VEKLURY Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Veklury, and what generic alternatives are available?
Veklury is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are sixteen patents protecting this drug.
This drug has three hundred and twenty-nine patent family members in forty-eight countries.
The generic ingredient in VEKLURY is remdesivir. One supplier is listed for this compound. Additional details are available on the remdesivir profile page.
DrugPatentWatch® Generic Entry Outlook for Veklury
Veklury was eligible for patent challenges on October 22, 2024.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 29, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for VEKLURY?
- What are the global sales for VEKLURY?
- What is Average Wholesale Price for VEKLURY?
Summary for VEKLURY
| International Patents: | 329 |
| US Patents: | 16 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 34 |
| Clinical Trials: | 6 |
| Patent Applications: | 2,734 |
| What excipients (inactive ingredients) are in VEKLURY? | VEKLURY excipients list |
| DailyMed Link: | VEKLURY at DailyMed |
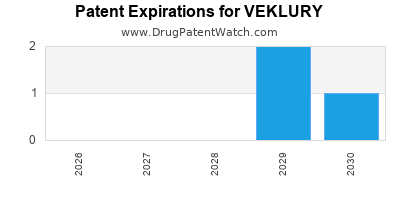
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VEKLURY
Generic Entry Date for VEKLURY*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INTRAVENOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VEKLURY
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Peninsula Clinical Trials Unit | Phase 4 |
| University of Exeter | Phase 4 |
| University Hospitals of Derby and Burton NHS Foundation Trust | Phase 4 |
Pharmacology for VEKLURY
US Patents and Regulatory Information for VEKLURY
VEKLURY is protected by sixteen US patents and eight FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VEKLURY is ⤷ Try for Free.
This potential generic entry date is based on patent ⤷ Try for Free.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VEKLURY | remdesivir | POWDER;INTRAVENOUS | 214787-001 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Gilead Sciences Inc | VEKLURY | remdesivir | SOLUTION;INTRAVENOUS | 214787-002 | Oct 22, 2020 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VEKLURY
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Ireland UC | Veklury | remdesivir | EMEA/H/C/005622 Veklury is indicated for the treatment of coronavirus disease 2019 (COVID 19) in:adults and paediatric patients (at least 4 weeks of age and weighing at least 3 kg) with pneumonia requiring supplemental oxygen (low- or high-flow oxygen or other non-invasive ventilation at start of treatment)adults and paediatric patients (weighing at least 40 kg) who do not require supplemental oxygen and who are at increased risk of progressing to severe COVID-19 |
Authorised | no | no | no | 2020-07-03 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VEKLURY
When does loss-of-exclusivity occur for VEKLURY?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2467
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 2468
Patent: MÉTODOS PARA LA PREPARACIÓN DE RIBÓSIDOS
Estimated Expiration: ⤷ Try for Free
Patent: 9850
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Australia
Patent: 15339222
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Patent: 15339223
Patent: Methods for treating Filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 18253483
Patent: Methods for treating Filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 19201232
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Patent: 20203892
Patent: Methods for treating Filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 21201474
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Patent: 22283772
Patent: Methods for treating Filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 23202679
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Brazil
Patent: 2015027413
Patent: métodos para tratar infecções pelo vírus filoviridae
Estimated Expiration: ⤷ Try for Free
Patent: 2017007636
Patent: métodos para a preparação de ribosídeos
Estimated Expiration: ⤷ Try for Free
Canada
Patent: 63832
Patent: METHODES POUR LE TRAITEMENT D'INFECTIONS VIRALES A FILOVIRIDAE (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 63907
Patent: PROCEDES DE PREPARATION DE RIBOSIDES (METHODS FOR THE PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 84285
Patent: PROCEDES DE PREPARATION DE RIBOSIDES (METHODS FOR THE PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Chile
Patent: 17001040
Patent: Métodos para tratar infecciones por el virus filoviridae
Estimated Expiration: ⤷ Try for Free
Patent: 17002693
Patent: Métodos para tratar infecciones por el virus filoviridae. divisional de solicitud n° 1040-2017.
