ZEJULA Drug Patent Profile
✉ Email this page to a colleague
When do Zejula patents expire, and when can generic versions of Zejula launch?
Zejula is a drug marketed by Glaxosmithkline and is included in two NDAs. There are eight patents protecting this drug.
This drug has two hundred and eighty patent family members in fifty-five countries.
The generic ingredient in ZEJULA is niraparib tosylate. One supplier is listed for this compound. Additional details are available on the niraparib tosylate profile page.
DrugPatentWatch® Generic Entry Outlook for Zejula
Zejula was eligible for patent challenges on March 27, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 27, 2038. This may change due to patent challenges or generic licensing.
There have been four patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ZEJULA?
- What are the global sales for ZEJULA?
- What is Average Wholesale Price for ZEJULA?
Summary for ZEJULA
| International Patents: | 280 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 68 |
| Clinical Trials: | 60 |
| Drug Prices: | Drug price information for ZEJULA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ZEJULA |
| What excipients (inactive ingredients) are in ZEJULA? | ZEJULA excipients list |
| DailyMed Link: | ZEJULA at DailyMed |
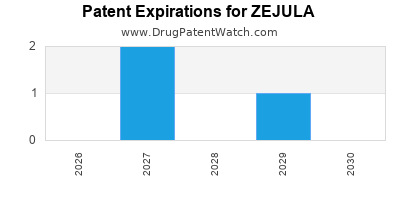

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ZEJULA
Generic Entry Dates for ZEJULA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
Generic Entry Dates for ZEJULA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ZEJULA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Zenith Epigenetics | Phase 1 |
| Haider Mahdi | Phase 1 |
| Institute of Cancer Research, United Kingdom | Phase 2 |
Pharmacology for ZEJULA
| Drug Class | Poly(ADP-Ribose) Polymerase Inhibitor |
| Mechanism of Action | Poly(ADP-Ribose) Polymerase Inhibitors |
US Patents and Regulatory Information for ZEJULA
ZEJULA is protected by nine US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ZEJULA is ⤷ Subscribe.
This potential generic entry date is based on patent 11,091,459.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | ZEJULA | niraparib tosylate | TABLET;ORAL | 214876-003 | Apr 26, 2023 | RX | Yes | Yes | 8,071,579 | ⤷ Subscribe | ⤷ Subscribe | ||||
| Glaxosmithkline | ZEJULA | niraparib tosylate | TABLET;ORAL | 214876-002 | Apr 26, 2023 | RX | Yes | No | 11,730,725 | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Glaxosmithkline | ZEJULA | niraparib tosylate | TABLET;ORAL | 214876-003 | Apr 26, 2023 | RX | Yes | Yes | 11,673,877 | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Glaxosmithkline | ZEJULA | niraparib tosylate | TABLET;ORAL | 214876-002 | Apr 26, 2023 | RX | Yes | No | 8,436,185 | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for ZEJULA
When does loss-of-exclusivity occur for ZEJULA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 18246214
Patent: Niraparib compositions
Estimated Expiration: ⤷ Subscribe
Patent: 21245223
Patent: Niraparib compositions
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 2019020211
Patent: composições de niraparib
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 58375
Patent: COMPOSITIONS DE NIRAPARIB (NIRAPARIB COMPOSITIONS)
Estimated Expiration: ⤷ Subscribe
China
Patent: 0944638
Patent: 尼拉帕尼组合物 (NIRAPARIB COMPOSITIONS)
Estimated Expiration: ⤷ Subscribe
Eurasian Patent Organization
Patent: 1992177
Patent: КОМПОЗИЦИИ НА ОСНОВЕ НИРАПАРИБА
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 00314
Patent: COMPOSITIONS DE NIRAPARIB (NIRAPARIB COMPOSITIONS)
Estimated Expiration: ⤷ Subscribe
Israel
Patent: 9630
Patent: פורמולציות של ניראפריב (Niraparib formulations)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 20512350
Patent: ニラパリブ組成物
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 19011496
Patent: COMPOSICIONES DE NIRAPARIB. (NIRAPARIB COMPOSITIONS.)
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 201909011P
Patent: NIRAPARIB COMPOSITIONS
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 200014736
Patent: 니라파립 조성물
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 61476
Estimated Expiration: ⤷ Subscribe
Patent: 1840315
Patent: NIRAPARIB compositions
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ZEJULA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2006528618 | ⤷ Subscribe | |
| Poland | 2336120 | ⤷ Subscribe | |
| Singapore | 150548 | DNA DAMAGE REPAIR INHIBITORS FOR TREATMENT OF CANCER | ⤷ Subscribe |
| Spain | 2339663 | ⤷ Subscribe | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ZEJULA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2109608 | CA 2018 00017 | Denmark | ⤷ Subscribe | PRODUCT NAME: NIRAPARIB OR A PHARMACEUTICALLY ACCEPTABLE SALT, STEREOISOMER OR TAUTOMER THEREOF, PARTICULARLY THE; REG. NO/DATE: EU/1/17/1235 20171120 |
| 1633724 | C01633724/01 | Switzerland | ⤷ Subscribe | PRODUCT NAME: OLAPARIB; REGISTRATION NO/DATE: SWISSMEDIC 65160 14.01.2016 |
| 1633724 | 92680 | Luxembourg | ⤷ Subscribe | PRODUCT NAME: OLAPARIB AINSI QUE DES SELS ET DES SOLVATES DE CELUI-CI. FIRST REGISTRATION: 20141218 |
| 2109608 | 1890020-9 | Sweden | ⤷ Subscribe | PRODUCT NAME: NIRAPARIB OR A PHARMACEUTICALLY ACCEPTABLE SALT, STEREOISOMER OR TAUTOMER THEREOF, PARTICULARY THE TOSYLATE OR A HYDRATE, ESPECIALLY THE TOSYLATE MONOHYDRATE; REG. NO/DATE: EU/1/17/1235 20171120 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ZEJULA Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


