In the dynamic world of pharmaceuticals, Teva Pharmaceutical Industries Limited stands as a formidable player, known for its extensive portfolio of generic and branded medications. This article delves into Teva's market position, strengths, and strategic insights, providing a comprehensive analysis of its competitive landscape in the branded pharmaceutical sector.
Teva's Market Position in the Pharmaceutical Industry
Teva Pharmaceutical Industries Limited has established itself as a global leader in the pharmaceutical industry. As the world's largest generic drug manufacturer, Teva has leveraged its expertise to build a strong presence in the branded pharmaceutical market as well[1]. The company's dual focus on generic and branded drugs has allowed it to capture a significant market share and maintain a competitive edge.
Global Reach and Market Presence
Teva's global footprint is impressive, with operations spanning over 60 countries[3]. This extensive reach allows the company to serve diverse markets and adapt to regional healthcare needs. Teva's ability to navigate different regulatory environments and market conditions has contributed significantly to its success in the branded pharmaceutical sector.
Teva is listed on the Tel Aviv Stock Exchange and the New York Stock Exchange. Its manufacturing facilities are located in Israel, North America, Europe, Australia, and South America[1].
Ranking Among Pharmaceutical Giants
In terms of overall pharmaceutical sales, Teva ranks as the 18th largest pharmaceutical company globally[1]. This positioning places Teva in direct competition with industry giants such as Pfizer, Novartis, and Johnson & Johnson[3]. Despite facing stiff competition, Teva has managed to carve out a niche for itself, particularly in the generic drug market, while steadily growing its branded pharmaceutical portfolio.
Teva's Branded Pharmaceutical Portfolio
Teva's branded pharmaceutical division focuses on developing innovative medicines to address unmet medical needs. The company's branded portfolio includes treatments for various therapeutic areas, with a particular emphasis on central nervous system disorders and respiratory conditions.
Key Branded Products
Some of Teva's primary branded products include:
-
Austedo (deutetrabenazine): Used for the treatment of chorea associated with Huntington's disease and tardive dyskinesia[1].
-
Ajovy (fremanezumab): A preventive treatment for migraine in adults[1].
-
Copaxone: A treatment for multiple sclerosis, primarily sold in the United States[1].
-
Bendeka and Treanda: Oncology treatments[1].
These branded medications form the backbone of Teva's innovative drug portfolio, contributing significantly to the company's revenue and market position.
Strengths and Competitive Advantages
Teva's success in the branded pharmaceutical market can be attributed to several key strengths and competitive advantages.
Diverse Product Portfolio
Teva's diverse product portfolio, spanning both generic and branded medications, provides a significant competitive advantage. This diversification helps mitigate risks associated with any single product or market segment, ensuring stability and sustained revenue growth[4].
Strong Research and Development Capabilities
Teva invests heavily in research and development to drive innovation and bring new drugs to market. The company's commitment to R&D allows it to develop novel treatments and maintain a competitive edge in the branded pharmaceutical sector[4].
Cost-Effective Manufacturing
Teva's efficient manufacturing processes enable the production of high-quality medicines at competitive prices. This cost-effectiveness is particularly advantageous in the branded pharmaceutical market, where pricing pressures are increasingly prevalent[6].
Global Distribution Network
Teva's robust distribution network ensures that its products reach patients in a timely manner. This strong distribution infrastructure enhances the company's market presence and customer satisfaction, particularly for its branded medications[6].
Strategic Insights and Future Outlook
To maintain and enhance its position in the branded pharmaceutical market, Teva has adopted several strategic initiatives.
Focus on Specialty Medicines
Teva is increasingly focusing on specialty medicines, which offer higher profit margins compared to generic drugs. The company's strategy involves developing and commercializing specialty medicines to meet specific patient needs and healthcare provider requirements[4].
Expansion into Emerging Markets
Teva recognizes the growth potential in emerging markets such as China, India, and Brazil. The company is actively pursuing expansion strategies in these regions to tap into the rising demand for healthcare products[4].
Investment in Biosimilars
Biosimilars represent a significant growth opportunity for Teva. The company is leveraging its scientific expertise and advanced manufacturing capabilities to develop and commercialize biosimilars, aiming to expand access to these treatments and drive down healthcare costs globally[4].
Strategic Partnerships and Collaborations
Teva actively seeks strategic partnerships and collaborations to enhance its capabilities and market reach. These partnerships often involve joint research and development efforts, co-marketing agreements, or technology sharing arrangements[6].
Challenges in the Competitive Landscape
Despite its strengths, Teva faces several challenges in the competitive pharmaceutical landscape.
