DESCOVY Drug Patent Profile
✉ Email this page to a colleague
When do Descovy patents expire, and what generic alternatives are available?
Descovy is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are three patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-two patent family members in fifty countries.
The generic ingredient in DESCOVY is emtricitabine; tenofovir alafenamide fumarate. There are eighteen drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the emtricitabine; tenofovir alafenamide fumarate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Descovy
A generic version of DESCOVY was approved as emtricitabine; tenofovir alafenamide fumarate by LUPIN LTD on December 13th, 2024.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for DESCOVY?
- What are the global sales for DESCOVY?
- What is Average Wholesale Price for DESCOVY?
Summary for DESCOVY
| International Patents: | 132 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 17 |
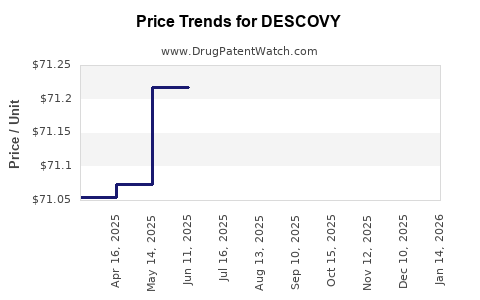
| Drug Prices: | Drug price information for DESCOVY |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DESCOVY |
| What excipients (inactive ingredients) are in DESCOVY? | DESCOVY excipients list |
| DailyMed Link: | DESCOVY at DailyMed |
Paragraph IV (Patent) Challenges for DESCOVY
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| DESCOVY | Tablets | emtricitabine; tenofovir alafenamide fumarate | 120 mg/15 mg | 208215 | 1 | 2022-10-31 |
| DESCOVY | Tablets | emtricitabine; tenofovir alafenamide fumarate | 200 mg/25 mg | 208215 | 6 | 2019-11-05 |
US Patents and Regulatory Information for DESCOVY
DESCOVY is protected by three US patents and one FDA Regulatory Exclusivity.
Expired US Patents for DESCOVY
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Try for Free | ⤷ Try for Free |
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Try for Free | ⤷ Try for Free |
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Try for Free | ⤷ Try for Free |
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Try for Free | ⤷ Try for Free |
| Gilead Sciences Inc | DESCOVY | emtricitabine; tenofovir alafenamide fumarate | TABLET;ORAL | 208215-001 | Apr 4, 2016 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for DESCOVY
When does loss-of-exclusivity occur for DESCOVY?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 39
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Try for Free
Argentina
Patent: 7546
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA
Estimated Expiration: ⤷ Try for Free
Australia
Patent: 12296622
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Try for Free
Patent: 14271320
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Try for Free
Brazil
Patent: 2014003420
Patent: hemifumarato de tenofovir alafenamida
Estimated Expiration: ⤷ Try for Free
Canada
Patent: 45553
Patent: HEMIFUMARATE DE TENOFOVIR ALAFENAMIDE (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Chile
Patent: 14000370
Patent: Hemifumarato de tenofovir alafenamida; metodo de preparación; composicion farmaceutica que lo comprende; metodo para preparar la composicion farmaceutica; y uso en el tratamiento de una infeccion por hiv.
