ZYKADIA Drug Patent Profile
✉ Email this page to a colleague
When do Zykadia patents expire, and what generic alternatives are available?
Zykadia is a drug marketed by Novartis and is included in two NDAs. There are seven patents protecting this drug.
This drug has two hundred and eighty-six patent family members in fifty-four countries.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this compound. One supplier is listed for this compound. Additional details are available on the ceritinib profile page.
DrugPatentWatch® Generic Entry Outlook for Zykadia
Zykadia was eligible for patent challenges on April 29, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be January 18, 2032. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ZYKADIA?
- What are the global sales for ZYKADIA?
- What is Average Wholesale Price for ZYKADIA?
Summary for ZYKADIA
| International Patents: | 286 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 98 |
| Clinical Trials: | 11 |
| Patent Applications: | 782 |
| Drug Prices: | Drug price information for ZYKADIA |
| What excipients (inactive ingredients) are in ZYKADIA? | ZYKADIA excipients list |
| DailyMed Link: | ZYKADIA at DailyMed |
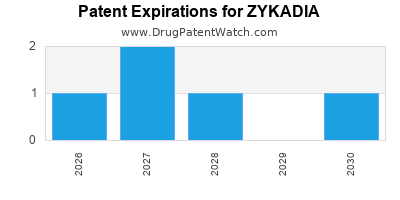

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ZYKADIA
Generic Entry Dates for ZYKADIA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
Generic Entry Dates for ZYKADIA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ZYKADIA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Plateforme labellisée Inca – Institut Bergonié, Bordeaux | Phase 3 |
| Plateforme labellisée Inca - Hôpital Européen Georges Pompidou, Paris | Phase 3 |
| Commissariat A L'energie Atomique | Phase 3 |
Pharmacology for ZYKADIA
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 2C9 Inhibitors Cytochrome P450 3A Inhibitors Tyrosine Kinase Inhibitors |
US Patents and Regulatory Information for ZYKADIA
ZYKADIA is protected by seven US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ZYKADIA is ⤷ Subscribe.
This potential generic entry date is based on patent 9,309,229.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | ZYKADIA | ceritinib | CAPSULE;ORAL | 205755-001 | Apr 29, 2014 | DISCN | Yes | No | 8,377,921 | ⤷ Subscribe | ⤷ Subscribe | ||||
| Novartis | ZYKADIA | ceritinib | CAPSULE;ORAL | 205755-001 | Apr 29, 2014 | DISCN | Yes | No | 7,964,592 | ⤷ Subscribe | Y | Y | ⤷ Subscribe | ||
| Novartis | ZYKADIA | ceritinib | CAPSULE;ORAL | 205755-001 | Apr 29, 2014 | DISCN | Yes | No | 8,399,450 | ⤷ Subscribe | Y | Y | ⤷ Subscribe | ||
| Novartis | ZYKADIA | ceritinib | TABLET;ORAL | 211225-001 | Mar 18, 2019 | RX | Yes | Yes | 8,039,479 | ⤷ Subscribe | Y | Y | ⤷ Subscribe | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ZYKADIA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novartis | ZYKADIA | ceritinib | TABLET;ORAL | 211225-001 | Mar 18, 2019 | 9,416,112 | ⤷ Subscribe |
| Novartis | ZYKADIA | ceritinib | TABLET;ORAL | 211225-001 | Mar 18, 2019 | 9,018,204 | ⤷ Subscribe |
| Novartis | ZYKADIA | ceritinib | TABLET;ORAL | 211225-001 | Mar 18, 2019 | 8,188,276 | ⤷ Subscribe |
| Novartis | ZYKADIA | ceritinib | CAPSULE;ORAL | 205755-001 | Apr 29, 2014 | 9,416,112 | ⤷ Subscribe |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for ZYKADIA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novartis Europharm Limited | Zykadia | ceritinib | EMEA/H/C/003819 Zykadia is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK) positive advanced non small cell lung cancer (NSCLC) previously treated with crizotinib. |
Authorised | no | no | no | 2015-05-06 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ZYKADIA
When does loss-of-exclusivity occur for ZYKADIA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 4309
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN-4-IL-FENIL)-N4-[2-(PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMINA
Estimated Expiration: ⤷ Subscribe
Patent: 2395
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN-4-IL-FENIL)-N4-[2-(PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMINA
Estimated Expiration: ⤷ Subscribe
Australia
Patent: 11343775
Patent: Crystalline forms of 5-chloro-N2-(2-isopropoxy-5-methyl- 4-piperidin-4-yl-phenyl)-N4[2-(propane-2-sulfonyl)-phenyl] -pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Brazil
Patent: 2013015000
Patent: formas cristalinas de 5-cloron2-(2-isopropóxi-5-metil-4-piperidin-4-il-fenil)-n4-[2-(propano-2-sulfonil)-fenil]-pirimidina-2,4-diamina
Estimated Expiration: ⤷ Subscribe
Canada
Patent: 21102
Patent: FORMES CRISTALLINES DE LA 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4-[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Chile
Patent: 13001723
Patent: Formas cristalinas de 5-cloro-n2-(2-isopropoxi-5-metil-4-piperidin-4-il-fenil)-n4-[2-(propan-2-sulfonil)-fenil]-pirimidin-2,4-diamina; metodos de preparacion; composicion farmaceutica; y el uso en el tratamiento de los trastornos mediados por la cinasa de linfoma anaplasico.
