MACRILEN Drug Patent Profile
✉ Email this page to a colleague
When do Macrilen patents expire, and when can generic versions of Macrilen launch?
Macrilen is a drug marketed by Novo and is included in one NDA. There is one patent protecting this drug.
This drug has eleven patent family members in ten countries.
The generic ingredient in MACRILEN is macimorelin acetate. One supplier is listed for this compound. Additional details are available on the macimorelin acetate profile page.
DrugPatentWatch® Generic Entry Outlook for Macrilen
Macrilen was eligible for patent challenges on December 20, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be October 12, 2027. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for MACRILEN?
- What are the global sales for MACRILEN?
- What is Average Wholesale Price for MACRILEN?
Summary for MACRILEN
| International Patents: | 11 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 5 |
| Clinical Trials: | 2 |
| Patent Applications: | 34 |
| Drug Prices: | Drug price information for MACRILEN |
| What excipients (inactive ingredients) are in MACRILEN? | MACRILEN excipients list |
| DailyMed Link: | MACRILEN at DailyMed |
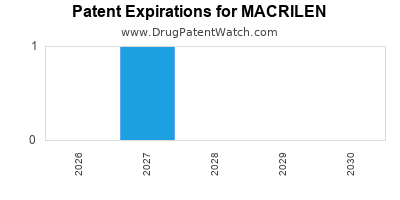

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for MACRILEN
Generic Entry Date for MACRILEN*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
FOR SOLUTION;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for MACRILEN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| VA Office of Research and Development | Phase 3 |
| AEterna Zentaris | Phase 3 |
| Novo Nordisk A/S | Phase 3 |
Pharmacology for MACRILEN
| Drug Class | Growth Hormone Secretagogue Receptor Agonist |
| Mechanism of Action | Growth Hormone Secretagogue Receptor Agonists |
US Patents and Regulatory Information for MACRILEN
MACRILEN is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of MACRILEN is ⤷ Try for Free.
This potential generic entry date is based on patent 8,192,719.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novo | MACRILEN | macimorelin acetate | FOR SOLUTION;ORAL | 205598-001 | Dec 20, 2017 | DISCN | Yes | No | 8,192,719 | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MACRILEN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novo | MACRILEN | macimorelin acetate | FOR SOLUTION;ORAL | 205598-001 | Dec 20, 2017 | 6,861,409 | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for MACRILEN
When does loss-of-exclusivity occur for MACRILEN?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Cyprus
Patent: 15080
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 84744
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 84744
Patent: METHODES ET KITS PERMETTANT DE DIAGNOSTIQUER UNE CARENCE EN HORMONE DE CROISSANCE (METHODS AND KITS TO DIAGNOSE GROWTH HORMONE DEFICIENCY)
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 52728
Estimated Expiration: ⤷ Try for Free
Patent: 09526989
Estimated Expiration: ⤷ Try for Free
Poland
Patent: 84744
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 84744
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 84744
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 04582
Estimated Expiration: ⤷ Try for Free
United Kingdom
Patent: 03295
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering MACRILEN around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 1736985 | ⤷ Try for Free | |
| Hong Kong | 1056555 | ⤷ Try for Free | |
| South Korea | 100825109 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MACRILEN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1289951 | 122019000074 | Germany | ⤷ Try for Free | PRODUCT NAME: MACIMORELIN, OPTIONAL IN FORM EINER ARZNEIMITTELZUSAMMENSETZUNG, OPTIONAL IN FORM EINES PHARMAZEUTISCH VERTRAEGLICHEN SALZES, INSBESONDERE MACIMORELIN ACETAT.; REGISTRATION NO/DATE: EU/1/18/1337 20190111 |
| 1289951 | 132019000000099 | Italy | ⤷ Try for Free | PRODUCT NAME: MACIMORELINA, OPZIONALMENTE NELLA FORMA DI UNA COMPOSIZIONE FARMACEUTICA, OPZIONALMENTE NELLA FORMA DI UN SALE FARMACEUTICAMENTE ACCETTABILE, COMPRESA MACIMORELINA ACETATO(MACIMORELIN AETERNA ZENTARIS - MACIMORELINA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/18/1337, 20190115 |
| 1289951 | CA 2019 00037 | Denmark | ⤷ Try for Free | PRODUCT NAME: MACIMORELIN, EN FARMACEUTISK SAMMENSAETNING DERAF ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/18/1337 20190115 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Macrilen (Macimorelin)
More… ↓


