WEGOVY Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Wegovy, and what generic alternatives are available?
Wegovy is a drug marketed by Novo and is included in one NDA. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and fifty-four patent family members in thirty-three countries.
The generic ingredient in WEGOVY is semaglutide. Two suppliers are listed for this compound. Additional details are available on the semaglutide profile page.
DrugPatentWatch® Generic Entry Outlook for Wegovy
Wegovy was eligible for patent challenges on December 5, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 5, 2031. This may change due to patent challenges or generic licensing.
There have been seven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for WEGOVY?
- What are the global sales for WEGOVY?
- What is Average Wholesale Price for WEGOVY?
Summary for WEGOVY
| International Patents: | 154 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 16 |
| Clinical Trials: | 12 |
| Patent Applications: | 702 |
| Drug Prices: | Drug price information for WEGOVY |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for WEGOVY |
| What excipients (inactive ingredients) are in WEGOVY? | WEGOVY excipients list |
| DailyMed Link: | WEGOVY at DailyMed |
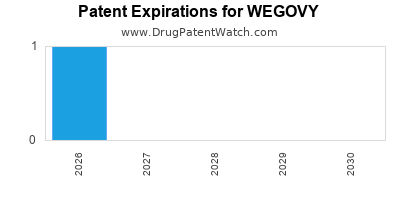
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for WEGOVY
Generic Entry Date for WEGOVY*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;SUBCUTANEOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for WEGOVY
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Merck Sharp & Dohme LLC | Phase 2 |
| Neurobiology Research Unit | Phase 2 |
| Psychiatric Centre Rigshospitalet | Phase 2 |
Pharmacology for WEGOVY
| Drug Class | GLP-1 Receptor Agonist |
| Mechanism of Action | Glucagon-like Peptide-1 (GLP-1) Agonists |
Paragraph IV (Patent) Challenges for WEGOVY
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| WEGOVY | Injection | semaglutide | 0.25 mg/0.5 mL 0.5 mg/0.5 mL 1 mg/0.5 mL 1.7 mg/0.75 mL 2.4 mg/0.75 mL | 215256 | 1 | 2022-10-20 |
US Patents and Regulatory Information for WEGOVY
WEGOVY is protected by eight US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of WEGOVY is ⤷ Try for Free.
This potential generic entry date is based on patent 8,129,343.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novo | WEGOVY | semaglutide | SOLUTION;SUBCUTANEOUS | 215256-004 | Jun 4, 2021 | RX | Yes | Yes | 9,764,003 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Novo | WEGOVY | semaglutide | SOLUTION;SUBCUTANEOUS | 215256-004 | Jun 4, 2021 | RX | Yes | Yes | 12,214,017 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | WEGOVY | semaglutide | SOLUTION;SUBCUTANEOUS | 215256-004 | Jun 4, 2021 | RX | Yes | Yes | 8,536,122 | ⤷ Try for Free | Y | Y | ⤷ Try for Free | ||
| Novo | WEGOVY | semaglutide | SOLUTION;SUBCUTANEOUS | 215256-005 | Jun 4, 2021 | RX | Yes | Yes | 9,764,003 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Novo | WEGOVY | semaglutide | SOLUTION;SUBCUTANEOUS | 215256-005 | Jun 4, 2021 | RX | Yes | Yes | 8,536,122 | ⤷ Try for Free | Y | Y | ⤷ Try for Free | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for WEGOVY
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novo Nordisk A/S | Ozempic | semaglutide | EMEA/H/C/004174 Treatment of adults with insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise:as monotherapy when metformin is considered inappropriate due to intolerance or contraindications;in addition to other medicinal products for the treatment of diabetes.For study results with respect to combinations, effects on glycaemic control and cardiovascular events, and the populations studied, see sections 4.4, 4.5 and 5.1. |
Authorised | no | no | no | 2018-02-08 | |
| Novo Nordisk A/S | Rybelsus | semaglutide | EMEA/H/C/004953 Rybelsus is indicated for the treatment of adults with insufficiently controlled type 2 diabetes mellitus to improve glycaemic control as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance or contraindicationsin combination with other medicinal products for the treatment of diabetes.For study results with respect to combinations, effects on glycaemic control and cardiovascular events, and the populations studied, see sections 4.4, 4.5 and 5.1. |
Authorised | no | no | no | 2020-04-03 | |
| Novo Nordisk A/S | Wegovy | semaglutide | EMEA/H/C/005422 Wegovy is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management, including weight loss and weight maintenance, in adults with an initial Body Mass Index (BMI) of- ≥30 kg/m² (obesity), or- ≥27 kg/m² to |
Authorised | no | no | no | 2022-01-06 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for WEGOVY
See the table below for patents covering WEGOVY around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Brazil | 112022013746 | FORMULAÇÃO AQUOSA DE CAGRILINTIDA, FORMULAÇÃO AQUOSA DE SEMAGLUTIDA, DISPOSITIVO MÉDICO, E, COMBINAÇÃO DE DOSE FIXA | ⤷ Try for Free |
| China | 101133082 | Acylated GLP-1 compounds | ⤷ Try for Free |
| Japan | 7132441 | ⤷ Try for Free | |
| Mexico | 2019008148 | USO DE PEPTIDOS GLP-1 DE ACCION PROLONGADA. (USE OF LONG-ACTING GLP-1 PEPTIDES.) | ⤷ Try for Free |
| Russian Federation | 2015101826 | ПРИМЕНЕНИЕ ДОЛГОДЕЙСТВУЮЩИХ ПЕПТИДОВ GLP-1 | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for WEGOVY
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1863839 | 2018/017 | Ireland | ⤷ Try for Free | PRODUCT NAME: OZEMPIC-SEMAGLUTIDE; REGISTRATION NO/DATE: EU/1/17/1251 20180208 |
| 1863839 | 22/2018 | Austria | ⤷ Try for Free | PRODUCT NAME: SEMAGLUTID; REGISTRATION NO/DATE: EU/1/17/1251 20180212 |
| 1863839 | 300936 | Netherlands | ⤷ Try for Free | PRODUCT NAME: SEMAGLUTIDE; REGISTRATION NO/DATE: EU/1/17/1251 20180208 |
| 1863839 | 661 | Finland | ⤷ Try for Free | |
| 1863839 | C01863839/01 | Switzerland | ⤷ Try for Free | PRODUCT NAME: SEMAGLUTIDE; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66604 02.07.2018 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Wegovy
More… ↓