Estimated Expiration: ⤷ Subscribe
China
Patent: 3282359
Patent: Crystalline forms of 5-chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Patent: 4262324
Patent: Crystalline Forms Of 5-chloro-n2-(2-isopropoxy-5-methyl- 4-piperidin-4-yl-phenyl)-n4[2-(propane-2-sulfonyl)-phenyl] -pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Patent: 6008462
Patent: 种ALK抑制剂的结晶形式 (Crystalline forms of ALK inhibitor)
Estimated Expiration: ⤷ Subscribe
Patent: 6831716
Patent: 种ALK抑制剂的结晶形式 (Crystallization of ALK inhibitor)
Estimated Expiration: ⤷ Subscribe
Patent: 7056751
Patent: 种ALK抑制剂的结晶形式 (crystalline forms of ALK inhibitor)
Estimated Expiration: ⤷ Subscribe
Patent: 2125884
Patent: 制备嘧啶-2,4-二胺二盐酸盐的方法 (Method for preparing pyrimidine-2, 4-diamine dihydrochloride)
Estimated Expiration: ⤷ Subscribe
Patent: 4989139
Patent: 制备嘧啶-2,4-二胺二盐酸盐的方法 (Method for preparing pyrimidine-2, 4-diamine dihydrochloride)
Estimated Expiration: ⤷ Subscribe
Colombia
Patent: 01792
Patent: Formas cristalinas de 5-cloro-n2-(2-isoporpoxi 5-metil-4-piperidin-4-il-fenil)-n4-[2-(propan-2-sulfonil)-fenil]-pirimidin-2,4-diamina.
Estimated Expiration: ⤷ Subscribe
Croatia
Patent: 0171477
Estimated Expiration: ⤷ Subscribe
Patent: 0181737
Estimated Expiration: ⤷ Subscribe
Cyprus
Patent: 19474
Estimated Expiration: ⤷ Subscribe
Patent: 21017
Estimated Expiration: ⤷ Subscribe
Denmark
Patent: 51918
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Estimated Expiration: ⤷ Subscribe
Ecuador
Patent: 13012770
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN-4-IL-FENIL)-N4-[2-(PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMINA
Estimated Expiration: ⤷ Subscribe
European Patent Office
Patent: 51918
Patent: FORMES CRISTALLINES DE LA 5-CHLORO-N2-(2-ISOPROPOXY-5-MÉTHYL-4-PIPÉRIDIN-4-YL-PHÉNYL)-N4[2-(PROPANE-2-SULFONYL)-PHÉNYL]-PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Patent: FORMES CRISTALLINES DE LA 5-CHLORO-N2-(2-ISOPROPOXY-5-MÉTHYL-4-PIPÉRIDIN-4-YL-PHÉNYL)-N4[2-(PROPANE-2-SULFONYL)-PHÉNYL]-PYRIMIDINE-2,4-DIAMINE (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 53708
Patent: PROCÉDÉ POUR LA PRÉPARATION DE 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N-2-(ISOPROPYLSULFONYL)PHENYL)-2,4-DIAMINE DI-HYDROCHLORIDE (PROCESS FOR THE PREPARATION OF 5-CHLORO-N-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N-2-(ISOPROPYLSULFONYL)PHENYL)-2,4-DIAMINE DI-HYDROCHLORIDE)
Estimated Expiration: ⤷ Subscribe
Guatemala
Patent: 1300153
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN-4-IL-FENIL)- N4-[2-(PROPAN-2-SULFONIL)-FENIL PIRIMIDIM-2,4-DIAMINA
Estimated Expiration: ⤷ Subscribe
Hungary
Patent: 41845
Estimated Expiration: ⤷ Subscribe
Israel
Patent: 6474
Patent: צורות גבישיות של 5-כלורו-n2-(2-איזופרופוקסי-5-מתיל-4-פיפרידנ-4-איל-פניל)-n4[2-(פרופאנ-2-סולפוניל)-פניל]פירימידינ-4,2-דיאמין (Crystalline forms of 5-chloro-n2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-n4[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine)
Estimated Expiration: ⤷ Subscribe
Japan
Patent: 16752
Estimated Expiration: ⤷ Subscribe
Patent: 13545812
Estimated Expiration: ⤷ Subscribe
Lithuania
Patent: 51918
