TEVA Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TEVA, and when can generic versions of TEVA drugs launch?
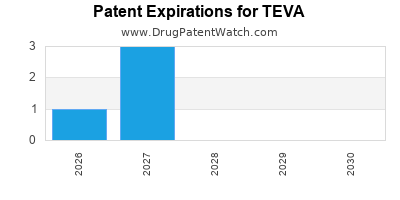
TEVA has seven hundred and thirty-nine approved drugs.
There are one hundred and three US patents protecting TEVA drugs. There are fifty-three tentative approvals on TEVA drugs.
There are nine hundred and forty-four patent family members on TEVA drugs in forty-nine countries and one thousand and eighty-three supplementary protection certificates in eighteen countries.
Summary for TEVA
| International Patents: | 944 |
| US Patents: | 103 |
| Tradenames: | 509 |
| Ingredients: | 444 |
| NDAs: | 739 |
| Patent Litigation for TEVA: | See patent lawsuits for TEVA |
| PTAB Cases with TEVA as petitioner: | See PTAB cases with TEVA as petitioner |
Drugs and US Patents for TEVA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-002 | Apr 3, 2017 | RX | Yes | No | ⤷ Subscribe | ⤷ Subscribe | |||||
| Teva Pharm | AIRDUO DIGIHALER | fluticasone propionate; salmeterol xinafoate | POWDER;INHALATION | 208799-004 | Jul 12, 2019 | DISCN | Yes | No | 10,124,131*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Teva Pharm | AIRDUO RESPICLICK | fluticasone propionate; salmeterol xinafoate | POWDER;INHALATION | 208799-001 | Jan 27, 2017 | RX | Yes | No | 9,415,008*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Teva Branded Pharm | PROAIR DIGIHALER | albuterol sulfate | POWDER, METERED;INHALATION | 205636-002 | Dec 21, 2018 | DISCN | Yes | No | 10,918,816 | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| Teva Pharm | ARMONAIR DIGIHALER | fluticasone propionate | POWDER;INHALATION | 208798-008 | Apr 8, 2022 | DISCN | Yes | No | 11,266,796*PED | ⤷ Subscribe | Y | ⤷ Subscribe | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TEVA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Teva Branded Pharm | LOSEASONIQUE | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 022262-001 | Oct 24, 2008 | 7,858,605 | ⤷ Subscribe |
| Teva Branded Pharm | QVAR 80 | beclomethasone dipropionate | AEROSOL, METERED;INHALATION | 020911-001 | Sep 15, 2000 | 5,766,573 | ⤷ Subscribe |
| Teva Pharm | AIRDUO DIGIHALER | fluticasone propionate; salmeterol xinafoate | POWDER;INHALATION | 208799-005 | Jul 12, 2019 | 6,871,646 | ⤷ Subscribe |
| Teva Branded Pharm | ZIAC | bisoprolol fumarate; hydrochlorothiazide | TABLET;ORAL | 020186-003 | Mar 26, 1993 | 4,258,062 | ⤷ Subscribe |
| Teva Branded Pharm | PROAIR RESPICLICK | albuterol sulfate | POWDER, METERED;INHALATION | 205636-001 | Mar 31, 2015 | 8,006,690 | ⤷ Subscribe |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for TEVA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 5 mg, 10 mg, 20 mg, 30 mg | ➤ Subscribe | 2009-11-18 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-02-26 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg/0.01 mg | ➤ Subscribe | 2008-01-22 |
| ➤ Subscribe | Tablets | 0.1 mg/0.02 mg and 0.01 mg | ➤ Subscribe | 2009-11-16 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Injection | 3.5 mg/vial | ➤ Subscribe | 2016-10-26 |
| ➤ Subscribe | Tablets | 0.5 mg and 1 mg | ➤ Subscribe | 2010-05-17 |
| ➤ Subscribe | Extended-release Capsule | 15 mg and 30 mg | ➤ Subscribe | 2008-08-11 |
| ➤ Subscribe | Injection | 40 mg/mL, 1 mL pre-filled syringe | ➤ Subscribe | 2014-01-29 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg | ➤ Subscribe | 2004-03-29 |
| ➤ Subscribe | Tablets | 0.15 mg/0.02 mg, 0.15 mg/0.025 mg, 0.15 mg/0.03 mg and 0.01 mg | ➤ Subscribe | 2013-07-10 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Tablets | 1 mg/0.02 mg and 75 mg | ➤ Subscribe | 2006-04-17 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
Premature patent expirations for TEVA
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Subscribe | ⤷ Subscribe |
International Patents for TEVA Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 5591941 | ⤷ Subscribe |
| Japan | 2014223544 | ⤷ Subscribe |
| China | 114306839 | ⤷ Subscribe |
| New Zealand | 610790 | ⤷ Subscribe |
| Eurasian Patent Organization | 201390490 | ⤷ Subscribe |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for TEVA Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1519731 | 132013902182575 | Italy | ⤷ Subscribe | PRODUCT NAME: AZELASTINA CLORIDRATO/FLUTICASONE PROPIONATO(DYMISTA); AUTHORISATION NUMBER(S) AND DATE(S): 2011/07125-REG, 20111024;041808015/M-027/M-039/M-041/M, 20130527 |
| 3141251 | 301099 | Netherlands | ⤷ Subscribe | PRODUCT NAME: A MEDICINAL PRODUCT CONSISTING OF A COMBINATION OF A FIRST DOSE PHARMACEUTICAL COMPOSITION AND A SECOND DOSE PHARMACEUTICAL COMPOSITION, THE FIRST DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, SODIUM SULPHATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE AND THE SECOND DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, ASCORBIC ACID, SODIUM ASCORBATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE; NATIONAL REGISTRATION NO/DATE: RVG 120195 20171114; FIRST REGISTRATION: IS IS/1/17/083/01 20171016 |
| 2782584 | 21C1058 | France | ⤷ Subscribe | PRODUCT NAME: COMPOSITION CONTENANT A LA FOIS DE L'ESTRADIOL (17SS-ESTRADIOL), Y COMPRIS SOUS FORME HEMIHYDRATEE, ET DE LA PROGESTERONE; NAT. REGISTRATION NO/DATE: NL51886 20210421; FIRST REGISTRATION: BE - BE582231 20210406 |
| 2924034 | LUC00114 | Luxembourg | ⤷ Subscribe | PRODUCT NAME: DORAVIRINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI EN COMBINAISON AVEC LA LAMIVUDINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI EN COMBINAISON AVEC DU TENOFOVIR OU UN DE SES ESTERS, EN PARTICULIER UN ESTER DE DISOPROXIL OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES, EN PARTICULIER UN SEL DE FUMARATE; AUTHORISATION NUMBER AND DATE: EU/1/18/1333 20181126 |
| 0770388 | 2009/012 | Ireland | ⤷ Subscribe | PRODUCT NAME: QLAIRA-ESTRADIOL VALERATE/DIENOGEST; NAT REGISTRATION NO/DATE: PA1410/58/1 20090109; FIRST REGISTRATION NO/DATE: BE327792 20081103 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.