Estimated Expiration: ⤷ Try for Free
China
Patent: 7073005
Patent: 治疗丝状病毒科病毒感染的方法 (Methods for treating filoviridae virus infections)
Estimated Expiration: ⤷ Try for Free
Patent: 7074902
Patent: 制备核糖核苷的方法 (Methods for the preparation of ribosides)
Estimated Expiration: ⤷ Try for Free
Patent: 3549120
Patent: 制备核糖核苷的方法 (Method for preparing ribonucleoside)
Estimated Expiration: ⤷ Try for Free
Patent: 3620992
Patent: 治疗丝状病毒科病毒感染的方法 (Method for treating filoviridae virus infection)
Estimated Expiration: ⤷ Try for Free
Patent: 4191438
Patent: 治疗丝状病毒科病毒感染的方法 (Methods of treating filoviridae virus infections)
Estimated Expiration: ⤷ Try for Free
Colombia
Patent: 17003960
Patent: Métodos para tratar infecciones por el virus filoviridae
Estimated Expiration: ⤷ Try for Free
Costa Rica
Patent: 170165
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 170483
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE (Divisional 2017-165)
Estimated Expiration: ⤷ Try for Free
Croatia
Patent: 0181130
Estimated Expiration: ⤷ Try for Free
Patent: 0200518
Estimated Expiration: ⤷ Try for Free
Cuba
Patent: 170056
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 170145
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Cyprus
Patent: 20893
Estimated Expiration: ⤷ Try for Free
Patent: 22946
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 12174
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Estimated Expiration: ⤷ Try for Free
Dominican Republic
Patent: 017000103
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Ecuador
Patent: 17025261
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 17072474
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
El Salvador
Patent: 17005424
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 17005561
Patent: METODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Eurasian Patent Organization
Patent: 2239
Patent: СПОСОБЫ ЛЕЧЕНИЯ ВИРУСНЫХ ИНФЕКЦИЙ FILOVIRIDAE (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 9561
Patent: СОЕДИНЕНИЯ ДЛЯ ЛЕЧЕНИЯ ВИРУСНЫХ ИНФЕКЦИЙ FILOVIRIDAE (COMPOUNDS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 1790597
Patent: СПОСОБЫ ЛЕЧЕНИЯ ВИРУСНЫХ ИНФЕКЦИЙ FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 1790630
Patent: СПОСОБЫ ПОЛУЧЕНИЯ РИБОЗИДОВ
Estimated Expiration: ⤷ Try for Free
Patent: 1990021
Patent: СПОСОБЫ ЛЕЧЕНИЯ ВИРУСНЫХ ИНФЕКЦИЙ FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 12174
Patent: MÉTHODES POUR LE TRAITEMENT D'INFECTIONS VIRALES À FILOVIRIDAE (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 12175
Patent: PROCÉDÉS DE PRÉPARATION DE RIBOSIDES (METHODS FOR THE PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Patent: PROCÉDÉS DE TRAITEMENT D'INFECTIONS PAR DES VIRUS FILOVIRIDÉS (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 95844
Patent: PROCÉDÉS DE TRAITEMENT D'INFECTIONS PAR DES VIRUS NIPAH (METHODS FOR TREATING NIPAH VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 36099
Patent: MÉTHODES POUR LA PRÉPARATION DE RIBOSIDES (METHODS FOR THE PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Hong Kong
Patent: 58795
Patent: 絲狀病毒科病毒感染的治療方法 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Hungary
Patent: 39231
Estimated Expiration: ⤷ Try for Free
Patent: 49192
Estimated Expiration: ⤷ Try for Free
Israel
Patent: 1707
Patent: שיטות לטיפול בזיהומי וירוס פילווירידאה (Methods for treating filoviridae virus infections)
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 20484
Estimated Expiration: ⤷ Try for Free
Patent: 87547
Estimated Expiration: ⤷ Try for Free
Patent: 71424
Estimated Expiration: ⤷ Try for Free
Patent: 57294
Estimated Expiration: ⤷ Try for Free
Patent: 58428
Estimated Expiration: ⤷ Try for Free
Patent: 17186358
Patent: フィロウイルス科ウイルス感染症を処置するための方法 (METHOD FOR TREATING FILOVIRIDAE VIRUS INFECTION)
Estimated Expiration: ⤷ Try for Free
Patent: 17533903
Patent: リボシドの調製のための方法
Estimated Expiration: ⤷ Try for Free
Patent: 17534614
Patent: フィロウイルス科ウイルス感染症を処置するための方法
Estimated Expiration: ⤷ Try for Free
Patent: 18172424
Patent: リボシドの調製のための方法 (METHODS FOR PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 19048901
Patent: リボシドの調製のための方法 (METHODS FOR PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 20090536
Patent: フィロウイルス科ウイルス感染症を処置するための方法 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 20097635
Patent: リボシドの調製のための方法 (METHODS FOR PREPARING RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 22065066
Patent: フィロウイルス科ウイルス感染症を処置するための方法
Estimated Expiration: ⤷ Try for Free
Patent: 22068297
Patent: リボシドの調製のための方法
Estimated Expiration: ⤷ Try for Free
Lithuania
Patent: 12174
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Estimated Expiration: ⤷ Try for Free
Malaysia
Patent: 5823
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
Mexico
Patent: 17005250
Patent: METODOS PARA TRATAR INFECCIONES POR EL VIRUS DE FILOVIRIDAE. (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS.)
