BEYAZ Drug Patent Profile
✉ Email this page to a colleague
When do Beyaz patents expire, and what generic alternatives are available?
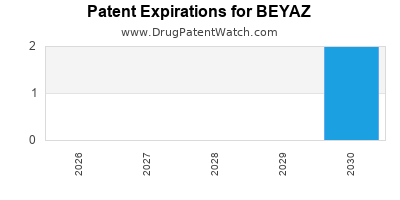
Beyaz is a drug marketed by Bayer Hlthcare and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
The generic ingredient in BEYAZ is drospirenone; ethinyl estradiol; levomefolate calcium. There are eleven drug master file entries for this compound. Three suppliers are listed for this compound. Additional details are available on the drospirenone; ethinyl estradiol; levomefolate calcium profile page.
DrugPatentWatch® Generic Entry Outlook for Beyaz
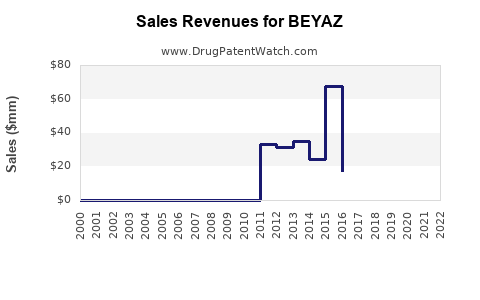
Annual sales in 2021 were $8mm indicating the motivation for generic entry (peak sales were $68mm in 2015).
There have been two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for BEYAZ?
- What are the global sales for BEYAZ?
- What is Average Wholesale Price for BEYAZ?
Summary for BEYAZ
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Patent Applications: | 8 |
| Drug Prices: | Drug price information for BEYAZ |
| Drug Sales Revenues: | Drug sales revenues for BEYAZ |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BEYAZ |
| What excipients (inactive ingredients) are in BEYAZ? | BEYAZ excipients list |
| DailyMed Link: | BEYAZ at DailyMed |
Paragraph IV (Patent) Challenges for BEYAZ
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| BEYAZ | Tablets | drospirenone; ethinyl estradiol; levomefolate calcium | 3 mg/0.02 mg/ 0.451 mg and 0.451 mg | 022532 | 1 | 2012-11-13 |
US Patents and Regulatory Information for BEYAZ
BEYAZ is protected by five US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | AB | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | ||
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | AB | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for BEYAZ
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | ⤷ Try for Free | ⤷ Try for Free |
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | ⤷ Try for Free | ⤷ Try for Free |
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | ⤷ Try for Free | ⤷ Try for Free |
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | ⤷ Try for Free | ⤷ Try for Free |
| Bayer Hlthcare | BEYAZ | drospirenone; ethinyl estradiol; levomefolate calcium | TABLET;ORAL | 022532-001 | Sep 24, 2010 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for BEYAZ
See the table below for patents covering BEYAZ around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hungary | 0001560 | ⤷ Try for Free | |
| Japan | 2013047279 | STABLE CRYSTALLINE 5-METHYLTETRAHYDROFOLATE SALT | ⤷ Try for Free |
| Norway | 20001886 | ⤷ Try for Free | |
| Russian Federation | 2265605 | КРИСТАЛЛИЧЕСКИЕ ЩЕЛОЧНО-ЗЕМЕЛЬНЫЕ СОЛИ 5-МЕТИЛТЕТРАГИДРОФОЛИЕВОЙ КИСЛОТЫ (ВАРИАНТЫ), СПОСОБ ИХ ПОЛУЧЕНИЯ И КОМПОЗИЦИЯ ДЛЯ ФАРМАЦЕВТИЧЕСКИХ СРЕДСТВ И ПИЩЕВЫХ ДОБАВОК (5-METHYLTETRAHYDROFOLIC ACID CRYSTALLINE ALKALINE-EARTH SALTS (VARIANTS), METHOD FOR THEIR PREPARING AND COMPOSITION FOR PHARMACEUTICAL AGENTS AND NUTRITION SUPPLEMENTS) | ⤷ Try for Free |
| Serbia | 20090299 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BEYAZ
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0398460 | 04C0022 | France | ⤷ Try for Free | PRODUCT NAME: ESTRADIOL ANHYDRE DROSPIRENONE; REGISTRATION NO/DATE IN FRANCE: NL 28661 DU 20040316; REGISTRATION NO/DATE AT EEC: RVG 27505 DU 20021211 |
| 0771217 | 07C0001 | France | ⤷ Try for Free | PRODUCT NAME: ETHINYLESTRADIOL BETADEX CLATHRATE; NAT. REGISTRATION NO/DATE: NL 32343 20060710; FIRST REGISTRATION: NL - RVG 31781 20050804 |
| 3632448 | 22C1031 | France | ⤷ Try for Free | PRODUCT NAME: DROSPIRENONE; NAT. REGISTRATION NO/DATE: NL49691 20191121; FIRST REGISTRATION: DK - 61678 20191016 |
| 2588114 | LUC00227 | Luxembourg | ⤷ Try for Free | PRODUCT NAME: DROSPIRENONE; AUTHORISATION NUMBER AND DATE: 31332 20191022 |
| 0398460 | SPC/GB04/032 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: ESTRADIOL, OPTIONALLY IN THE FORM OF A HYDRATE, TOGETHER WITH DROSPIRENONE; REGISTERED: NL RVG 27505 20021211; UK PL 00053/0341 20040310 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Beyaz
More… ↓


