VALTOCO Drug Patent Profile
✉ Email this page to a colleague
When do Valtoco patents expire, and what generic alternatives are available?
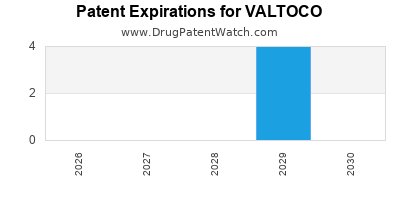
Valtoco is a drug marketed by Neurelis Inc and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has ninety-eight patent family members in twenty-eight countries.
The generic ingredient in VALTOCO is diazepam. There are eight drug master file entries for this compound. Forty-five suppliers are listed for this compound. Additional details are available on the diazepam profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Valtoco
A generic version of VALTOCO was approved as diazepam by MYLAN on September 4th, 1985.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for VALTOCO?
- What are the global sales for VALTOCO?
- What is Average Wholesale Price for VALTOCO?
Summary for VALTOCO
| International Patents: | 98 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 68 |
| Clinical Trials: | 2 |
| Patent Applications: | 3,894 |
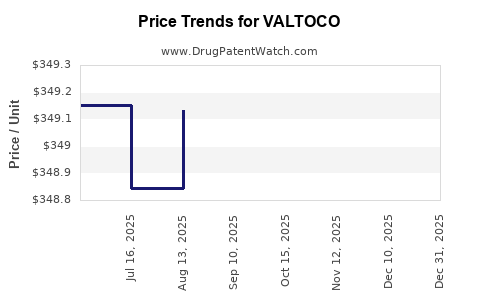
| Drug Prices: | Drug price information for VALTOCO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VALTOCO |
| What excipients (inactive ingredients) are in VALTOCO? | VALTOCO excipients list |
| DailyMed Link: | VALTOCO at DailyMed |
Recent Clinical Trials for VALTOCO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Cincinnati | Phase 4 |
| Neurelis, Inc. | Phase 1/Phase 2 |
Pharmacology for VALTOCO
| Drug Class | Benzodiazepine |
US Patents and Regulatory Information for VALTOCO
VALTOCO is protected by six US patents and one FDA Regulatory Exclusivity.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-001 | Jan 10, 2020 | RX | Yes | No | 8,927,497 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-001 | Jan 10, 2020 | RX | Yes | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-001 | Jan 10, 2020 | RX | Yes | No | 8,895,546 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-002 | Jan 10, 2020 | RX | Yes | No | 11,241,414 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-002 | Jan 10, 2020 | RX | Yes | No | 8,927,497 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-003 | Jan 10, 2020 | RX | Yes | Yes | 8,895,546 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Neurelis Inc | VALTOCO | diazepam | SPRAY;NASAL | 211635-003 | Jan 10, 2020 | RX | Yes | Yes | 10,265,402 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for VALTOCO
See the table below for patents covering VALTOCO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2756690 | ADMINISTRATION DE COMPOSITIONS DE BENZODIAZEPINE (ADMINISTRATION OF BENZODIAZEPINE COMPOSITIONS) | ⤷ Try for Free |
| European Patent Office | 3415139 | ADMINISTRATION DE BENZODIAZÉPINE (ADMINISTRATION OF BENZODIAZEPINE) | ⤷ Try for Free |
| Germany | 112019000683 | ⤷ Try for Free | |
| Hong Kong | 1250647 | 苯二氮卓組合物的投與 (ADMINISTRATION OF BENZODIAZEPINE COMPOSITIONS) | ⤷ Try for Free |
| Hungary | E060917 | ⤷ Try for Free | |
| Japan | 2021193091 | 疾患の処置のための鼻腔内エピネフリン製剤及び方法 (INTRANASAL EPINEPHRINE PREPARATIONS AND METHODS FOR TREATMENT OF DISEASE) | ⤷ Try for Free |
| World Intellectual Property Organization (WIPO) | 2012054500 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VALTOCO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3678649 | 122025000010 | Germany | ⤷ Try for Free | PRODUCT NAME: KOMBINATION AUS EPINEPHRIN AND DODECYLMALTOSID, IN ALLEN DEM SCHUTZ DES GRUNDPATENTS UNTERLIEGENDEN FORMEN; REGISTRATION NO/DATE: EU/1/24/1846 20240822 |
| 3678649 | 301317 | Netherlands | ⤷ Try for Free | PRODUCT NAME: COMBINATIE VAN EPINEFRINE OF EEN ZOUT DAARVAN, EN DODECYLMALTOSIDE; REGISTRATION NO/DATE: EU1/24/1846 20240823 |
| 3678649 | C20250011 | Finland | ⤷ Try for Free | |
| 3678649 | CA 2025 00007 | Denmark | ⤷ Try for Free | PRODUCT NAME: KOMBINATION AF EPINEPHRIN ELLER ET SALT DERAF, OG DODECYLMATOSID; REG. NO/DATE: EU/1/24/1846 20240823 |
| 3678649 | PA2025507 | Lithuania | ⤷ Try for Free | PRODUCT NAME: VISU FORMU EPINEFRINO IR DODECILMALTOZIDO DERINYS SAUGOMASPAGRINDINIO PATENTO; REGISTRATION NO/DATE: EU/1/24/1846 20240822 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of VALTOCO
More… ↓