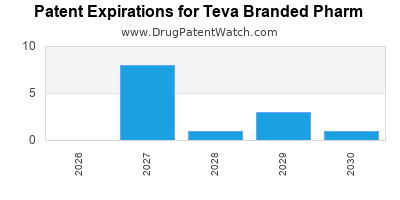
Patent Expirations
Like all pharmaceutical companies, Teva must contend with patent expirations on its branded medications. As patents expire, the company faces increased competition from generic manufacturers, potentially impacting revenue from its branded portfolio[6].
Pricing Pressures
The pharmaceutical industry is experiencing increasing pressure to reduce drug prices. This trend poses a challenge for Teva's branded medications, which typically command higher prices than generic alternatives[6].
Regulatory Hurdles
The pharmaceutical industry is heavily regulated, and changes in regulatory requirements can impact Teva's operations, time to market, and overall profitability. Navigating these regulatory challenges is crucial for the company's success in the branded pharmaceutical market[4].
Teva's Commitment to Quality and Compliance
In the highly regulated pharmaceutical industry, maintaining high standards of quality and compliance is essential. Teva places a strong emphasis on these aspects to ensure the safety and efficacy of its products.
Quality Control Measures
Teva implements rigorous quality control measures throughout its manufacturing and distribution processes. The company's commitment to quality is evident in its state-of-the-art manufacturing facilities and adherence to Good Manufacturing Practices (GMP)[2].
Regulatory Compliance
Teva dedicates significant resources to ensure compliance with regulatory requirements across all markets in which it operates. This focus on compliance helps the company maintain its reputation and build trust with healthcare providers and patients[2].
Innovation and Future Growth
Innovation is at the heart of Teva's strategy for future growth in the branded pharmaceutical market.
Investment in Research and Development
Teva continues to invest heavily in research and development to bring innovative treatments to market. The company's R&D efforts focus on addressing unmet medical needs and improving patient outcomes[3].
Digital Health Initiatives
Recognizing the growing importance of digital health, Teva is exploring opportunities to integrate digital technologies into its branded pharmaceutical offerings. These initiatives aim to enhance patient care and improve treatment outcomes[6].
Teva's Corporate Social Responsibility
As a leading pharmaceutical company, Teva recognizes its responsibility to contribute to global health and sustainability.
Access to Medicines
Teva is committed to improving access to medicines globally, particularly in underserved communities. The company collaborates with governments, non-profit organizations, and other stakeholders to increase access to essential medicines and healthcare services[4].
Environmental Sustainability
Teva is taking steps to reduce its environmental impact and promote sustainability across its operations. These efforts include reducing carbon emissions, minimizing waste, and implementing sustainable manufacturing practices[6].
Key Takeaways
- Teva Pharmaceutical Industries Limited is a global leader in both generic and branded pharmaceuticals, ranking as the 18th largest pharmaceutical company worldwide.
- The company's branded pharmaceutical portfolio includes key products like Austedo, Ajovy, and Copaxone, focusing on central nervous system disorders and respiratory conditions.
- Teva's strengths include its diverse product portfolio, strong R&D capabilities, cost-effective manufacturing, and global distribution network.
- Strategic initiatives focus on specialty medicines, expansion into emerging markets, investment in biosimilars, and strategic partnerships.
- Challenges include patent expirations, pricing pressures, and regulatory hurdles.
- Teva maintains a strong commitment to quality, compliance, innovation, and corporate social responsibility.
FAQs
-
What is Teva's position in the global pharmaceutical market?
Teva is the world's largest generic drug manufacturer and ranks as the 18th largest pharmaceutical company overall.
-
What are some of Teva's key branded products?
Teva's key branded products include Austedo, Ajovy, Copaxone, Bendeka, and Treanda.
-
How is Teva addressing the challenges of patent expirations?
Teva is focusing on developing new innovative medicines and expanding its specialty pharmaceutical portfolio to offset potential revenue losses from patent expirations.
-
What role does research and development play in Teva's strategy?
R&D is crucial to Teva's strategy, driving innovation and the development of new branded medications to address unmet medical needs.
-
How is Teva contributing to global health beyond its product offerings?
Teva is committed to improving access to medicines in underserved communities and implementing sustainable practices across its operations.
Sources cited:
- https://en.wikipedia.org/wiki/Teva_Pharmaceuticals
- https://www.tevausa.com/about-teva/article-pages/our-quality/
- https://canvasbusinessmodel.com/blogs/growth-strategy/teva-pharmaceuticals-growth-strategy
- https://pitchgrade.com/companies/teva-pharmaceutical-industries
- https://canvasbusinessmodel.com/blogs/competitors/teva-pharmaceuticals-competitive-landscape