Estimated Expiration: ⤷ Try for Free
China
Patent: 3732594
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Try for Free
Patent: 0343135
Patent: 替诺福韦艾拉酚胺(TENOFOVIR ALAFENAMIDE)半反丁烯二酸盐 (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Colombia
Patent: 80063
Patent: Hemifumarato de tenofovir alafenamida
Estimated Expiration: ⤷ Try for Free
Costa Rica
Patent: 140072
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA
Estimated Expiration: ⤷ Try for Free
Croatia
Patent: 0161696
Estimated Expiration: ⤷ Try for Free
Cyprus
Patent: 18385
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 44810
Estimated Expiration: ⤷ Try for Free
Ecuador
Patent: 14013206
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA
Estimated Expiration: ⤷ Try for Free
Eurasian Patent Organization
Patent: 7768
Patent: ТЕНОФОВИР АЛАФЕНАМИДА ГЕМИФУМАРАТ (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 1490208
Patent: ТЕНОФОВИР АЛАФЕНАМИДА ГЕМИФУМАРАТ
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 44810
Patent: HÉMIFUMARATE DE TÉNOFOVIR ALAFÉNAMIDE (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 70088
Patent: TÉNOFOVIR ALAFENAMIDE HÉMIFUMARATE (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 31832
Patent: TÉNOFOVIR ALAFENAMIDE HÉMIFUMARATE (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Finland
Patent: 44810
Estimated Expiration: ⤷ Try for Free
Hong Kong
Patent: 99026
Patent: 替諾福韋艾拉酚胺 半反丁烯二酸鹽 (TENOFOVIR ALAFENAMIDE HEMIFUMARATE (TENOFOVIRALAFENAMIDE))
Estimated Expiration: ⤷ Try for Free
Hungary
Patent: 31253
Estimated Expiration: ⤷ Try for Free
India
Patent: 12DEN2014
Estimated Expiration: ⤷ Try for Free
Israel
Patent: 0949
Patent: טנופוביר אלאפנאמיד המיפומאראט (Tenofovir alafenamide hemifumarate)
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 51275
Estimated Expiration: ⤷ Try for Free
Patent: 56537
Estimated Expiration: ⤷ Try for Free
Patent: 80162
Estimated Expiration: ⤷ Try for Free
Patent: 14528924
Patent: テノホビルアラフェナミドヘミフマレート
Estimated Expiration: ⤷ Try for Free
Patent: 15038149
Patent: テノホビルアラフェナミドヘミフマレート (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 16169228
Patent: テノホビルアラフェナミドヘミフマレート (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 18065870
Patent: テノホビルアラフェナミドヘミフマレート (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Patent: 20040972
Patent: テノホビルアラフェナミドヘミフマレート (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Lithuania
Patent: 44810
Estimated Expiration: ⤷ Try for Free
Mexico
Patent: 6627
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA. (TENOFOVIR ALAFENAMIDE HEMIFUMARATE.)
Estimated Expiration: ⤷ Try for Free
Patent: 14001549
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA. (TENOFOVIR ALAFENAMIDE HEMIFUMARATE.)
Estimated Expiration: ⤷ Try for Free
Moldova, Republic of
Patent: 08
Patent: Hemifumarat de tenofovir alafenamidă şi utilizările sale medicale (Tenofovir alafenamide hemifumarate and its medical uses)
Estimated Expiration: ⤷ Try for Free
Patent: 140011
Patent: Tenofovir alafenamid hemifumarat (Tenofovir alafenamide hemifumarate)
Estimated Expiration: ⤷ Try for Free
Montenegro
Patent: 612
Patent: TENOFOVIR ALAFENAMID HEMIFUMARAT (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Morocco
Patent: 350
Patent: Hemifumarate de tenofovir alafenamide
Estimated Expiration: ⤷ Try for Free
New Zealand
Patent: 0421
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Try for Free
Peru
Patent: 141328
Patent: TENOFOVIR ALAFENAMIDA HEMIFUMARATO
Estimated Expiration: ⤷ Try for Free
Poland
Patent: 44810
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 44810
Estimated Expiration: ⤷ Try for Free
San Marino
Patent: 01600476
Patent: TENOFOVIR ALAFENAMMIDE EMIFUMARATO
