CICLOPIROX Drug Patent Profile
✉ Email this page to a colleague
When do Ciclopirox patents expire, and when can generic versions of Ciclopirox launch?
Ciclopirox is a drug marketed by Cosette, Fougera Pharms, Glenmark Pharms, Padagis Israel, Taro, Alvogen, Glenmark Pharms Inc, Padagis Us, Actavis Mid Atlantic, Encube, Acella, Chartwell Rx, Epic Pharma Llc, Hikma, Mylan Pharms Inc, Rising, Taro Pharm Inds, and Teva Pharms. and is included in twenty-eight NDAs.
The generic ingredient in CICLOPIROX is ciclopirox. There are ten drug master file entries for this compound. Fourteen suppliers are listed for this compound. Additional details are available on the ciclopirox profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Ciclopirox
A generic version of CICLOPIROX was approved as ciclopirox by FOUGERA PHARMS on August 6th, 2004.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for CICLOPIROX?
- What are the global sales for CICLOPIROX?
- What is Average Wholesale Price for CICLOPIROX?
Summary for CICLOPIROX
| US Patents: | 0 |
| Applicants: | 18 |
| NDAs: | 28 |
| Finished Product Suppliers / Packagers: | 14 |
| Raw Ingredient (Bulk) Api Vendors: | 107 |
| Clinical Trials: | 13 |
| Patent Applications: | 4,085 |
| Drug Prices: | Drug price information for CICLOPIROX |
| Drug Sales Revenues: | Drug sales revenues for CICLOPIROX |
| What excipients (inactive ingredients) are in CICLOPIROX? | CICLOPIROX excipients list |
| DailyMed Link: | CICLOPIROX at DailyMed |
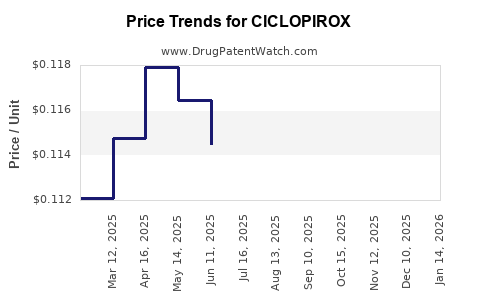
See drug prices for CICLOPIROX
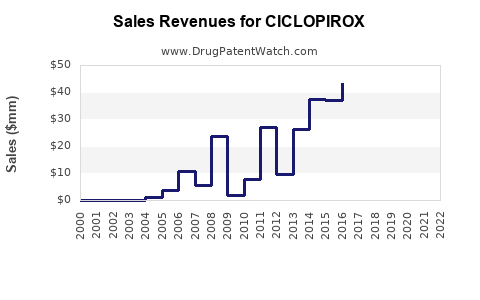
Recent Clinical Trials for CICLOPIROX
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Universidad Complutense de Madrid | Phase 4 |
| Atlas Molecular Pharma | Phase 1 |
| BCDiabetes.Ca | N/A |
Pharmacology for CICLOPIROX
| Mechanism of Action | Protein Synthesis Inhibitors |
| Physiological Effect | Decreased DNA Replication Decreased Protein Synthesis Decreased RNA Replication |
Anatomical Therapeutic Chemical (ATC) Classes for CICLOPIROX
Paragraph IV (Patent) Challenges for CICLOPIROX
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CICLOPIROX | Gel | ciclopirox | 0.77% | 020519 | 1 | 2006-05-10 |
US Patents and Regulatory Information for CICLOPIROX
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taro | CICLOPIROX | ciclopirox | SUSPENSION;TOPICAL | 077092-001 | Aug 10, 2005 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Padagis Israel | CICLOPIROX | ciclopirox | CREAM;TOPICAL | 077364-001 | Mar 3, 2006 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Encube | CICLOPIROX | ciclopirox | SOLUTION;TOPICAL | 077687-001 | Sep 18, 2007 | AT | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Fougera Pharms | CICLOPIROX | ciclopirox | GEL;TOPICAL | 077896-001 | Jun 10, 2008 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Taro Pharm Inds | CICLOPIROX | ciclopirox | SOLUTION;TOPICAL | 078144-001 | Sep 18, 2007 | AT | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Cosette | CICLOPIROX | ciclopirox | CREAM;TOPICAL | 078463-001 | Dec 20, 2010 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CICLOPIROX
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Alvogen | CICLOPIROX | ciclopirox | GEL;TOPICAL | 020519-001 | Jul 21, 1997 | ⤷ Try for Free | ⤷ Try for Free |
| Alvogen | CICLOPIROX | ciclopirox | GEL;TOPICAL | 020519-001 | Jul 21, 1997 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for CICLOPIROX
See the table below for patents covering CICLOPIROX around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Argentina | 008858 | PREPARADOS FARMACEUTICOS QUE CONTIENEN DERIVADOS 1-HIDROXI-2-(1H) PIRIDONA, PROCEDIMIENTO PARA SU OBTENCION Y EMPLEO DE LOS MISMOS PARA LA PREPARACION DEUN MEDICAMENTO PARA EL TRATAMIENTO Y PROFILAXIS DE MICOSIS CUTANEAS. | ⤷ Try for Free |
| Bulgaria | 103262 | ⤷ Try for Free | |
| Serbia | 49596 | PRIMENA FARMACEUTSKE KOMPOZICIJE ZA DOBIJANJE LEKA U OBLIKU GELA (USE OF PHARMACEUTICAL COMPOSITION FOR OBTAINING GEL FORM OF A MEDICINE) | ⤷ Try for Free |
| Slovenia | 0928192 | ⤷ Try for Free | |
| South Korea | 20000048620 | ⤷ Try for Free | |
| Turkey | 9900723 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Ciclopirox Market Dynamics and Financial Trajectory
More… ↓


