
The Amlodipine and Olmesartan market, which includes Azor, was valued at USD 597.95 million in 2023 and is projected to reach USD 824.65 million by 2031, growing at a Compound Annual Growth Rate (CAGR) of 4.10% from 2024 to 2031[1].
Key Drivers of Market Growth
Increasing Prevalence of Hypertension
The global incidence of cardiovascular diseases, including hypertension, is on the rise. This trend is expected to drive the demand for antihypertensive medications like Azor. In regions such as North America and the Asia Pacific, the high prevalence of hypertension among the adult population is a significant driver of market growth[1].
Advancements in Drug Formulations
Advancements in drug formulations and the increased emphasis on combination therapies are expected to enhance patient outcomes, thereby boosting the demand for Azor. New formulations and generic versions entering the market are likely to cater to a broader patient base[1].
Regulatory and Public Health Initiatives
Strong regulatory support and favorable reimbursement policies in regions like North America encourage the adoption of innovative healthcare solutions, including Azor. Public health initiatives aimed at reducing cardiovascular risks also contribute to the growing demand for this medication[1].
Market Challenges
Generic Competition
The entry of generic versions of Azor poses a significant challenge to the market growth of branded products. Generic alternatives, such as those launched by Aurobindo Pharma Limited and Hikma Pharmaceuticals, can lead to price erosion and reduced profit margins for branded products[1].
Adverse Drug Reactions
Potential side effects associated with Azor, such as dizziness, swelling, or gastrointestinal issues, may deter some patients from continuing their prescribed treatment. This can impact market demand and patient adherence to the medication[1].
Regulatory Hurdles
The lengthy and complex approval process for new formulations and combination therapies can delay the introduction of innovative products in the market, limiting growth opportunities[1].
Economic Factors
Economic instability and healthcare budget constraints can affect drug pricing and reimbursement policies, limiting access to essential medications like Azor, particularly in low- and middle-income countries[1].
Regional Market Dynamics
North America
North America is the dominant region in the Amlodipine and Olmesartan market, driven by a robust healthcare infrastructure, high prevalence of hypertension, and increased awareness of cardiovascular diseases. The United States and Canada are key contributors to market growth due to the presence of well-established pharmaceutical companies and advanced research and development facilities[1].
Asia Pacific
The Asia Pacific region is the fastest-growing market for Azor, driven by rapid urbanization, rising disposable incomes, and increasing awareness of health and wellness. Countries like India and China are experiencing significant growth due to the rising prevalence of lifestyle-related diseases, including hypertension[1].
Application Segments
Hospitals
The hospitals segment dominates the market due to comprehensive patient management capabilities and the availability of specialized medical personnel to prescribe and administer Azor. Hospitals play a crucial role in the initial treatment and stabilization of patients with hypertension[1].
Drug Stores
The drug stores segment is the fastest-growing segment, driven by increasing patient self-management of chronic conditions and a growing preference for over-the-counter access to antihypertensive medications[1].
Financial Impact of Generic Versions
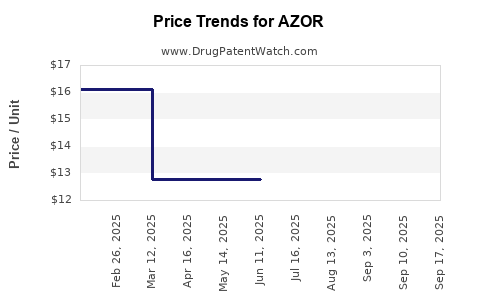
The launch of generic versions of Azor, such as those by Aurobindo Pharma Limited and Hikma Pharmaceuticals, is expected to significantly impact the financial trajectory of the branded product. Generic versions can reduce the market share and revenue of branded Azor, as they offer lower-priced alternatives. For example, the generic version of Azor can cost as low as $98.51 for 30 tablets, compared to the branded version which can cost around $410 for the same quantity[1][5].
Industry Expert Insights
Industry experts highlight the importance of managing hypertension effectively to reduce cardiovascular risks. According to the American Heart Association, there is a growing emphasis on managing hypertension, which has resulted in a surge in prescriptions for medications like Azor[1].
Illustrative Statistics
- Approximately 47% of adults in the U.S. have hypertension, underscoring the high demand for effective antihypertensive medications like Azor[1].
- The World Health Organization (WHO) estimates that hypertension affects about 30% of the adult population in countries like India and China, highlighting a vast potential customer base for these medications[1].
Key Takeaways
- The Amlodipine and Olmesartan market, including Azor, is projected to grow at a CAGR of 4.10% from 2024 to 2031.
- The market is driven by the increasing prevalence of hypertension, advancements in drug formulations, and strong regulatory support.
- Generic competition, adverse drug reactions, regulatory hurdles, and economic factors pose significant challenges to market growth.
- North America and the Asia Pacific are key regions driving market growth.
- Hospitals and drug stores are the primary application segments, with hospitals dominating due to comprehensive patient management.
FAQs
1. What is Azor used for?
Azor is used to treat hypertension (high blood pressure) by combining amlodipine, a calcium channel blocker, and olmesartan, an angiotensin II receptor blocker.
2. What is the projected market size of Azor by 2031?
The Amlodipine and Olmesartan market, which includes Azor, is projected to reach USD 824.65 million by 2031[1].
3. What are the main challenges facing the Azor market?
The main challenges include generic competition, adverse drug reactions, regulatory hurdles, and economic factors such as healthcare budget constraints[1].
4. Which regions are driving the growth of the Azor market?
North America and the Asia Pacific are the key regions driving market growth, with North America being the dominant region and the Asia Pacific being the fastest-growing region[1].
5. How do generic versions impact the Azor market?
Generic versions of Azor can lead to price erosion and reduced profit margins for branded products, as they offer lower-priced alternatives to patients[1][5].
Cited Sources:
- Verified Market Research: Amlodipine And Olmesartan Market Size, Share, Trends & Forecast.
- Daiichi Sankyo: Annual Report.
- ASPE: Explaining the Growth in Prescription Drug Spending: A Review of Recent Studies.
- iHealthcareAnalyst: Antihypertensive Drugs Market and Forecast 2024-2031.
- Drugs.com: Azor Prices, Coupons, Copay Cards & Patient Assistance.