Estimated Expiration: ⤷ Try for Free
Patent: 17005252
Patent: METODOS PARA LA PREPARACION DE RIBOSIDOS. (METHODS FOR THE PREPARATION OF RIBOSIDES.)
Estimated Expiration: ⤷ Try for Free
Patent: 20012560
Patent: METODOS PARA TRATAR INFECCIONES POR EL VIRUS DE FILOVIRIDAE. (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS.)
Estimated Expiration: ⤷ Try for Free
Moldova, Republic of
Patent: 170046
Patent: Metode pentru tratamentul infecţiilor virale cu Filoviridae (Methods for treating Filoviridae virus infections)
Estimated Expiration: ⤷ Try for Free
Montenegro
Patent: 070
Patent: POSTUPCI LIJEČENJA VIRUSNIH FILOVIRIDAE INFEKCIJA (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Morocco
Patent: 867
Patent: MÉTHODES POUR LE TRAITEMENT D'INFECTIONS VIRALES À FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 872
Patent: MÉTHODES POUR LA PRÉPARATION DE RIBOSIDES
Estimated Expiration: ⤷ Try for Free
Patent: 201
Patent: PROCÉDÉS DE TRAITEMENT D'INFECTIONS PAR DES VIRUS FILOVIRIDÉS
Estimated Expiration: ⤷ Try for Free
Patent: 506
Patent: PROCÉDÉS DE TRAITEMENT D'INFECTIONS PAR DES VIRUS NIPAH
Estimated Expiration: ⤷ Try for Free
New Zealand
Patent: 0803
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Patent: 0809
Patent: Methods for treating filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 5328
Patent: Methods for treating filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Peru
Patent: 171439
Patent: METODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 180202
Patent: METODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Philippines
Patent: 017500631
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
Patent: 020551055
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
Poland
Patent: 12174
Estimated Expiration: ⤷ Try for Free
Patent: 12175
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 12174
Estimated Expiration: ⤷ Try for Free
Patent: 12175
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Estimated Expiration: ⤷ Try for Free
Saudi Arabia
Patent: 7381419
Patent: طرق لعلاج حالات الإصابة بعدوى فيروس من عائلة الفيروسات الخيطية (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Serbia
Patent: 425
Patent: POSTUPCI LEČENJA VIRUSNIH FILOVIRIDAE INFEKCIJA (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Singapore
Patent: 202008772U
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
Patent: 201702903T
Patent: METHODS FOR THE PREPARATION OF RIBOSIDES
Estimated Expiration: ⤷ Try for Free
Patent: 201702904R
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 12174
Estimated Expiration: ⤷ Try for Free
Patent: 12175
Estimated Expiration: ⤷ Try for Free
Patent: 66295
Estimated Expiration: ⤷ Try for Free
South Africa
Patent: 1800414
Patent: METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS
Estimated Expiration: ⤷ Try for Free
South Korea
Patent: 1822348
Estimated Expiration: ⤷ Try for Free
Patent: 2337664
Estimated Expiration: ⤷ Try for Free
Patent: 2453808
Estimated Expiration: ⤷ Try for Free
Patent: 170066665
Patent: 필로비리다에 바이러스 감염을 치료하는 방법 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 170067898
Patent: 필로비리다에 바이러스 감염을 치료하는 방법 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 170077167
Patent: 리보시드의 제조 방법 (METHODS FOR THE PREPARATION OF RIBOSIDES)
Estimated Expiration: ⤷ Try for Free
Patent: 210152015
