COREG CR Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Coreg Cr, and what generic alternatives are available?
Coreg Cr is a drug marketed by Waylis Therap and is included in one NDA. There is one patent protecting this drug and three Paragraph IV challenges.
This drug has twenty-four patent family members in fourteen countries.
The generic ingredient in COREG CR is carvedilol phosphate. There are thirty-seven drug master file entries for this compound. Ten suppliers are listed for this compound. Additional details are available on the carvedilol phosphate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Coreg Cr
A generic version of COREG CR was approved as carvedilol phosphate by SUN PHARM INDUSTRIES on October 25th, 2017.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for COREG CR?
- What are the global sales for COREG CR?
- What is Average Wholesale Price for COREG CR?
Summary for COREG CR
| International Patents: | 24 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 3 |
| Raw Ingredient (Bulk) Api Vendors: | 28 |
| Patent Applications: | 29 |
| Drug Prices: | Drug price information for COREG CR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for COREG CR |
| What excipients (inactive ingredients) are in COREG CR? | COREG CR excipients list |
| DailyMed Link: | COREG CR at DailyMed |


Pharmacology for COREG CR
| Drug Class | alpha-Adrenergic Blocker beta-Adrenergic Blocker |
| Mechanism of Action | Adrenergic alpha-Antagonists Adrenergic beta1-Antagonists Adrenergic beta2-Antagonists |
Paragraph IV (Patent) Challenges for COREG CR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| COREG CR | Extended-release Capsules | carvedilol phosphate | 10 mg and 20 mg | 022012 | 1 | 2008-03-18 |
| COREG CR | Extended-release Capsules | carvedilol phosphate | 40 mg | 022012 | 1 | 2007-12-21 |
| COREG CR | Extended-release Capsules | carvedilol phosphate | 80 mg | 022012 | 1 | 2007-11-19 |
US Patents and Regulatory Information for COREG CR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Waylis Therap | COREG CR | carvedilol phosphate | CAPSULE, EXTENDED RELEASE;ORAL | 022012-001 | Oct 20, 2006 | DISCN | Yes | No | 8,101,209*PED | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Waylis Therap | COREG CR | carvedilol phosphate | CAPSULE, EXTENDED RELEASE;ORAL | 022012-002 | Oct 20, 2006 | DISCN | Yes | No | 8,101,209*PED | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Waylis Therap | COREG CR | carvedilol phosphate | CAPSULE, EXTENDED RELEASE;ORAL | 022012-003 | Oct 20, 2006 | DISCN | Yes | No | 8,101,209*PED | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Waylis Therap | COREG CR | carvedilol phosphate | CAPSULE, EXTENDED RELEASE;ORAL | 022012-004 | Oct 20, 2006 | DISCN | Yes | No | 8,101,209*PED | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for COREG CR
International Patents for COREG CR
See the table below for patents covering COREG CR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Eurasian Patent Organization | 200500091 | ФОСФАТНЫЕ СОЛИ КАРВЕДИЛОЛА И/ИЛИ ИХ СОЛЬВАТЫ, СООТВЕТСТВУЮЩИЕ КОМПОЗИЦИИ И/ИЛИ СПОСОБЫ ЛЕЧЕНИЯ | ⤷ Try for Free |
| European Patent Office | 0709087 | Microcapsules médicamenteuses et/ou nutritionnelles pour administration per os (Medicinal and/or nutritional microcapsules for oral administration) | ⤷ Try for Free |
| Germany | 69602424 | ⤷ Try for Free | |
| Japan | H10509427 | ⤷ Try for Free | |
| Japan | H0123462 | ⤷ Try for Free | |
| Latvia | 5234 | (Karbazolil-4-oksi)propanolamina atvasinajumu un to saju iegusanas panemiens (varianti) | ⤷ Try for Free |
| Mexico | PA04012923 | SALES DE FOSFATO DE CARVEDILOL Y/O SOLVATOS DE LAS MISMAS, COMPOSICIONES CORRESPONDIENTES Y/O METODOS DE TRATAMIENTO. (CARVEDILOL PHOSPHATE SALTS AND/OR SOLVATES THEREOF, CORRESPONDINQ COMPOSITIONS, AND/OR METHODS OF TREATMENT.) | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
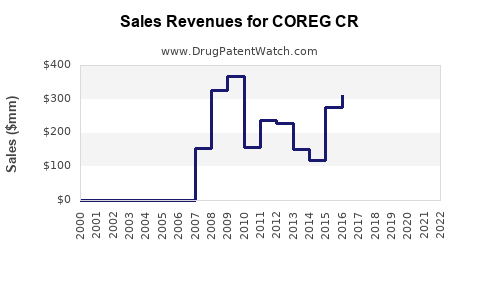
Market Dynamics and Financial Trajectory for COREG CR
More… ↓


