ORTHO TRI-CYCLEN LO Drug Patent Profile
✉ Email this page to a colleague
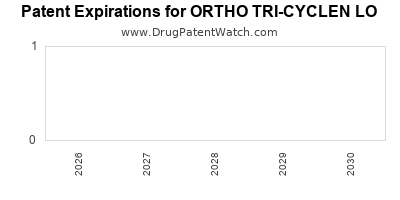
Which patents cover Ortho Tri-cyclen Lo, and when can generic versions of Ortho Tri-cyclen Lo launch?
Ortho Tri-cyclen Lo is a drug marketed by Janssen Pharms and is included in one NDA.
The generic ingredient in ORTHO TRI-CYCLEN LO is ethinyl estradiol; norgestimate. There are twenty-six drug master file entries for this compound. Nineteen suppliers are listed for this compound. Additional details are available on the ethinyl estradiol; norgestimate profile page.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ORTHO TRI-CYCLEN LO?
- What are the global sales for ORTHO TRI-CYCLEN LO?
- What is Average Wholesale Price for ORTHO TRI-CYCLEN LO?
Summary for ORTHO TRI-CYCLEN LO

See drug prices for ORTHO TRI-CYCLEN LO
Recent Clinical Trials for ORTHO TRI-CYCLEN LO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Gilead Sciences | Phase 1 |
| Lexicon Pharmaceuticals | Phase 1 |
| Aegerion Pharmaceuticals, Inc. | Phase 1 |
US Patents and Regulatory Information for ORTHO TRI-CYCLEN LO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | DISCN | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ORTHO TRI-CYCLEN LO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | ⤷ Try for Free | ⤷ Try for Free |
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | ⤷ Try for Free | ⤷ Try for Free |
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | ⤷ Try for Free | ⤷ Try for Free |
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | ⤷ Try for Free | ⤷ Try for Free |
| Janssen Pharms | ORTHO TRI-CYCLEN LO | ethinyl estradiol; norgestimate | TABLET;ORAL-28 | 021241-001 | Aug 22, 2002 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ORTHO TRI-CYCLEN LO
See the table below for patents covering ORTHO TRI-CYCLEN LO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2356747 | CONTRACEPTIF ORAL TRIPHASE (TRIPHASIC ORAL CONTRACEPTIVE) | ⤷ Try for Free |
| Czech Republic | 302568 | Trífázový orální antikoncepcní prostredek (Triphasic oral contraceptive composition) | ⤷ Try for Free |
| New Zealand | 512404 | Triphasic oral contraceptive comprising an estrogen and a progestogen administrated over 21 days | ⤷ Try for Free |
| Norway | 20013144 | ⤷ Try for Free | |
| Taiwan | 589311 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ORTHO TRI-CYCLEN LO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1380301 | 2009C/007 | Belgium | ⤷ Try for Free | PRODUCT NAME: DROSPIRENONE-ETHINYLESTRADIOL; AUTHORISATION NUMBER AND DATE: BE321386 20080811 |
| 1453521 | 300814 | Netherlands | ⤷ Try for Free | PRODUCT NAME: LEVONORGESTREL EN ETHINYLESTRADIOL; NATIONAL REGISTRATION NO/DATE: RVG 117453 20151211; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 1453521 | 39/2015 | Austria | ⤷ Try for Free | PRODUCT NAME: ETHINYLESTRADIOL UND EINE KOMBINATION VON LEVONORGESTREL UND ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: 136021 20150224; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 0771217 | CA 2006 00038 | Denmark | ⤷ Try for Free | PRODUCT NAME: ETHINYLESTRADIOL (SOM BETA-CYCLODEXTRIN-CLATHRAT) OG DROSPIRENON; NAT. REG. NO/DATE: 38687 20060627; FIRST REG. NO/DATE: EU RVG 31781 20050804 |
| 1214076 | SZ 49/2008 | Austria | ⤷ Try for Free | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
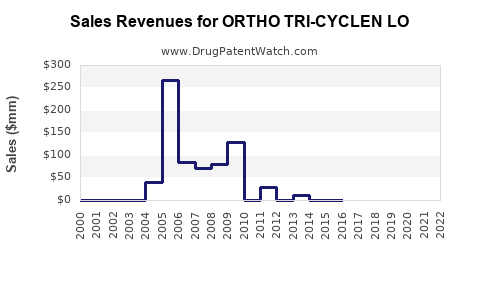
Market Dynamics and Financial Trajectory for Ortho Tri-Cyclen Lo
More… ↓


