Introduction to Albuterol Sulfate
Albuterol sulfate, a short-acting beta agonist, is a widely used medication for managing respiratory conditions such as asthma and chronic obstructive pulmonary disease (COPD). Its efficacy in preventing and treating symptoms like wheezing, difficulty breathing, and coughing has made it a cornerstone in respiratory care.
Market Size and Growth Projections
The global albuterol sulfate market is poised for significant growth over the next few years. Here are some key projections:
- The market was valued at USD 5.22 billion in 2023 and is expected to reach USD 9.14 billion by 2030, growing at a Compound Annual Growth Rate (CAGR) of 8.32% during the forecast period[1].
- Another report indicates that the market size was valued at USD 2.38 billion in 2023 and is expected to reach USD 4.07 billion by 2031, growing at a CAGR of 7.1% from 2024 to 2031[3].
Key Drivers of Market Growth
Several factors are driving the growth of the albuterol sulfate market:
Increasing Prevalence of Respiratory Diseases
The rising incidence of respiratory disorders such as asthma, COPD, and bronchitis is a major driver. These conditions are becoming more prevalent due to factors like air pollution, urbanization, and lifestyle changes[4].
Advancements in Inhalation Therapy
Innovations in inhaler technology, including metered-dose and dry powder inhalers, are expanding treatment options and improving patient compliance. Regulatory approvals for generic formulations also contribute to market growth[3].
Growing Awareness and Diagnosis
Improved awareness and diagnostic techniques have led to earlier detection and better management of respiratory conditions. This increased diagnosis rate translates into higher demand for albuterol sulfate[4].
Lifestyle Changes
Modern lifestyles, characterized by sedentary habits, poor dietary choices, and exposure to environmental pollutants, contribute to the development and exacerbation of respiratory diseases. Albuterol plays a crucial role in managing these lifestyle-related disorders[4].
Government Initiatives and Healthcare Expenditure
Governments and healthcare organizations are investing in awareness campaigns, research, and healthcare infrastructure to manage respiratory diseases. These initiatives increase access to albuterol medications, driving market growth[4].
Regional Market Dynamics
North America
North America dominates the albuterol sulfate inhalation aerosol market, accounting for 40% of the global market share. This dominance is attributed to the region's robust healthcare infrastructure, high prevalence of respiratory disorders, and strategic advancements in pharmaceutical development[1].
Financial Impact and Cost Savings
Formulary Substitutions
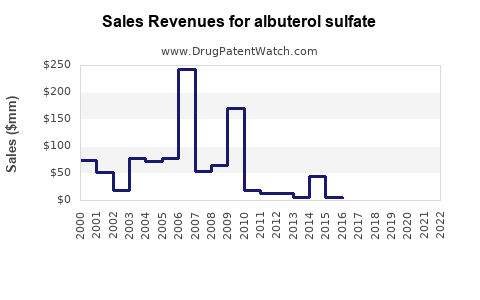
Studies have shown that formulary substitutions can lead to significant cost savings. For instance, substituting ipratropium-albuterol nebulization solution for metered-dose inhalers (MDIs) resulted in a three-month savings of $99,359 in drug costs and an extrapolated full-year savings of $397,436. Despite additional costs for equipment and personnel, the overall savings were substantial[2].
Cost Variability
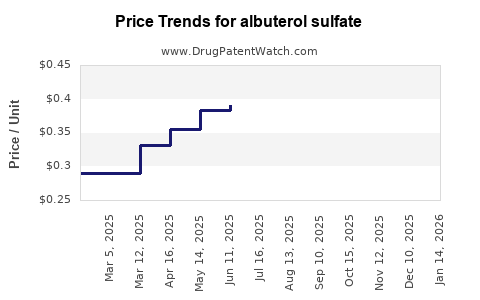
The cost of albuterol sulfate can vary significantly based on factors such as insurance coverage, treatment plan, and the pharmacy used. Generic forms of albuterol tend to be less expensive than brand-name versions, and strategies like obtaining a 90-day supply can help reduce long-term costs[5].
Market Opportunities
Combination Therapies
Combination therapies that include albuterol along with other respiratory medications offer significant opportunities for market growth. These therapies provide enhanced efficacy and convenience, simplifying treatment regimens for patients[4].
Innovations in Delivery Systems
Ongoing research into new formulations and delivery systems, such as vibrating mesh nebulizers, is expected to further advance the market. Patient education programs and healthcare reforms promoting respiratory health awareness also support market expansion[3].
Challenges and Considerations
Regulatory Framework
The market is influenced by regulatory approvals and healthcare policies. Changes in these areas can impact the availability and affordability of albuterol sulfate medications[4].
Patient Compliance
Ensuring patient compliance with treatment regimens is crucial. Innovations in inhaler technology and patient education programs can help improve compliance rates[3].
Key Takeaways
- The albuterol sulfate market is expected to grow significantly, driven by increasing respiratory disease prevalence, advancements in inhalation therapy, and government initiatives.
- North America is the largest market due to its robust healthcare infrastructure and high prevalence of respiratory disorders.
- Formulary substitutions and the use of generic formulations can lead to substantial cost savings.
- Combination therapies and innovations in delivery systems offer opportunities for market growth.
- Patient compliance and regulatory frameworks are critical factors influencing market dynamics.
FAQs
Q: What is the projected growth rate of the albuterol sulfate market?
A: The market is expected to grow at a CAGR of 8.32% from 2023 to 2030, according to some reports, and at a CAGR of 7.1% from 2024 to 2031, according to others[1][3].
Q: Which region dominates the albuterol sulfate inhalation aerosol market?
A: North America dominates the market, accounting for 40% of the global market share due to its robust healthcare infrastructure and high prevalence of respiratory disorders[1].
Q: How can formulary substitutions impact costs?
A: Formulary substitutions, such as switching from MDIs to nebulization solutions, can result in significant cost savings, despite additional costs for equipment and personnel[2].
Q: What factors influence the cost of albuterol sulfate?
A: The cost can vary based on insurance coverage, treatment plan, pharmacy used, and whether the medication is in brand-name or generic form[5].
Q: What are some opportunities for market growth in the albuterol sulfate sector?
A: Opportunities include combination therapies, innovations in delivery systems, and patient education programs that promote respiratory health awareness[3][4].
Sources
- Exactitude Consultancy, "Albuterol Sulfate Inhalation Aerosol Market Trend Shows Outstanding Growth at a CAGR of 8.32% by 2030," EIN Presswire, April 2, 2024.
- American Society of Health-System Pharmacists, Inc., "Financial effect of converting ipratropium-albuterol therapy from metered-dose inhalers to nebulization solution," PubMed, February 1, 2016.
- Market Research Intellect, "Global Albuterol Sulfate Inhalation Aerosol Market Size and Projections," Market Research Intellect.
- Data Bridge Market Research, "Global Albuterol Market – Industry Trends and Forecast to 2030," Data Bridge Market Research.
- Medical News Today, "Albuterol cost 2024: Coupons and more," Medical News Today, May 25, 2021.