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Estimated Expiration: ⤷ Subscribe
Malaysia
Patent: 4810
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4-[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2, 4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Patent: 7742
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Mexico
Patent: 8210
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN -4-IL-FENIL)-N4-[2- (PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMI NA. (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERID IN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4 -DIAMINE.)
Estimated Expiration: ⤷ Subscribe
Patent: 13006952
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN -4-IL-FENIL)-N4-[2- (PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMI NA. (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERID IN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4 -DIAMINE.)
Estimated Expiration: ⤷ Subscribe
Morocco
Patent: 771
Patent: طريقة لتحديد الخلايا التي لها حساسية من تعديل إشارة مستقبل عامل نمو الخلايا الليفية او الأصناف الاخرى.
Estimated Expiration: ⤷ Subscribe
New Zealand
Patent: 0713
Patent: Crystalline forms of 5-chloro-n2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-n4-[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Peru
Patent: 140698
Patent: FORMAS CRISTALINAS DE 5-CLORO-N2-(2-ISOPROPOXI-5-METIL-4-PIPERIDIN-4-IL-FENIL)-N4-[2-(PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2, 4-DIAMINA
Estimated Expiration: ⤷ Subscribe
Poland
Patent: 51918
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Estimated Expiration: ⤷ Subscribe
Portugal
Patent: 51918
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Estimated Expiration: ⤷ Subscribe
Russian Federation
Patent: 99785
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N2-(2-ИЗОПРОПОКСИ-5-МЕТИЛ-4-ПИПЕРИДИН-4-ИЛ-ФЕНИЛ)-N4-[2-(ПРОПАН-2-СУЛЬФОНИЛ)-ФЕНИЛ]-ПИРИМИДИН-2,4-ДИАМИНА (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4-[2-(PROPANE-2-SULPHONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 46159
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N2-(2-ИЗОПРОПОКСИ-5-МЕТИЛ-4-ПИПЕРИДИН-4-ИЛ-ФЕНИЛ)-N4-[2-(ПРОПАН-2-СУЛЬФОНИЛ)-ФЕНИЛ]-ПИРИМИДИН-2,4-ДИАМИНА (CRYSTAL FORMS 5-CHLORO-N2 - (2-ISOPROPOXY-5-METHYL-4-PIPERIDINE-4-YL-PHENYL) - N 4 - [2-(PROPANE-2-SULFONYL)- PHENYL] - PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 13132947
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N2-(2-ИЗОПРОПОКСИ-5-МЕТИЛ-4-ПИПЕРИДИН-4-ИЛ-ФЕНИЛ)-N4-[2-(ПРОПАН-2-СУЛЬФОНИЛ)-ФЕНИЛ]-ПИРИМИДИН-2,4-ДИАМИНА
Estimated Expiration: ⤷ Subscribe
Patent: 16136823
Patent: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 5-ХЛОР-N2-(2-ИЗОПРОПОКСИ-5-МЕТИЛ-4-ПИПЕРИДИН-4-ИЛ-ФЕНИЛ)-N4-[2-(ПРОПАН-2-СУЛЬФОНИЛ)-ФЕНИЛ]-ПИРИМИДИН-2,4-ДИАМИНА
Estimated Expiration: ⤷ Subscribe
Serbia
Patent: 771
Patent: KRISTALNI OBLICI 5-HLORO-N2-(2-IZOPROPOKSI-5-METIL-4-PIPERIDIN-4-IL-FENIL)-N4-[2-(PROPAN-2-SULFONIL)-FENIL]-PIRIMIDIN-2,4-DIAMINA (CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Singapore
Patent: 0856
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Patent: 201510082X
