Market Dynamics and Financial Trajectory for the Drug: Vimovo
Introduction
Vimovo, a fixed-dose combination tablet of naproxen and esomeprazole, has been a significant player in the pharmaceutical market, particularly in the treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. This article delves into the market dynamics and financial trajectory of Vimovo, highlighting its acquisition history, sales performance, pricing strategies, and the impact on the healthcare system.
Acquisition History
Vimovo's journey involves several key acquisitions that have shaped its market presence. Initially developed by AstraZeneca, the rights to Vimovo were partially acquired by Grünenthal in 2018. Grünenthal purchased the European rights to Nexium and the global rights to Vimovo (excluding the U.S. and Japan) for up to $922 million. This deal was part of Grünenthal's strategy to expand its pain therapeutics business and AstraZeneca's effort to streamline its drug pipeline[1].
Prior to this, in 2013, AstraZeneca sold the U.S. rights to Vimovo to Horizon Pharma for $40 million, with additional annual payments of $7.5 million[1].
Sales Performance
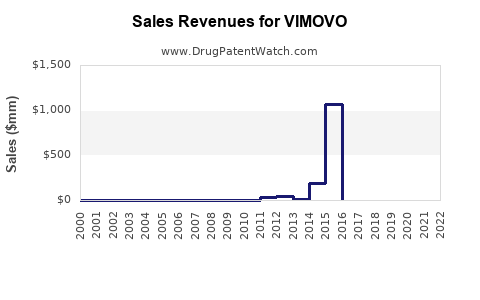
The sales figures for Vimovo have been notable, despite some fluctuations. In the first half of 2018, Vimovo sales worldwide, excluding the U.S. and Japan, were $37 million. However, Grünenthal reported that Vimovo sales for the entire year of 2017 were $79 million[1].
Under Horizon Pharma, Vimovo generated significant revenue. Since 2014, Vimovo's net sales have exceeded $455 million annually. However, by the first quarter of 2017, net sales had dropped to just under $5 million, an 81% decline from the first quarter of 2016, due to reduced insurance coverage and higher rebates demanded by insurers[4].
Pricing Strategies and Market Impact
Vimovo is a combination of two readily available generic medications: naproxen (Aleve) and esomeprazole. Despite the components being cheaply available over-the-counter, Vimovo is priced as a specialty drug, leading to significant costs for the healthcare system.
Horizon Pharma employed a strategic approach to maintain high sales. The company made deals with insurance payers and pharmacy benefit managers, offering special rebates and administrative fees. This strategy ensured that Vimovo remained covered by insurers, despite its high cost. Additionally, Horizon expanded its sales force and focused marketing efforts on doctors who preferred brand-name drugs, emphasizing the convenience of a single pill and improved patient compliance[4].
Financial Trajectory
The financial performance of Vimovo under different ownerships reflects the drug's market dynamics.
- Under AstraZeneca and Grünenthal: The acquisition by Grünenthal marked a significant investment in the company's history, with an upfront payment of $115 million and potential future milestones and sales-related payments of up to $17 million[1].
- Under Horizon Pharma: Despite the high revenue generated, the financial filings of Horizon Pharma indicated challenges in maintaining sales. By 2017, fewer insurance companies were willing to cover Vimovo, leading to higher rebates and reduced net sales[4].
In the fourth quarter of 2022, Horizon Therapeutics (the successor to Horizon Pharma) reported that Vimovo's net sales continued to be a small fraction of their overall revenue, highlighting the declining significance of Vimovo in their portfolio[5].
Impact on the Healthcare System
The pricing and marketing strategies of Vimovo have been criticized for contributing to healthcare cost inflation. Critics argue that the high cost of Vimovo, despite being a combination of cheap generic drugs, is a form of waste in the healthcare system. The strategy of covering patient out-of-pocket costs and offering incentives to doctors and pharmacies has been seen as a way to circumvent cost-control measures[4].
Cost Savings Potential
Studies have shown that prescribing the generic components of Vimovo separately could significantly reduce healthcare costs. For instance, the Center for Improving Value in Health Care (CIVHC) analyzed data from the Colorado All-Payer Claims Database and found that the price of Vimovo and similar drugs has been increasing annually, while the cost of their generic components remains much lower[3].
Consumer and Payer Perspectives
From a consumer perspective, the convenience of a single pill can be appealing, especially when out-of-pocket costs are covered by the manufacturer. However, this convenience comes at a higher cost to insurers and the overall healthcare system. Payers have increasingly demanded larger rebates, reflecting their resistance to the high pricing of Vimovo[4].
Regulatory and Patent Considerations
The patent protection for Vimovo has played a crucial role in its market dynamics. While Nexium lost compound patent protection in most global markets, Vimovo remained patent-protected in most European markets until 2025. This patent protection has allowed Grünenthal to maintain exclusive rights and pricing power in these markets[1].
Key Takeaways
- Acquisition and Ownership: Vimovo's rights have been acquired by Grünenthal and Horizon Pharma at different times, reflecting strategic shifts in the pharmaceutical market.
- Sales Performance: Despite fluctuations, Vimovo has generated significant revenue, particularly under Horizon Pharma.
- Pricing Strategies: The high pricing of Vimovo, despite being a combination of generic drugs, has been a subject of criticism and has contributed to healthcare cost inflation.
- Impact on Healthcare: The strategies employed by manufacturers have circumvented cost-control measures, leading to higher costs for the healthcare system.
- Regulatory and Patent Considerations: Patent protection has been crucial in maintaining market exclusivity and pricing power.
FAQs
What is Vimovo and how is it used?
Vimovo is a fixed-dose combination tablet of naproxen, a non-steroidal anti-inflammatory drug (NSAID), and esomeprazole, a gastroprotective agent. It is used for the symptomatic treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis in patients at risk of developing NSAID-associated gastric and/or duodenal ulcers.
Who are the current owners of Vimovo?
The rights to Vimovo are currently held by Grünenthal for all markets except the U.S. and Japan. Horizon Therapeutics holds the rights for the U.S. market.
Why is Vimovo so expensive despite its components being generic?
Vimovo's high price is due to its marketing and pricing strategies, including deals with insurance payers and pharmacy benefit managers, and the convenience of a single pill, which justifies the premium pricing.
How has the sales performance of Vimovo changed over time?
Vimovo's sales have seen significant fluctuations. Initially, it generated over $455 million in annual sales under Horizon Pharma, but by 2017, sales had dropped significantly due to reduced insurance coverage and higher rebates.
What are the criticisms against Vimovo's pricing strategy?
Critics argue that the high cost of Vimovo contributes to healthcare cost inflation and is a form of waste, as the generic components are much cheaper when prescribed separately.
What is the patent status of Vimovo?
Vimovo is patent-protected in most European markets until 2025, which has allowed Grünenthal to maintain exclusive rights and pricing power in these markets.
Sources
- Grünenthal Acquires Partial Rights to Nexium, Vimovo from AstraZeneca for Up to $922M. Genetic Engineering & Biotechnology News.
- Horizon Therapeutics plc Reports Third-Quarter 2022 Financial Results. BioSpace.
- Drug Cost Savings Potential Vimovo and Duexis. APCD Council.
- How Two Common Medications Became One $455 Million Specialty Pill. ProPublica.
- Horizon Therapeutics plc Reports Fourth-Quarter and Full-Year 2022 Financial Results. Business Wire.