Introduction
Prempro, a combination hormone therapy drug consisting of conjugated estrogens and medroxyprogesterone acetate, was once a blockbuster medication for menopausal symptoms. However, its market dynamics and financial trajectory have been significantly influenced by several key events, particularly the findings of the Women’s Health Initiative (WHI) study.
Rise to Prominence
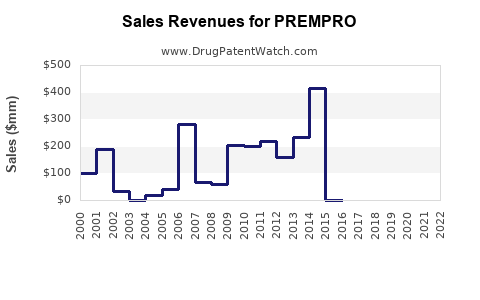
Before the WHI study, Prempro, along with its sister drug Premarin, achieved widespread acceptance and generated over $2 billion in annual sales. Wyeth, the pharmaceutical company behind Prempro, aggressively marketed the drug, touting its benefits for various menopausal symptoms such as hot flashes, osteoporosis, and skin elasticity. The drug was heavily promoted through various channels, including direct-to-consumer advertising, free samples, and office-based detailing[4].
Impact of the Women’s Health Initiative (WHI) Study
The publication of the WHI study in July 2002 marked a significant turning point for Prempro. The study revealed that Prempro increased the risk of breast cancer, cardiovascular disease, thromboembolic events, and other health issues. This led to a rapid decline in prescriptions and promotional spending.
- Prescription Decline: Within nine months of the report’s publication, hormone therapy prescriptions decreased by 32%. By the fourth quarter of 2003, the use of Prempro had declined by 80%, from 4.4 million prescriptions in Q2 of 2002 to just 0.9 million prescriptions[1].
- Promotional Spending: There was a substantial reduction in promotional activities, with a 100% decrease in direct-to-consumer advertising and a 61% decrease in promotional spending for standard-dose Prempro by the first quarter of 2003[1].
Financial Consequences
The decline in prescriptions and promotional spending had significant financial implications for Wyeth and later Pfizer, which acquired Wyeth.
- Revenue Drop: The sales of Prempro and other hormone therapies plummeted following the WHI report. This led to a substantial decline in revenue for Wyeth and subsequently Pfizer[4].
- Legal and Settlement Costs: Over 10,000 women who developed estrogen-dependent breast cancers after taking Prempro filed lawsuits. These legal battles resulted in significant costs and settlements, further impacting the financial trajectory of the drug[4].
Shift to Lower-Dose Formulations
In response to the decline, pharmaceutical companies began promoting lower-dose formulations of Prempro and Premarin.
- New Promotional Strategies: By the latter part of 2003, promotional spending shifted towards lower-dose formulations, which saw modest increases in prescriptions. This strategy included new promotional expenditures of $13.5 million by the fourth quarter of 2003 for these lower-dose formulations[1].
- Market Adaptation: The introduction of lower-dose formulations was part of a broader strategy to adapt to the changing market landscape and mitigate the negative impact of the WHI study findings[1].
Current Market Status
While Prempro is no longer the blockbuster it once was, it still maintains a presence in the market, albeit with significantly reduced sales and a more cautious approach to promotion.
- Regulatory and Public Perception: The drug's reputation has been irreparably damaged by the WHI study findings, leading to stricter regulatory oversight and a more skeptical public perception[4].
- Competition and Alternatives: The market has seen the introduction of new treatments for menopausal symptoms, such as Imvexxy and Annovera, which have further eroded Prempro's market share[3].
Financial Performance of Pfizer
Pfizer, the current owner of Prempro, has seen significant fluctuations in its financial performance, partly due to the decline of Prempro and other factors such as the impact of COVID-19 products.
- Revenue Trends: Pfizer's revenues have been influenced by the decline of Prempro and other legacy products, as well as the rise and fall of COVID-19 related products like Comirnaty and Paxlovid. For example, in 2023, Pfizer reported a 42% decline in revenues compared to 2022, largely due to the decrease in sales of these COVID-19 products[2][5].
Key Takeaways
- The WHI study significantly impacted the market dynamics and financial trajectory of Prempro.
- The drug saw a rapid decline in prescriptions and promotional spending following the study's publication.
- Pharmaceutical companies adapted by promoting lower-dose formulations.
- The financial performance of Pfizer has been influenced by the decline of Prempro and other factors.
- The market has become more competitive with the introduction of new treatments for menopausal symptoms.
FAQs
What was the impact of the WHI study on Prempro prescriptions?
The WHI study led to a 32% decrease in hormone therapy prescriptions within nine months of its publication, with Prempro prescriptions declining by 80% by the fourth quarter of 2003[1].
How did the WHI study affect promotional spending for Prempro?
The study resulted in a substantial reduction in promotional activities, including a 100% decrease in direct-to-consumer advertising and a 61% decrease in promotional spending for standard-dose Prempro[1].
What was the financial impact on Wyeth and Pfizer due to the decline of Prempro?
The decline led to significant revenue drops and legal costs associated with lawsuits filed by women who developed breast cancer after taking Prempro[4].
How have pharmaceutical companies adapted to the decline of Prempro?
Companies have shifted their focus to promoting lower-dose formulations of Prempro and Premarin, which have seen modest increases in prescriptions[1].
What is the current market status of Prempro?
Prempro still exists in the market but with significantly reduced sales and a more cautious approach to promotion, facing competition from new treatments for menopausal symptoms[3].
How has Pfizer's financial performance been affected by the decline of Prempro?
Pfizer's revenues have been influenced by the decline of Prempro, as well as the impact of COVID-19 related products. The company reported a 42% decline in revenues in 2023 compared to 2022[2][5].
Sources
- Promotion and Prescribing of Hormone Therapy After Report of the Women’s Health Initiative (WHI) Study - JAMA
- Pfizer Reports Full-Year 2023 Results and Reaffirms Full-Year 2024 Guidance - Pfizer
- Oppenheimer NDR_TXMD Final 3 - TherapeuticsMD Inc
- Prempro - Hormone Replacement Therapy (HRT) - Hausfeld
- PFIZER REPORTS RECORD FULL-YEAR 2022 RESULTS AND PROVIDES FULL-YEAR 2023 GUIDANCE - Pfizer