Introduction
Triazolam, a small molecule drug developed by Pfizer Inc., has been a significant player in the pharmaceutical industry since its approval in 1982. This article delves into the market dynamics and financial trajectory of Triazolam, highlighting its mechanism of action, therapeutic applications, market trends, and financial performance.
Mechanism of Action and Therapeutic Applications
Triazolam targets the GABAA receptor, enhancing the effects of gamma-aminobutyric acid (GABA), a neurotransmitter that inhibits brain activity. This mechanism allows Triazolam to induce sedation and promote sleep, making it effective in treating sleep initiation and maintenance disorders, as well as serving as an anesthetic[1][3].
Approval and Global Recognition
Triazolam has received approval in multiple countries, including the United States, since its initial approval in November 1982. This widespread recognition underscores its safety and efficacy as a medication[1].
Market Demand and Growth
The demand for Triazolam is driven by the increasing prevalence of sleep disorders and anxiety-related conditions. The global benzodiazepine drugs market, which includes Triazolam, is projected to grow at a CAGR of 2.8% to reach US$ 3.1 billion by 2032. This growth is fueled by rising cases of anxiety-related disorders and the impact of urbanization and work pressure on mental health[5].
Short-Term Outlook (2022-2025)
In the short term, the market for Triazolam is expected to benefit from the rising use of benzodiazepine drugs for mental disorders. The immediate need for effective treatments for insomnia and anxiety will drive the demand for Triazolam[5].
Medium-Term Outlook (2025-2028)
During the medium term, product development focusing on minimizing side effects will be crucial. As manufacturers develop benzodiazepines with improved safety profiles, the acceptance and use of Triazolam are likely to increase[5].
Long-Term Outlook (2028-2032)
Long-term growth will be driven by rising urbanization and the increasing pressure of modern work culture, which contribute to higher rates of mental health issues. This trend is expected to sustain the demand for Triazolam and other benzodiazepine drugs[5].
Financial Performance
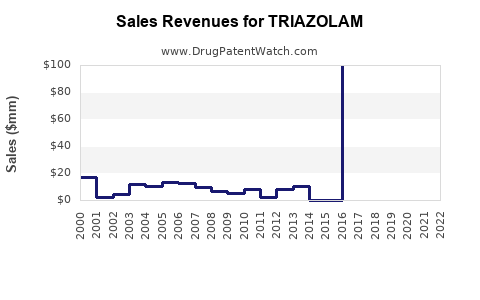
While specific financial data for Triazolam alone is not readily available, the broader context of the benzodiazepine market provides insights into its financial trajectory. The global benzodiazepine market's projected growth indicates a stable and increasing revenue stream for drugs like Triazolam.
Revenue Projections
The global benzodiazepine drugs market is expected to reach US$ 3.1 billion by 2032. This growth will be influenced by the increasing demand for effective treatments for sleep disorders and anxiety, which are key indications for Triazolam[5].
Competitive Landscape
Triazolam operates within a competitive landscape that includes other benzodiazepines and non-benzodiazepine hypnotics. However, its short half-life and minimal daytime residual effects make it a preferred option for many patients, contributing to its financial stability[3].
Clinical and Research Aspects
Triazolam has been extensively studied in both sleep laboratory and non-laboratory clinical trials. These studies have shown that Triazolam is effective in inducing and maintaining sleep, with a duration of efficacy up to six months in non-laboratory settings. Its short half-life of 2-5 hours reduces the risk of daytime residual effects, making it a safe and effective treatment option[3].
Regulatory and Approval Status
Triazolam's approval in multiple countries, including the United States, reflects its compliance with stringent regulatory standards. This widespread approval is a testament to its safety and efficacy profile, which is crucial for maintaining a strong market presence[1].
Side Effects and Safety Profile
While Triazolam is generally safe and effective, it can have side effects such as drowsiness, dizziness, and impaired coordination. These side effects are common among GABAA receptor agonists and highlight the need for medical supervision during its use[1].
Conclusion
Triazolam's market dynamics are characterized by a strong demand driven by the increasing prevalence of sleep and anxiety disorders. Its financial trajectory is expected to remain positive, aligned with the growth of the global benzodiazepine market. The drug's unique pharmacological profile, including its short half-life and minimal residual effects, positions it as a valuable treatment option in the pharmaceutical industry.
Key Takeaways
- Mechanism of Action: Triazolam targets the GABAA receptor, enhancing GABA's inhibitory effects.
- Therapeutic Applications: Used for sleep initiation and maintenance disorders, and as an anesthetic.
- Market Growth: Part of the benzodiazepine market projected to grow at a CAGR of 2.8% to US$ 3.1 billion by 2032.
- Financial Performance: Expected to benefit from rising demand for benzodiazepines.
- Clinical Efficacy: Effective in inducing and maintaining sleep with minimal daytime residual effects.
- Regulatory Status: Approved in multiple countries, including the United States.
FAQs
What is the primary mechanism of action of Triazolam?
Triazolam targets the GABAA receptor, enhancing the effects of gamma-aminobutyric acid (GABA), which inhibits brain activity[1].
What are the common therapeutic applications of Triazolam?
Triazolam is primarily used for the treatment of sleep initiation and maintenance disorders and as an anesthetic[1][3].
How does the market for Triazolam project to grow in the coming years?
The market for Triazolam is expected to grow as part of the broader benzodiazepine market, projected to reach US$ 3.1 billion by 2032 with a CAGR of 2.8%[5].
What are the key side effects of Triazolam?
Common side effects include drowsiness, dizziness, and impaired coordination, which are typical for GABAA receptor agonists[1].
Why is Triazolam preferred over other benzodiazepines in some cases?
Triazolam's short half-life and minimal daytime residual effects make it a preferred option for many patients, reducing the risk of prolonged sedation[3].
Is Triazolam approved for use in multiple countries?
Yes, Triazolam has received approval in multiple countries, including the United States, since its initial approval in November 1982[1].