KISQALI Drug Patent Profile
✉ Email this page to a colleague
When do Kisqali patents expire, and when can generic versions of Kisqali launch?
Kisqali is a drug marketed by Novartis and is included in two NDAs. There are nine patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and eighty-seven patent family members in fifty-three countries.
The generic ingredient in KISQALI is letrozole; ribociclib succinate. There are twenty-four drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the letrozole; ribociclib succinate profile page.
DrugPatentWatch® Generic Entry Outlook for Kisqali
Kisqali was eligible for patent challenges on March 13, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 14, 2036. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for KISQALI?
- What are the global sales for KISQALI?
- What is Average Wholesale Price for KISQALI?
Summary for KISQALI
| International Patents: | 187 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 140 |
| Clinical Trials: | 35 |
| Patent Applications: | 921 |
| Drug Prices: | Drug price information for KISQALI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for KISQALI |
| What excipients (inactive ingredients) are in KISQALI? | KISQALI excipients list |
| DailyMed Link: | KISQALI at DailyMed |
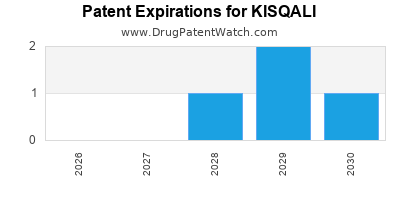

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for KISQALI
Generic Entry Date for KISQALI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for KISQALI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Michigan Rogel Cancer Center | Phase 1 |
| AstraZeneca | Phase 1/Phase 2 |
| Nationwide Children's Hospital | Phase 2 |
Pharmacology for KISQALI
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 3A Inhibitors Kinase Inhibitors |
Paragraph IV (Patent) Challenges for KISQALI
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| KISQALI | Tablets | ribociclib succinate | 200 mg | 209092 | 4 | 2021-03-15 |
US Patents and Regulatory Information for KISQALI
KISQALI is protected by twenty-seven US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of KISQALI is ⤷ Try for Free.
This potential generic entry date is based on patent 10,799,506.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
International Patents for KISQALI
When does loss-of-exclusivity occur for KISQALI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 4257
Estimated Expiration: ⤷ Try for Free
Australia
Patent: 16248017
Estimated Expiration: ⤷ Try for Free
Patent: 19201929
Estimated Expiration: ⤷ Try for Free
Patent: 20250190
Estimated Expiration: ⤷ Try for Free
Patent: 22215155
Estimated Expiration: ⤷ Try for Free
Patent: 24227794
Estimated Expiration: ⤷ Try for Free
Brazil
Patent: 2017021283
Estimated Expiration: ⤷ Try for Free
Canada
Patent: 82425
Estimated Expiration: ⤷ Try for Free
Chile
Patent: 17002593
Estimated Expiration: ⤷ Try for Free
China
Patent: 7530292
Estimated Expiration: ⤷ Try for Free
Patent: 5554257
Estimated Expiration: ⤷ Try for Free
Colombia
Patent: 17010510
Estimated Expiration: ⤷ Try for Free
Croatia
Patent: 0230053
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 83058
Estimated Expiration: ⤷ Try for Free
Ecuador
Patent: 17075052
Estimated Expiration: ⤷ Try for Free
Eurasian Patent Organization
Patent: 1792290
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 83058
Estimated Expiration: ⤷ Try for Free
Patent: 97530
Estimated Expiration: ⤷ Try for Free
Finland
Patent: 83058
Estimated Expiration: ⤷ Try for Free
Hungary
Patent: 61213
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 18514523
Estimated Expiration: ⤷ Try for Free
Mexico
Patent: 17013350
Estimated Expiration: ⤷ Try for Free
Peru
Patent: 180035
Estimated Expiration: ⤷ Try for Free
Philippines
Patent: 017501820
Estimated Expiration: ⤷ Try for Free
Poland
Patent: 83058
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 83058
Estimated Expiration: ⤷ Try for Free
Singapore
Patent: 201708084P
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 83058
Estimated Expiration: ⤷ Try for Free
South Korea
Patent: 170137101
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 38261
Estimated Expiration: ⤷ Try for Free
Taiwan
Patent: 1642864
Estimated Expiration: ⤷ Try for Free
Tunisia
Patent: 17000422
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering KISQALI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Dominican Republic | P2011000057 | COMPUESTOS DE PIRROLO-PIRIMIDINA COMO INHIBIDORES DE CDK | ⤷ Try for Free |
| Ecuador | SP17075052 | Tableta de Ribociclib | ⤷ Try for Free |
| Eurasian Patent Organization | 016301 | ПИРРОЛОПИРИМИДИНЫ И ИХ ПРИМЕНЕНИЕ (PYRROLOPYRIMIDINE COMPOUNDS AND THEIR USES) | ⤷ Try for Free |
| New Zealand | 572549 | PYRROLOPYRIMIDINE COMPOUNDS AND THEIR USES | ⤷ Try for Free |
| South Korea | 20130140222 | PYRROLOPYRIMIDINE COMPOUNDS AS CDK INHIBITORS | ⤷ Try for Free |
| Spain | 2623133 | ⤷ Try for Free | |
| Tunisia | 2011000062 | PYRROLOPYRIMIDINE COMPOUNDS OF CDK INHIBITORS | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for KISQALI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2331547 | 1790067-1 | Sweden | ⤷ Try for Free | PRODUCT NAME: RIBOCICLIB AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REG. NO/DATE: EU/1/17/1221 20170824 |
| 2331547 | 2017/060 | Ireland | ⤷ Try for Free | PRODUCT NAME: RIBOCICLIB OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/17/1221 20170824 |
| 2331547 | 2017059 | Norway | ⤷ Try for Free | PRODUCT NAME: RIBOSIKLIB ELLER ET; REG. NO/DATE: EU/1/17/1221 20170830 |
| 2331547 | 2017C/052 | Belgium | ⤷ Try for Free | PRODUCT NAME: KISQALI - RIBOCOCLIB; AUTHORISATION NUMBER AND DATE: EU/1/17/1221 20170824 |
| 2331547 | 291 26-2017 | Slovakia | ⤷ Try for Free | PRODUCT NAME: RIBOCIKLIB VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/17/1221 20170824 |
| 2331547 | 664 | Finland | ⤷ Try for Free | |
| 2331547 | C201730045 | Spain | ⤷ Try for Free | PRODUCT NAME: RIBOCICLIB O UNA SAL DE ESTE FARMACEUTICAMENTE ACEPTABLE; NATIONAL AUTHORISATION NUMBER: EU/1/17/1221; DATE OF AUTHORISATION: 20170822; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/17/1221; DATE OF FIRST AUTHORISATION IN EEA: 20170822 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for KISQALI (Ribociclib; LEE011)
More… ↓


