PRADAXA Drug Patent Profile
✉ Email this page to a colleague
When do Pradaxa patents expire, and when can generic versions of Pradaxa launch?
Pradaxa is a drug marketed by Boehringer Ingelheim and is included in two NDAs. There are two patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and twenty patent family members in forty-four countries.
The generic ingredient in PRADAXA is dabigatran etexilate mesylate. There are twenty-six drug master file entries for this compound. Fourteen suppliers are listed for this compound. Additional details are available on the dabigatran etexilate mesylate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Pradaxa
A generic version of PRADAXA was approved as dabigatran etexilate mesylate by ALKEM LABS LTD on March 11th, 2020.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for PRADAXA?
- What are the global sales for PRADAXA?
- What is Average Wholesale Price for PRADAXA?
Summary for PRADAXA
| International Patents: | 120 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 3 |
| Raw Ingredient (Bulk) Api Vendors: | 12 |
| Clinical Trials: | 55 |
| Drug Prices: | Drug price information for PRADAXA |
| Drug Sales Revenues: | Drug sales revenues for PRADAXA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PRADAXA |
| What excipients (inactive ingredients) are in PRADAXA? | PRADAXA excipients list |
| DailyMed Link: | PRADAXA at DailyMed |
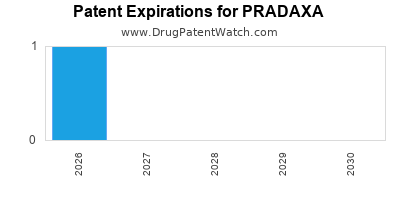
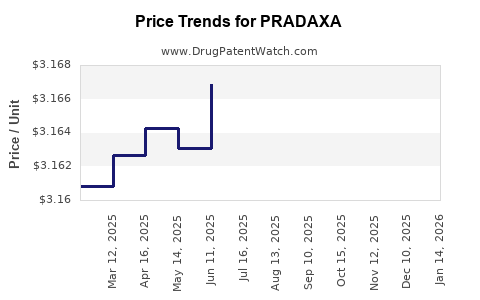
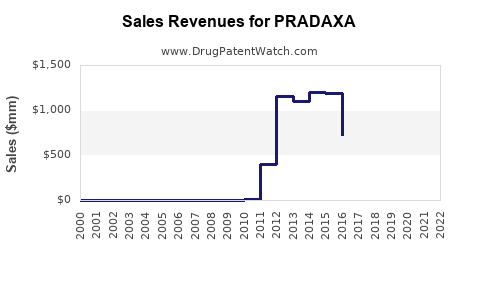
Recent Clinical Trials for PRADAXA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Pfizer | Phase 1 |
| Laval University | Phase 2 |
| Bayer | Phase 1 |
Pharmacology for PRADAXA
| Drug Class | Direct Thrombin Inhibitor |
| Mechanism of Action | Thrombin Inhibitors |
Paragraph IV (Patent) Challenges for PRADAXA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PRADAXA | Capsules | dabigatran etexilate mesylate | eq. to 110 mg base | 022512 | 2 | 2015-12-15 |
| PRADAXA | Capsules | dabigatran etexilate mesylate | eq. to 75 mg base and 150 mg base | 022512 | 17 | 2014-10-20 |
US Patents and Regulatory Information for PRADAXA
PRADAXA is protected by two US patents.
International Patents for PRADAXA
See the table below for patents covering PRADAXA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 1485094 | FORME POSOLOGIQUE POUR ADMINISTRATION PAR VOIE ORALE DE L'ETHYLESTER D'ACIDE 3-[(2-{[4-(HEXYLOXYCARBONYLAMINO-IMINO-METHYL)-PHENYLAMINO]-METHYL}-1-METHYL-1H-BENZIMIDAZOL-5-CARBONYL)-PYRIDIN-2-YL-AMINO] PROPIONIQUE OU SES SELS (DOSAGE FORM FOR ORAL ADMINISTRATION OF 3-[(2-{[4-(HEXYLOXYCARBONYLAMINO-IMINO-METHYL)-PHENYLAMINO]-METHYL}-1-METHYL-1H-BENZIMIDAZOL-5-CARBONYL)-PYRIDIN-2-YL-AMINO] PROPIONIC ACID ETHYL ESTER OR ITS SALTS) | ⤷ Try for Free |
| Luxembourg | 91437 | ⤷ Try for Free | |
| New Zealand | 535663 | Administration form for the oral application of 3-[(2-{[4-(hexyloxycarbonylamino-imino-methyl)-phenylamino]-methyl}-1-methyl-1H-benzimidazol-5-carbonyl)-pyridin-2-yl-amino] propionic acid ethyl ester and the salts thereof | ⤷ Try for Free |
| Norway | 20130778 | ⤷ Try for Free | |
| Portugal | 1485094 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PRADAXA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1485094 | 132012902112338 | Italy | ⤷ Try for Free | PRODUCT NAME: DABIGATRAN ETEXILATO MESILATO(PRADAXA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/08/442/009-011-012-013-014, 20110801 |
| 1485094 | 92117 | Luxembourg | ⤷ Try for Free | |
| 2525812 | C201730027 | Spain | ⤷ Try for Free | PRODUCT NAME: IDARUCIZUMAB; NATIONAL AUTHORISATION NUMBER: EU/1/15/1056; DATE OF AUTHORISATION: 20151120; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/15/1056; DATE OF FIRST AUTHORISATION IN EEA: 20151120 |
| 2525812 | CR 2017 00031 | Denmark | ⤷ Try for Free | PRODUCT NAME: IDARUCIZUMAB; REG. NO/DATE: EU/1/15/1056 20151124 |
| 2525812 | PA2017021,C2525812 | Lithuania | ⤷ Try for Free | PRODUCT NAME: IDARUCIZUMABAS; REGISTRATION NO/DATE: EU/1/15/1056/001 20151120 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of PRADAXA
More… ↓


