PREDNISONE Drug Patent Profile
✉ Email this page to a colleague
When do Prednisone patents expire, and what generic alternatives are available?
Prednisone is a drug marketed by Hikma, Xttrium Labs Inc, Actavis Labs Fl Inc, Aiping Pharm Inc, Allied, Am Therap, Amneal, Amneal Pharms Ny, Aurobindo Pharma Ltd, Bundy, Chartwell Molecular, Chartwell Rx, Contract Pharmacal, Duramed Pharms Barr, Elkins Sinn, Endo Operations, Everylife, Ferrante, Geneyork Pharms, Halsey, Heather, Hikma Pharms, Impax Labs, Inwood Labs, Ivax Sub Teva Pharms, Jubilant Cadista, Kv Pharm, Lederle, Marshall Pharma, Mutual Pharm, Mylan, Novitium Pharma, Nylos, Panray, Pharmavite, Phoenix Labs Ny, Purepac Pharm, Pvt Form, Rexall, Rising, Roxane, Scherer Labs, Sperti, Strides Pharma, Sun Pharm Industries, Superpharm, Teva, UDL, Upsher Smith, Valeant Pharm Intl, Vangard, Vitarine, Watson Labs, and Whiteworth Town Plsn. and is included in one hundred and sixteen NDAs.
The generic ingredient in PREDNISONE is prednisone. There are sixteen drug master file entries for this compound. Forty-nine suppliers are listed for this compound. Additional details are available on the prednisone profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Prednisone
A generic version of PREDNISONE was approved as prednisone by WATSON LABS on December 31st, 1969.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for PREDNISONE?
- What are the global sales for PREDNISONE?
- What is Average Wholesale Price for PREDNISONE?
Summary for PREDNISONE
| US Patents: | 0 |
| Applicants: | 54 |
| NDAs: | 116 |
| Finished Product Suppliers / Packagers: | 48 |
| Raw Ingredient (Bulk) Api Vendors: | 90 |
| Clinical Trials: | 2,021 |
| Patent Applications: | 4,491 |
| Drug Prices: | Drug price information for PREDNISONE |
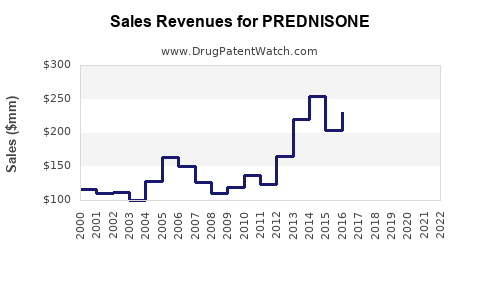
| Drug Sales Revenues: | Drug sales revenues for PREDNISONE |
| DailyMed Link: | PREDNISONE at DailyMed |
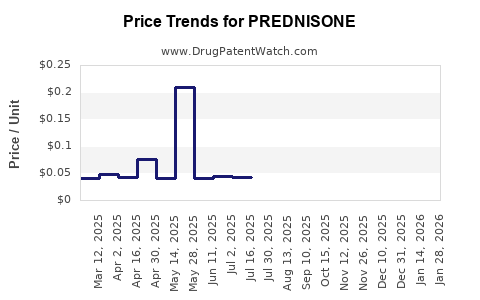
See drug prices for PREDNISONE
Recent Clinical Trials for PREDNISONE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Zhejiang University | Phase 3 |
| Merck Sharp & Dohme LLC | Phase 3 |
| Orion Corporation, Orion Pharma | Phase 3 |
Pharmacology for PREDNISONE
| Drug Class | Corticosteroid |
| Mechanism of Action | Corticosteroid Hormone Receptor Agonists |
Medical Subject Heading (MeSH) Categories for PREDNISONE
Anatomical Therapeutic Chemical (ATC) Classes for PREDNISONE
Paragraph IV (Patent) Challenges for PREDNISONE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| RAYOS | Delayed-release Tablets | prednisone | 5 mg | 202020 | 1 | 2012-11-26 |
US Patents and Regulatory Information for PREDNISONE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mylan | PREDNISONE | prednisone | TABLET;ORAL | 080292-001 | Approved Prior to Jan 1, 1982 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Watson Labs | PREDNISONE | prednisone | TABLET;ORAL | 080356-001 | Approved Prior to Jan 1, 1982 | AB | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Sperti | PREDNISONE | prednisone | TABLET;ORAL | 080359-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Phoenix Labs Ny | PREDNISONE | prednisone | TABLET;ORAL | 083807-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Ivax Sub Teva Pharms | PREDNISONE | prednisone | TABLET;ORAL | 084133-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Everylife | PREDNISONE | prednisone | TABLET;ORAL | 084440-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Duramed Pharms Barr | PREDNISONE | prednisone | TABLET;ORAL | 088395-001 | Oct 4, 1983 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Prednisone
More… ↓