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Slovenia
Patent: 51918
Estimated Expiration: ⤷ Subscribe
Patent: 21171
Estimated Expiration: ⤷ Subscribe
South Africa
Patent: 1303599
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE2-,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
South Korea
Patent: 2325775
Estimated Expiration: ⤷ Subscribe
Patent: 130130022
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Patent: 180032680
Patent: 5-클로로-N2--페닐]-피리미딘-2,4-디아민의 결정질 형태 (5--2-2--5--4--4---4[2--2--]--24- CRYSTALLINE FORMS OF 5-CHLORO-N2-2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL-N4[2-PROPANE-2-SULFONYL-PHENYL]-PYRIMIDINE-24-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 190022903
Patent: 5-클로로-N2--페닐]-피리미딘-2,4-디아민의 결정질 형태 (5--2-2--5--4--4---4[2--2--]--24- CRYSTALLINE FORMS OF 5-CHLORO-N2-2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL-N4[2-PROPANE-2-SULFONYL-PHENYL]-PYRIMIDINE-24-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Patent: 200039021
Patent: 5-클로로-N2--페닐]-피리미딘-2,4-디아민의 결정질 형태 (5--2-2--5--4--4---4[2--2--]--24- CRYSTALLINE FORMS OF 5-CHLORO-N2-2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL-N4[2-PROPANE-2-SULFONYL-PHENYL]-PYRIMIDINE-24-DIAMINE)
Estimated Expiration: ⤷ Subscribe
Spain
Patent: 43016
Estimated Expiration: ⤷ Subscribe
Patent: 96526
Estimated Expiration: ⤷ Subscribe
Patent: 05973
Estimated Expiration: ⤷ Subscribe
Taiwan
Patent: 76343
Estimated Expiration: ⤷ Subscribe
Patent: 76344
Estimated Expiration: ⤷ Subscribe
Patent: 1307299
Patent: Crystalline forms of 5-chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Patent: 1629021
Patent: Crystalline forms of 5-chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine
Estimated Expiration: ⤷ Subscribe
Tunisia
Patent: 13000216
Patent: CRYSTALLINE FORMS OF 5-CHLORO-N2-(2-ISOPROPOXY-5-METHYL-4-PIPERIDIN-4-YL-PHENYL)-N4[2-(PROPANE-2-SULFONYL)-PHENYL]-PYRIMIDINE-2,4-DIAMINE
Estimated Expiration: ⤷ Subscribe
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ZYKADIA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2009032703 | ⤷ Subscribe | |
| Eurasian Patent Organization | 201100688 | СОЕДИНЕНИЯ И КОМПОЗИЦИИ В КАЧЕСТВЕ ИНГИБИТОРОВ ПРОТЕИНКИНАЗЫ | ⤷ Subscribe |
| Lithuania | 3121171 | ⤷ Subscribe | |
| Israel | 166241 | 2,4-PYRIMIDINEDIAMINE COMPOUNDS FOR USE IN THE TREATMENT OF AUTOIMMUNE DISEASES | ⤷ Subscribe |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ZYKADIA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2091918 | PA2015034,C2091918 | Lithuania | ⤷ Subscribe | PRODUCT NAME: CERITINIBAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/15/999 20150506 |
| 2091918 | 300763 | Netherlands | ⤷ Subscribe | PRODUCT NAME: CERITINIB, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/15/999 20150508 |
| 2091918 | PA2015034 | Lithuania | ⤷ Subscribe | PRODUCT NAME: CERITINIBUM; REGISTRATION NO/DATE: EU/1/15/999 20150506 |
| 2091918 | 528 | Finland | ⤷ Subscribe | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ZYKADIA Market Analysis and Financial Projection Experimental
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