Estimated Expiration: ⤷ Try for Free
Serbia
Patent: 353
Patent: TENOFOVIR ALAFENAMID HEMIFUMARAT (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Singapore
Patent: 14011548
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 44810
Estimated Expiration: ⤷ Try for Free
South Africa
Patent: 1400582
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Try for Free
South Korea
Patent: 1612642
Estimated Expiration: ⤷ Try for Free
Patent: 140054068
Patent: TENOFOVIR ALAFENAMIDE HEMIFUMARATE
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 08871
Estimated Expiration: ⤷ Try for Free
Taiwan
Patent: 16499
Estimated Expiration: ⤷ Try for Free
Patent: 1321396
Patent: Tenofovir alafenamide hemifumarate
Estimated Expiration: ⤷ Try for Free
Ukraine
Patent: 5311
Patent: ГЕМІФУМАРАТ ТЕНОФОВІРУ АЛАФЕНАМІДУ (TENOFOVIR ALAFENAMIDE HEMIFUMARATE)
Estimated Expiration: ⤷ Try for Free
Uruguay
Patent: 262
Patent: HEMIFUMARATO DE TENOFOVIR ALAFENAMIDA, SUS COMPOSICIONES, MÉTODO DE PREPARACIÓN, MÉTODOS PARA TRATAR INFECCIONES VIRALES Y SU USO PARA PREPARAR MEDI CAMENTOS
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering DESCOVY around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Bulgaria | 107572 | ⤷ Try for Free | |
| European Patent Office | 1808434 | ⤷ Try for Free | |
| European Patent Office | 0984013 | Activité antivirale et résolution de 2-hydroxymethyl-5-(5-fluorocytosin-1-yl)-1,3-oxathiolane (Antiviral activity and resolution of 2-hydroxymethyl-5-(5-fluorocytosin-1-yl)-1, 3-oxathiolane) | ⤷ Try for Free |
| Netherlands | 300148 | ⤷ Try for Free | |
| Russian Federation | 2125558 | METHOD OF SYNTHESIS OF 2',3'-DIDEOXY-3'-THIACYTYDINE OR 2',3'- -DIDEOXY-3'-THIA-5-FLUOROCYTIDINE, NUCLEOSIDE ENRICHED WITH ENANTIOMER, METHOD OF SYNTHESIS OF 1,3-OXATHIOLANE, METHOD OF SYNTHESIS OF 2-ACYLOXY-5-ACYLOXY-1,3-OXATHIOLANE ENRICHED WITH ENANTIOMER, ENANTIOMERS, METHOD OF CLEAVAGE OF NUCLEOSIDE ENANTIOMERS | ⤷ Try for Free |
| South Korea | 20140054068 | TENOFOVIR ALAFENAMIDE HEMIFUMARATE | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DESCOVY
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1301519 | 132016000035069 | Italy | ⤷ Try for Free | PRODUCT NAME: TENOFOVIR ALAFENAMIDE O UN SUO SALE O UN SOLVATO DELLO STESSO, IN PARTICOLARE TENOFOVIR ALAFENAMIDE FUMARATO(GENVOYA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/15/1061, 20151123 |
| 1663240 | 15C0071 | France | ⤷ Try for Free | PRODUCT NAME: COMBINAISON DE RILPIVIRINE OU SES FORMES THERAPEUTIQUEMENT EQUIVALENTES PROTEGEES PAR LE BREVET DE BASE TELLES QUE LES SELS PHARMACEUTIQUEMENT ACCEPTABLES DE RILPIVIRINE Y COMPRIS LE SEL D'ACIDE CHLORHYDRIQUE DE RILPIVIRINE AINSI QUE D'EMTRICITABINE; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 1301519 | 1690012-8 | Sweden | ⤷ Try for Free | PRODUCT NAME: TENOFOVIR ALAFENAMIDE OR A SALT OR SOLVATE THEREOF, IN PARTICULAR TENOFOVIR ALAFENAMIDE FUMARATE; REG. NO/DATE: EU/1/15/1061/001-002 20151123 |
| 1301519 | 300803 | Netherlands | ⤷ Try for Free | PRODUCT NAME: TENOFOVIRALAFENAMIDE OF EEN ZOUT OF SOLVAAT DAARVAN, MET NAME TENOFOVIRALAFENAMIDEFUMARAAT; NATIONAL REGISTRATION NO/DATE: EU/1/15/1061/002 20151123; FIRST REGISTRATION: EU EU/1/15/1061/001 20151123 |
| 0513200 | C00513200/01 | Switzerland | ⤷ Try for Free | PRODUCT NAME: EMTRICITABINE; REGISTRATION NUMBER/DATE: SWISSMEDIC 56880 25.10.2004 |
| 1301519 | CR 2016 00012 | Denmark | ⤷ Try for Free | PRODUCT NAME: TENOFOVIRALAFENAMID ELLER ET SALT ELLER SOLVAT DERAF, I SAERDELESHED TENOFOVIRALAFENAMIDFUMARAT; REG. NO/DATE: EU/1/15/1061/001-002 20151123 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of Descovy
More… ↓


