Market Overview
The ketoconazole market is experiencing significant growth, driven by several key factors. As of 2023, the global ketoconazole market was valued at USD 782.3 million and is projected to reach USD 1.2 billion by 2031, growing at a compound annual growth rate (CAGR) of 4.6% from 2024 to 2031[1].
Driving Factors
Increasing Prevalence of Fungal Infections
The rising incidence of fungal infections, particularly in tropical and subtropical regions, is a major driver of the ketoconazole market. This increase is attributed to compromised immune systems and the adaptation of harmful pathogens to warmer temperatures[4].
Growing Demand for Antifungal Medications
There is a growing need for potent antifungal medications like ketoconazole, which is fueling market growth. The increased awareness and accessibility of over-the-counter antifungal drugs have expanded the market reach[1].
Expansion of the Pharmaceutical Sector
The continuous growth and development of the pharmaceutical sector, along with ongoing research and development of new formulations, are enhancing the market potential for ketoconazole. Pharmaceutical companies are investing heavily in finding new therapeutic options, which includes the development of antifungal agents[4].
Rising Prevalence of Skin Diseases
The increasing prevalence of skin diseases such as seborrheic dermatitis and dandruff, which can be effectively treated with ketoconazole-based treatments, is another significant factor driving market growth[1].
Market Segmentation
By Application
The ketoconazole market is segmented into various applications, including fungal infections, dermatological conditions, seborrheic dermatitis, dandruff, and tinea infections. The antifungal segment holds a significant share due to the surge in compromised immune systems and the adaptation of harmful pathogens[4].
By Product Type
The market is segmented by product type into ketoconazole cream, shampoo, tablets, solution, and foam. The ketoconazole topical segment, particularly the shampoo, has a substantial market share due to its effectiveness in treating scalp conditions like dandruff[4].
Geographical Analysis
Regional Growth
The market is geographically segmented into North America, Europe, Asia-Pacific, South America, and the Middle East and Africa. The Asia-Pacific region is expected to grow with the highest CAGR during the forecast period due to the growing number of ketoconazole manufacturing companies and increased demand for skincare products in this region[4].
Country-Specific Trends
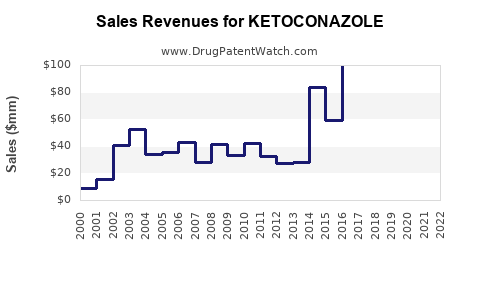
North America is also a significant market, with the U.S. annual sales for ketoconazole shampoo alone totaling approximately $69.2 million as of July 2024. This indicates a strong demand for ketoconazole products in this region[3].
Competitive Landscape
Key Players
The market is competitive, with key players such as Mylan, Johnson & Johnson, and ANI Pharmaceuticals. These companies are focusing on expanding their product portfolios and launching new products to address patient needs. For instance, ANI Pharmaceuticals recently received FDA approval and launched a generic version of ketoconazole shampoo, 2%, which is expected to boost their revenue and market presence[3].
Strategic Moves
Companies are implementing various strategies to strengthen their market position. This includes new product launches, expansions, mergers and acquisitions, and partnerships. The competitive landscape is further analyzed through Porter’s Five Forces analysis, which includes buyers' bargaining power, suppliers' bargaining power, the threat of new entrants, the threat of substitutes, and the degree of competition[1].
Financial Trajectory
Market Size and Growth
The ketoconazole market is expected to grow significantly, with a projected market size of USD 1.2 billion by 2031. This growth is driven by the increasing demand for antifungal medications and the expanding pharmaceutical sector[1].
Revenue and Profit Margins
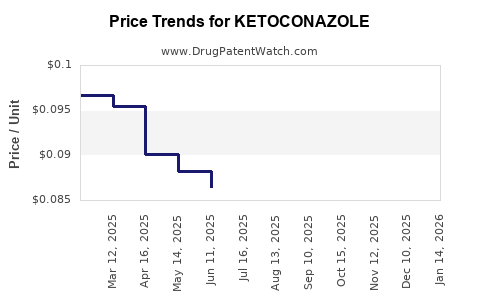
The launch of new products, such as ANI Pharmaceuticals' ketoconazole shampoo, is expected to contribute to revenue growth. However, the actual impact will depend on the company's ability to capture market share and compete with other generic versions. Generic drug pricing can be competitive, so manufacturing efficiency and distribution strategies will be crucial in determining profit margins[3].
Challenges and Opportunities
Competition and Pricing
The market faces competition from both branded and generic products, which can impact pricing and profit margins. However, the growing demand for antifungal medications presents a significant opportunity for companies to expand their market share[3].
Regulatory Approvals
Regulatory approvals, such as the FDA approval for ANI Pharmaceuticals' ketoconazole shampoo, are critical for market entry and growth. Companies must navigate regulatory landscapes to ensure timely and successful product launches[3].
Technological and R&D Trends
Advances in Formulations
The market is witnessing advances in formulations, with a focus on developing better therapeutic options. Companies are investing in research and development to create more effective and safer antifungal agents[4].
Emerging Technologies
Emerging technologies, such as improved fungal control and better product constitutions, are trends in the ketoconazole market. These advancements are expected to enhance the efficacy and safety of ketoconazole products[4].
Consumer Behavior and Market Trends
Increasing Urban Population
The rising urban population and changing lifestyles are contributing to the increased demand for skincare products, including antifungal medications. This trend is particularly evident in the Asia-Pacific region[4].
Consumer Awareness
Increased consumer awareness of antifungal therapies and the availability of over-the-counter antifungal drugs are driving market growth. Consumers are becoming more health-conscious, leading to higher demand for effective treatments[1].
Key Takeaways
- The ketoconazole market is valued at USD 782.3 million in 2023 and is expected to reach USD 1.2 billion by 2031.
- The market is driven by the increasing prevalence of fungal infections, growing demand for antifungal medications, and the expansion of the pharmaceutical sector.
- The Asia-Pacific region is expected to grow with the highest CAGR during the forecast period.
- Key players are focusing on new product launches and strategic expansions to capture market share.
- Regulatory approvals and technological advancements are crucial for market growth.
Frequently Asked Questions (FAQs)
Q1: What is the current market size of the ketoconazole market?
The ketoconazole market was valued at USD 782.3 million in 2023[1].
Q2: What is the expected CAGR of the ketoconazole market from 2024 to 2031?
The ketoconazole market is expected to grow at a CAGR of 4.6% from 2024 to 2031[1].
Q3: Which region is expected to grow with the highest CAGR during the forecast period?
The Asia-Pacific region is expected to grow with the highest CAGR during the forecast period[4].
Q4: What are the main driving factors for the growth of the ketoconazole market?
The main driving factors include the increasing prevalence of fungal infections, growing demand for antifungal medications, and the expansion of the pharmaceutical sector[1].
Q5: Which segment has the largest share of the ketoconazole market by product type?
The ketoconazole topical segment, particularly the shampoo, has a substantial market share[4].
Cited Sources:
- Market Research Intellect: Ketoconazole Market Size, Share and Forecast to 2031
- Cognitive Market Research: Ketoconazole Market Report 2024 (Global Edition)
- Stock Titan: ANI Pharmaceuticals Announces the FDA Approval and Launch of Ketoconazole Shampoo, 2%
- UnivDatos: Ketoconazole Market: Share, Size, Trends, Growth & Forecast to 2030