PHOSLYRA Drug Patent Profile
✉ Email this page to a colleague
When do Phoslyra patents expire, and what generic alternatives are available?
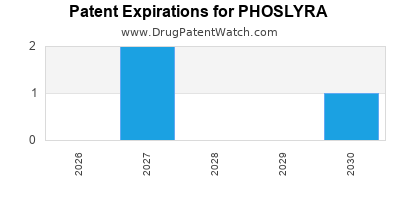
Phoslyra is a drug marketed by Fresenius Medcl and is included in one NDA. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has twenty-three patent family members in sixteen countries.
The generic ingredient in PHOSLYRA is calcium acetate. There are two hundred and eighty-two drug master file entries for this compound. Twenty-one suppliers are listed for this compound. Additional details are available on the calcium acetate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Phoslyra
A generic version of PHOSLYRA was approved as calcium acetate by HIKMA on February 26th, 2008.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for PHOSLYRA?
- What are the global sales for PHOSLYRA?
- What is Average Wholesale Price for PHOSLYRA?
Summary for PHOSLYRA
| International Patents: | 23 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 60 |
| Clinical Trials: | 2 |
| Drug Prices: | Drug price information for PHOSLYRA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PHOSLYRA |
| What excipients (inactive ingredients) are in PHOSLYRA? | PHOSLYRA excipients list |
| DailyMed Link: | PHOSLYRA at DailyMed |

Recent Clinical Trials for PHOSLYRA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Vifor Fresenius Medical Care Renal Pharma | Phase 3 |
| Fresenius Medical Care North America | Phase 4 |
Paragraph IV (Patent) Challenges for PHOSLYRA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PHOSLYRA | Oral Solution | calcium acetate | 667 mg/5 mL | 022581 | 2 | 2013-12-05 |
US Patents and Regulatory Information for PHOSLYRA
PHOSLYRA is protected by three US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresenius Medcl | PHOSLYRA | calcium acetate | SOLUTION;ORAL | 022581-001 | Apr 18, 2011 | DISCN | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Fresenius Medcl | PHOSLYRA | calcium acetate | SOLUTION;ORAL | 022581-001 | Apr 18, 2011 | DISCN | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Fresenius Medcl | PHOSLYRA | calcium acetate | SOLUTION;ORAL | 022581-001 | Apr 18, 2011 | DISCN | Yes | No | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for PHOSLYRA
See the table below for patents covering PHOSLYRA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hong Kong | 1202815 | 醋酸鈣的液體組合物 (LIQUID COMPOSITIONS OF CALCIUM ACETATE) | ⤷ Try for Free |
| New Zealand | 574331 | LIQUID COMPOSITIONS OF CALCIUM ACETATE | ⤷ Try for Free |
| Russian Federation | 2009104774 | ЖИДКАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ (ВАРИАНТЫ) И СПОСОБ СВЯЗЫВАНИЯ ФОСФОРА В ЖЕЛУДОЧНО-КИШЕЧНОМ ТРАКТЕ | ⤷ Try for Free |
| South Africa | 200900472 | Liquid compositions of calcium acetate | ⤷ Try for Free |
| Spain | 2382993 | ⤷ Try for Free | |
| World Intellectual Property Organization (WIPO) | 2008011126 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PHOSLYRA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2365988 | 122018000012 | Germany | ⤷ Try for Free | PRODUCT NAME: PATIROMER SORBITEX CALCIUM UND SALZE DAVON IN ALLEN VOM GRUNDPATENT GESCHUETZTEN FORMEN; REGISTRATION NO/DATE: EU/1/17/1179 20170719 |
| 2957286 | 122018000145 | Germany | ⤷ Try for Free | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTRATION NO/DATE: EU/1/17/1179 20170719 |
| 2957286 | 300962 | Netherlands | ⤷ Try for Free | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTRATION NO/DATE: EU/1/17/1179 20170721 |
| 2957286 | 6/2019 | Austria | ⤷ Try for Free | PRODUCT NAME: PATIROMER UND SEINE CALCIUM-SALZE; REGISTRATION NO/DATE: EU/1/17/1179 (MITTEILUNG) 20170721 |
| 0521471 | C300125 | Netherlands | ⤷ Try for Free | PRODUCT NAME: ROSUVASTATINUM, DESGEWENST IN DE VORM VAN EEN NIET-TOXISCH FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER HET CALCIUMZOUT; REGISTRATION NO/DATE: RVG 26872 - RVG 26874 20021106 |
| 0933372 | PA2008006,C0933372 | Lithuania | ⤷ Try for Free | PRODUCT NAME: FOSAMPRENAVIR CALCIUM; REGISTRATION NO/DATE: EU/1/04/282/001-002 20040712 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
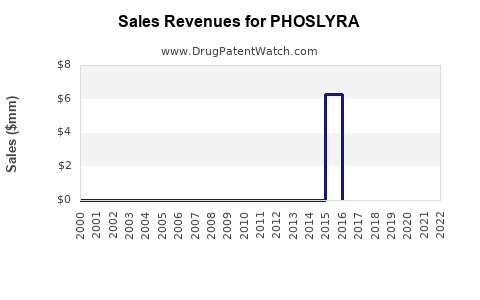
Market Dynamics and Financial Trajectory for PHOSLYRA
More… ↓


