NORVASC Drug Patent Profile
✉ Email this page to a colleague
When do Norvasc patents expire, and when can generic versions of Norvasc launch?
Norvasc is a drug marketed by Viatris and is included in one NDA.
The generic ingredient in NORVASC is amlodipine besylate. There are fifty drug master file entries for this compound. Forty-five suppliers are listed for this compound. Additional details are available on the amlodipine besylate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Norvasc
A generic version of NORVASC was approved as amlodipine besylate by COREPHARMA on May 23rd, 2007.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for NORVASC?
- What are the global sales for NORVASC?
- What is Average Wholesale Price for NORVASC?
Summary for NORVASC
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 4 |
| Raw Ingredient (Bulk) Api Vendors: | 132 |
| Clinical Trials: | 58 |
| Patent Applications: | 4,227 |
| Drug Prices: | Drug price information for NORVASC |
| What excipients (inactive ingredients) are in NORVASC? | NORVASC excipients list |
| DailyMed Link: | NORVASC at DailyMed |
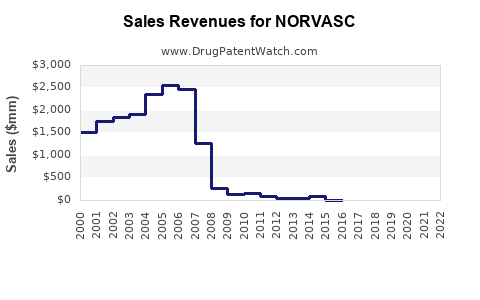
Recent Clinical Trials for NORVASC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Accutest Research Laboratories (I) Pvt. Ltd. | Phase 1 |
| Brillian Pharma Inc. | Phase 1 |
| Overseas Pharmaceuticals, Ltd. | Phase 1 |
Pharmacology for NORVASC
| Drug Class | Calcium Channel Blocker Dihydropyridine Calcium Channel Blocker |
| Mechanism of Action | Calcium Channel Antagonists Cytochrome P450 3A Inhibitors |
US Patents and Regulatory Information for NORVASC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-001 | Jul 31, 1992 | AB | RX | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-002 | Jul 31, 1992 | AB | RX | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-003 | Jul 31, 1992 | AB | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for NORVASC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-002 | Jul 31, 1992 | 4,572,909*PED | ⤷ Try for Free |
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-002 | Jul 31, 1992 | 4,879,303*PED | ⤷ Try for Free |
| Viatris | NORVASC | amlodipine besylate | TABLET;ORAL | 019787-001 | Jul 31, 1992 | 4,572,909*PED | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for NORVASC
See the table below for patents covering NORVASC around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 1023800 | ⤷ Try for Free | |
| European Patent Office | 0089167 | DIHYDROPYRIDINE ANTI-ISCHAEMIC AND ANTIHYPERTENSIVE AGENTS, PROCESSES FOR THEIR PRODUCTION, AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | ⤷ Try for Free |
| Kenya | 3778 | DIHYDROPYRIDINE ANTI-ISCHAEMIC AND ANTIHYPERTENSIVE AGENTS,PROCESSES FOR THEIR PRODUCTION,AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for NORVASC
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0443983 | 2007C/043 | Belgium | ⤷ Try for Free | PRODUCT NAME: AMLODIPINE ET VALSARTAN; NATL. REGISTRATION NO/DATE: EU/1/06/370/001 20070118; FIRST REGISTRATION: CH 57771 20061222 |
| 0502314 | C300478 | Netherlands | ⤷ Try for Free | PRODUCT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN AMLODIPINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER AMLODIPINEBESILAAT; REGISTRATION NO/DATE: EU/1/10/648/001-028 20101007 |
| 0678503 | C300499 | Netherlands | ⤷ Try for Free | PRODUCT NAME: COMBINATIE OMVATTEND ALISKIREN OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, EN AMLODIPINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/11/686/001-056 20110114 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of Amlodipine Besylate (NORVASC)
More… ↓


