DULERA Drug Patent Profile
✉ Email this page to a colleague
When do Dulera patents expire, and what generic alternatives are available?
Dulera is a drug marketed by Organon Llc and is included in one NDA.
The generic ingredient in DULERA is formoterol fumarate; mometasone furoate. There are nineteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the formoterol fumarate; mometasone furoate profile page.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for DULERA?
- What are the global sales for DULERA?
- What is Average Wholesale Price for DULERA?
Summary for DULERA
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 4 |
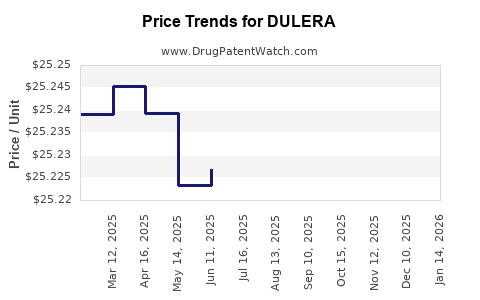
| Drug Prices: | Drug price information for DULERA |
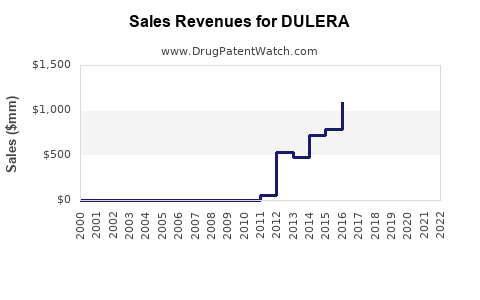
| Drug Sales Revenues: | Drug sales revenues for DULERA |
| What excipients (inactive ingredients) are in DULERA? | DULERA excipients list |
| DailyMed Link: | DULERA at DailyMed |
Recent Clinical Trials for DULERA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Sanofi | Phase 2 |
| Regeneron Pharmaceuticals | Phase 2 |
| Asthma Management Systems | Phase 4 |
Pharmacology for DULERA
| Drug Class | Corticosteroid beta2-Adrenergic Agonist |
| Mechanism of Action | Adrenergic beta2-Agonists Corticosteroid Hormone Receptor Agonists |
US Patents and Regulatory Information for DULERA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-001 | Jun 22, 2010 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-002 | Jun 22, 2010 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-003 | Aug 12, 2019 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for DULERA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-002 | Jun 22, 2010 | 6,068,832 | ⤷ Try for Free |
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-001 | Jun 22, 2010 | 6,068,832 | ⤷ Try for Free |
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-001 | Jun 22, 2010 | 6,677,323 | ⤷ Try for Free |
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-001 | Jun 22, 2010 | 5,889,015*PED | ⤷ Try for Free |
| Organon Llc | DULERA | formoterol fumarate; mometasone furoate | AEROSOL, METERED;INHALATION | 022518-002 | Jun 22, 2010 | 6,677,323 | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for DULERA
See the table below for patents covering DULERA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Australia | 691880 | ⤷ Try for Free | |
| China | 101164542 | ⤷ Try for Free | |
| Hungary | T74884 | ⤷ Try for Free | |
| Spain | 2369516 | ⤷ Try for Free | |
| United Kingdom | 9904919 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DULERA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2435024 | 21C1020 | France | ⤷ Try for Free | PRODUCT NAME: COMBINAISON DE FORMOTEROL (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI), GLYCOPYRROLATE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI) ET BUDESONIDE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI); REGISTRATION NO/DATE: EU/1/20/1498 20201210 |
| 2435025 | CR 2019 00032 | Denmark | ⤷ Try for Free | PRODUCT NAME: KOMBINATION AF GLYCOPYRROLAT, HERUNDER ALLE FARMACEUTISK ACCEPTABLE SALTE, ESTERE, ENANTIOMERER ELLER SOLVATER DERAF, OG FORMOTEROL, HERUNDER ALLE FARMACEUTISK ACCEPTABLE SALTE, ESTERE, ENANTIOMERE ELLER SOLVATER DERAF; REG. NO/DATE: EU/1/18/1339 20181220 |
| 2435025 | LUC00124 | Luxembourg | ⤷ Try for Free | PRODUCT NAME: UNE COMBINAISON DE GLYCOPYRROLATE (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES) ET DE FORMOTEROL (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES); AUTHORISATION NUMBER AND DATE: EU/1/18/1339 20181220 |
| 2435025 | PA2019014,C2435025 | Lithuania | ⤷ Try for Free | PRODUCT NAME: GLIKOPIRONIO BROMIDAS/FORMOTEROLIS; REGISTRATION NO/DATE: EU/1/18/1339 20181218 |
| 2435024 | SPC/GB21/029 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: A COMBINATION OF FORMOTEROL, INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ESTERS AND SOLVATES THEREOF, GLYCOPYRROLATE, INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ESTERS AND SOLVATES THEREOF, AND BUDESONIDE INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ES; REGISTERED: UK EU/1/20/1498 (NI) 20201210; UK PLGB 17901/0352-001 20201210 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for DULERA
More… ↓


