NUEDEXTA Drug Patent Profile
✉ Email this page to a colleague
When do Nuedexta patents expire, and when can generic versions of Nuedexta launch?
Nuedexta is a drug marketed by Avanir Pharms and is included in one NDA. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has seventy-five patent family members in twenty-one countries.
The generic ingredient in NUEDEXTA is dextromethorphan hydrobromide; quinidine sulfate. There are twenty-three drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the dextromethorphan hydrobromide; quinidine sulfate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Nuedexta
A generic version of NUEDEXTA was approved as dextromethorphan hydrobromide; quinidine sulfate by ACTAVIS ELIZABETH on October 10th, 2017.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for NUEDEXTA?
- What are the global sales for NUEDEXTA?
- What is Average Wholesale Price for NUEDEXTA?
Summary for NUEDEXTA
| International Patents: | 75 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Clinical Trials: | 11 |
| Drug Prices: | Drug price information for NUEDEXTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for NUEDEXTA |
| What excipients (inactive ingredients) are in NUEDEXTA? | NUEDEXTA excipients list |
| DailyMed Link: | NUEDEXTA at DailyMed |
Paragraph IV (Patent) Challenges for NUEDEXTA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| NUEDEXTA | Capsules | dextromethorphan hydrobromide; quinidine sulfate | 20 mg/10 mg | 021879 | 1 | 2011-03-07 |
US Patents and Regulatory Information for NUEDEXTA
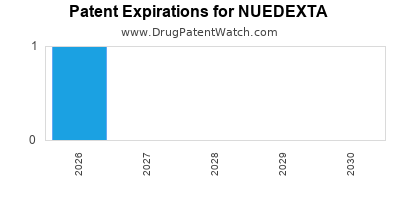
NUEDEXTA is protected by one US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Avanir Pharms | NUEDEXTA | dextromethorphan hydrobromide; quinidine sulfate | CAPSULE;ORAL | 021879-001 | Oct 29, 2010 | AB | RX | Yes | Yes | 7,659,282 | ⤷ Try for Free | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for NUEDEXTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Avanir Pharms | NUEDEXTA | dextromethorphan hydrobromide; quinidine sulfate | CAPSULE;ORAL | 021879-001 | Oct 29, 2010 | 8,227,484 | ⤷ Try for Free |
| Avanir Pharms | NUEDEXTA | dextromethorphan hydrobromide; quinidine sulfate | CAPSULE;ORAL | 021879-001 | Oct 29, 2010 | RE38115 | ⤷ Try for Free |
| Avanir Pharms | NUEDEXTA | dextromethorphan hydrobromide; quinidine sulfate | CAPSULE;ORAL | 021879-001 | Oct 29, 2010 | 5,166,207 | ⤷ Try for Free |
| Avanir Pharms | NUEDEXTA | dextromethorphan hydrobromide; quinidine sulfate | CAPSULE;ORAL | 021879-001 | Oct 29, 2010 | 5,206,248 | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for NUEDEXTA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Jenson Pharmaceutical Services Limited | Nuedexta | dextromethorphan hydrobromide, quinidine sulfate | EMEA/H/C/002560 Nuedexta is indicated for the symptomatic treatment of pseudobulbar affect (PBA) in adults. Efficacy has only been studied in patients with underlying amyotrophic lateral sclerosis or multiple sclerosis. |
Withdrawn | no | no | no | 2013-06-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for NUEDEXTA
See the table below for patents covering NUEDEXTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Australia | 2019200415 | Pharmaceutical compositions comprising dextromethorphan and quinidine for the treatment of neurological disorders | ⤷ Try for Free |
| Japan | 2017036337 | 神経疾患治療のためのデキストロメトルファンおよびキニジンを含む薬剤組成物 (PHARMACEUTICAL COMPOSITIONS COMPRISING DEXTROMETHORPHAN AND QUINIDINE FOR TREATMENT OF NEUROLOGICAL DISORDERS) | ⤷ Try for Free |
| Poland | 374026 | Kompozycje farmaceutyczne zawierające dekstrometorfan i chinidynę do leczenia zaburzeń neurologicznych (PHARMACEUTICAL COMPOSITIONS COMPRISING DEXTROMETHORPHAN AND QUINIDINE FOR THE TREATMENT OF NEUROLOGICAL DISORDERS) | ⤷ Try for Free |
| Taiwan | 200403997 | Pharmaceutical compositions comprising dextromethorphan and quinidine for the treatment of neurological disorders | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for NUEDEXTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1539166 | 122013000090 | Germany | ⤷ Try for Free | PRODUCT NAME: KOMBINATION AUS: (A) DEXTROMETHORPHAN ODER EINEM PHARMAZEUTISCH VERTRAEGLICHEN SALZ, Z.B. DEXTROMETHORPHAN-HYDROBROMID UND INSBESONDERE DEXTROMETHORPHAN-HYDROBROMID-MONOHYDRAT; UND (B) CHINIDIN ODER EINEM PHARMAZEUTISCH VERTRAEGLICHEN SALZ DAVON, Z.B.CHINIDIN-SULFAT UND INSBESONDERE CHINIDIN-SULFAT-DIHYDRAT; REGISTRATION NO/DATE: EU/1/13/833 20130624 |
| 1539166 | 60/2013 | Austria | ⤷ Try for Free | PRODUCT NAME: KOMBINATION VON A) DEXTROMETHORPHAN ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON, BEISPIELSWEISE DEXTROMETHORPHANHYDROBROMID, UND INSBESONDERE DEXTROMETHORPHANHYDROBROMIDMONOHYDRAT UND B) CHINIDIN ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON, BEISPIELSWEISE CHINIDINSULFAT, UND INSBESONDERE CHINIDINSULFATDIHYDRAT.; REGISTRATION NO/DATE: EU/1/13/833 20130626 |
| 1539166 | C300626 | Netherlands | ⤷ Try for Free | PRODUCT NAME: COMBINATIE VAN: (A) DEXTROMETHORFAN, DESGEWENST; REGISTRATION NO/DATE: EU/1/13/833 20130624 |
| 1539166 | SPC/GB13/070 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: THE COMBINATION OF: (A) DEXTROMETHORPHAN OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, E.G. DEXTROMETHORPHAN HYDROBROMIDE AND IN PARTICULAR DEXTROMETHORPHAN HYDROBROMIDE MONOHYDRATE; AND (B) QUINIDINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, E.G. Q; REGISTERED: UK EU/1/13/833 20130626 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
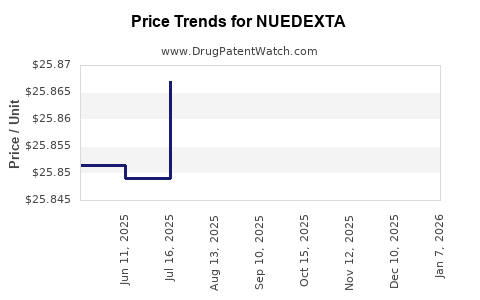
Market Dynamics and Financial Trajectory of Nuedexta
More… ↓