Patent: 필로비리다에 바이러스 감염을 치료하는 방법 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Patent: 220140656
Patent: 필로비리다에 바이러스 감염을 치료하는 방법 (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 74806
Estimated Expiration: ⤷ Try for Free
Patent: 85034
Estimated Expiration: ⤷ Try for Free
Patent: 04298
Estimated Expiration: ⤷ Try for Free
Taiwan
Patent: 87432
Estimated Expiration: ⤷ Try for Free
Patent: 98444
Estimated Expiration: ⤷ Try for Free
Patent: 40546
Estimated Expiration: ⤷ Try for Free
Patent: 67201
Estimated Expiration: ⤷ Try for Free
Patent: 1629076
Patent: Methods for treating filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 1630925
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Patent: 2039526
Patent: Methods for treating Filoviridae virus infections
Estimated Expiration: ⤷ Try for Free
Patent: 2115098
Patent: Methods for the preparation of ribosides
Estimated Expiration: ⤷ Try for Free
Turkey
Patent: 1809518
Estimated Expiration: ⤷ Try for Free
Ukraine
Patent: 1485
Patent: СПОСОБИ ЛІКУВАННЯ ВІРУСНИХ ІНФЕКЦІЙ FILOVIRIDAE (METHODS FOR TREATING FILOVIRIDAE VIRUS INFECTIONS)
Estimated Expiration: ⤷ Try for Free
Uruguay
Patent: 376
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Patent: 464
Patent: MÉTODOS PARA TRATAR INFECCIONES POR EL VIRUS FILOVIRIDAE
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VEKLURY around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| African Regional IP Organization (ARIPO) | 3076 | Carba-nucleoside analogs for antiviral treatment | ⤷ Try for Free |
| China | 105343098 | ⤷ Try for Free | |
| Denmark | 2268642 | ⤷ Try for Free | |
| Denmark | 3785717 | ⤷ Try for Free | |
| European Patent Office | 4036099 | MÉTHODES POUR LA PRÉPARATION DE RIBOSIDES (METHODS FOR THE PREPARATION OF RIBOSIDES) | ⤷ Try for Free |
| Japan | 5425187 | ⤷ Try for Free | |
| Japan | 2023528810 | レムデシビル治療方法 | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VEKLURY
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2595980 | 122020000084 | Germany | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR ODER PHARMAZEUTISCH AKZEPTABLES SALZ ODER PHARMAZEUTISCH AKZEPTABLER ESTER DAVON; REGISTRATION NO/DATE: EU/1/20/1459 20200703 |
| 2595980 | 2020/061 | Ireland | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR ESTER THEREOF; REGISTRATION NO/DATE: EU/1/20/1459 20200703 |
| 2595980 | 301084 | Netherlands | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF ESTER DAARVAN; REGISTRATION NO/DATE: EU/1/20/1459 20200703 |
| 2595980 | C02595980/01 | Switzerland | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 68043 25.11.2020 |
| 2595980 | C202030068 | Spain | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR O UNA SAL O UN ESTER FARMACEUTICAMENTE ACEPTABLES DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/20/1459; DATE OF AUTHORISATION: 20200703; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1459; DATE OF FIRST AUTHORISATION IN EEA: 20200703 |
| 2937350 | CA 2020 00060 | Denmark | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/20/1459 20200703 |
| 2595980 | SPC/GB20/072 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: REMDESIVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR ESTER THEREOF; REGISTERED: UK EU/1/20/1459 (NI) 20200703; UK PL GB11972/0036 20200703 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of Veklury (Remdesivir)
More… ↓


