RYALTRIS Drug Patent Profile
✉ Email this page to a colleague
When do Ryaltris patents expire, and when can generic versions of Ryaltris launch?
Ryaltris is a drug marketed by Glenmark Speclt and is included in one NDA. There are fifteen patents protecting this drug.
This drug has ninety-two patent family members in thirty-four countries.
The generic ingredient in RYALTRIS is mometasone furoate; olopatadine hydrochloride. There are thirty drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the mometasone furoate; olopatadine hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Ryaltris
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for RYALTRIS?
- What are the global sales for RYALTRIS?
- What is Average Wholesale Price for RYALTRIS?
Summary for RYALTRIS
| International Patents: | 92 |
| US Patents: | 15 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
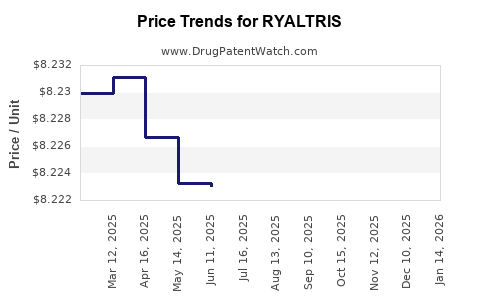
| Drug Prices: | Drug price information for RYALTRIS |
| What excipients (inactive ingredients) are in RYALTRIS? | RYALTRIS excipients list |
| DailyMed Link: | RYALTRIS at DailyMed |
Pharmacology for RYALTRIS
| Drug Class | Corticosteroid Histamine-1 Receptor Antagonist Histamine-1 Receptor Inhibitor Mast Cell Stabilizer |
| Mechanism of Action | Corticosteroid Hormone Receptor Agonists Histamine H1 Receptor Antagonists |
| Physiological Effect | Decreased Histamine Release |
US Patents and Regulatory Information for RYALTRIS
RYALTRIS is protected by fifteen US patents and one FDA Regulatory Exclusivity.
International Patents for RYALTRIS
See the table below for patents covering RYALTRIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Brazil | 112020016817 | método para tratar sintomas associados à rinite alérgica, método para fornecer um início de ação mais rápido para alívio da rinite alérgica e método para tratar um sujeito humano pediátrico que sofre de rinite alérgica | ⤷ Try for Free |
| China | 109475495 | ⤷ Try for Free | |
| China | 115444851 | 用于治疗鼻炎的分配装置和药物组合物 (Dispensing device and pharmaceutical composition for treating rhinitis) | ⤷ Try for Free |
| Hungary | E056448 | ⤷ Try for Free | |
| Japan | 2021514947 | ⤷ Try for Free | |
| Poland | 3043773 | ⤷ Try for Free | |
| Ukraine | 120587 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for RYALTRIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3043773 | 122021000085 | Germany | ⤷ Try for Free | PRODUCT NAME: MOMETASON ODER EIN SALZ DAVON UND OLOPATADIN ODER EIN SALZ DAVON; NAT. REGISTRATION NO/DATE: 2205824.00.00 20211117; FIRST REGISTRATION: OESTERREICH 140638 20210426 |
| 3043773 | 132021000000191 | Italy | ⤷ Try for Free | PRODUCT NAME: MOMETASONE O UN SUO SALE E OLOPATADINA O UN SUO SALE(RYALTRIS); AUTHORISATION NUMBER(S) AND DATE(S): 048499014, 048499026, 048499038, 20210726;140638, 20210426 |
| 3043773 | 2021045 | Norway | ⤷ Try for Free | PRODUCT NAME: MOMETASON ELLER ET SALT DERAV OG OLOPATADIN ELLER ET SALT DERAV; NAT. REG. NO/DATE: 20-13278 20210709; FIRST REG. NO/DATE: 140638 20210426 |
| 3043773 | 2190041-0 | Sweden | ⤷ Try for Free | PRODUCT NAME: MOMETASONE OR A SALT THEREOF AND OLOPATADINE OR A SALT THEREOF; NAT. REG. NO/DATE: MT NR 60226 20210519; FIRST REG.: AT APPROVAL NR 140638 20210426 |
| 3043773 | 57/2021 | Austria | ⤷ Try for Free | PRODUCT NAME: MOMETASON ODER EIN SALZ DAVON UND OLOPATADIN ODER EIN SALZ DAVON; REGISTRATION NO/DATE: 140638 20210426 |
| 0112669 | 96C0002 | Belgium | ⤷ Try for Free | PRODUCT NAME: MOMETASONE FUROATE; NAT. REGISTRATION NO/DATE: NL 19601 19950919; FIRST REGISTRATION: GR - 10681/89 19900622 |
| 3043773 | SPC/GB21/077 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: MOMETASONE OR A SALT THEREOF AND OLOPATADINE OR A SALT THEREOF; REGISTERED: AT 140638 20210426; UK PL 25258/0331 - 0001 20210511 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for RYALTRIS
More… ↓


